Surgery, Gastroenterology and Oncology

|
|
Remnant gastric cancer (RGC) is a rare but clinically significant malignancy that develops in the remaining part of the stomach after partial gastrectomy. In terms of diagnosis, therapy, and prognosis, it represents special difficulties. Although the precise cause of RGC is still unknown, several factors have been identified as potential risk factors, including Helicobacter pylori infection, prior gastric surgery, postoperative complications, and patient-related factors. Recent studies have given insight into certain genetic mutations, chromosomal abnormalities, and molecular pathways related to the pathogenesis of RGC, which involves complex molecular modifications. Despite improvements in diagnostic methods, including molecular markers and imaging modalities, RGC is frequently identified at an advanced stage with a bad prognosis. Multidisciplinary collaboration and further research efforts are necessary to improve our understanding of RGC and develop effective management strategies to enhance patient outcomes.
Introduction
Gastric cancer is considered the fourth most common malignancy worldwide. Although advances in cancer diagnosis and treatment have significantly improved the prognosis for gastric cancer, it is still the second most common reason for cancer-related mortality globally (1-3). After a gastrectomy, a remnant gastric cancer (RGC) carcinoma can develop in the stomach's remaining tissue. RGC was initially noted by Balfour in 1922 without a specific nomenclature as stomach cancer occurring in patients who had gastric ulcer surgery. Since Balfour's publication, carcinoma resulting from a stomach remnant after a gastrectomy surgery due to gastric cancer has also been included in RGC (4-5). However, its definition and nomenclature were not standardized for a long time. Some researchers defined RGC as stomach cancer detected more than five years after surgery. In contrast, it was characterized by other researchers as gastric cancer detected more than ten years following cancer surgery (6). RGC is currently defined as carcinoma arising from the remaining stomach tissue, regardless of the initial disease or duration following prior surgery (7).
Many papers have outlined the characteristics of gastric cancer to this point, but only a small number of published studies have focused on RGC. There is still a need for a comprehensive study of RGC worldwide. Additionally, a systematic review and meta-analysis have not yet been used to analyze the characteristics and follow-up results of RGC. This study aims to deliver an updated understanding of the most recent studies on RGC as well as existing issues and concerns.
Remnant gastric cancer clinicopathological features
The incidence of remnant gastric cancer is 1-3% and is more common in men than in women (the ratio is 3-5:1) (8). Depending on the underlying surgical reason. There may be a delay before the diagnosis of remnant stomach cancer is made: After surgery for a malignant disease, the recovery time is typically shorter (around 10 years) compared to the following gastrectomy for a benign disease (approximately 30 years). It has been suggested that this variation in the interval by initial disease results from differences in carcinogenesis (9). RGC development in the remnant stomach in initial benign diseases is caused by environmental factors, including mucosa denervation following gastrectomy and continuous activation by bile reflux. Additionally, gastrojejunostomy probably has more incidence of remnant gastric cancer than gastroduodenostomy reconstruction. As a consequence of the anatomical changes after the first surgery, the lymphatic outflow from the remnant stomach differs depending on whether a gastroduodenostomy or gastrojejunostomy was performed (9-10).
Additionally, the reason for the first surgery has an impact on lymphatic drainage because the benign disease would have required less or no lymph node dissection. In contrast, the malignant disease would have required a more extensive lymphadenectomy. As a result, the surgical technique should be differentiated based on the reconstruction type and the cause of the previous surgery (11).
Cancer in the remnant tissue of the stomach might metastasize to the splenic veins, short gastric vessels, superior mesenteric vein, and hepatoduodenal ligament post-gastroduodenostomy. The drainage of lymphatics post-gastrojejunostomy is comparable to gastroduodenostomy, allowing the malignancy to metastasize to the short gastric and splenic vessels (12). However, the lymphatic outflow is different post-gastrojejunostomy because it can also penetrate through the anastomosis site and reach the mesentery of the jejunum. Therefore, cancer metastasis of the remnant stomach can go into the lymphatic system surrounding the original residual arteries when the right gastric, right gastroepiploic, and left gastric vessels are kept following the prior operation (properly post-surgery of benign condition) (13). Because of this, all D2 lymphadenectomy for gastric cancer has to be excised even if there is no clear D2 lymphadenectomy for remnant stomach cancer (fig. 1). Additionally, careful dissection of the lymph node in this region is necessary because the lymphatics near the splenic arteries represent the remnant stomach's primary lymphatic supply. Similar to the upper third of stomach cancer, routine splenectomy is not advised; however, complete lymphadenectomy is recommended for completion of total gastrectomy for remnant gastric cancer and spleen-preserving surgery (14).
Figure 1 - (a) The flow of lymphatics after gastroduodenostomy in remnant gastric cancer.

(b) The flow of lymphatics after gastrojejunostomy in remnant gastric cancer.
Pathogenesis of remnant gastric cancer
In contrast to normal gastric mucosa, experimental research has shown that the remnant stomach has a higher risk of developing gastric cancer. However, epidemiological investigations have yielded no solid findings (9). In cohort studies prospectively, specific papers from the Western area have found a link between the presence of a remnant stomach following distal gastrectomy for the disease of peptic ulcer and a risk of developing stomach cancer. In contrast, papers from Japanese have found the opposite. These differences might be caused by the variance in the prevalence of primary stomach cancer among the population; Japan has one of the highest prevalence rates of primary stomach cancer worldwide (15). In order to understand the development of newly developed RGC, authors have looked into RGC after distal gastrectomy for benign diseases. According to studies, denervation of the gastric mucosa and persistent damage caused by duodenogastric reflux are the two leading causes of changes in the remnant gastric mucosa after surgery (16).
Over the past two decades, gastrectomy for benign disease has declined due to advancements in medication therapy for duodenal and stomach ulcers. However, because carcinogenesis requires a long
latency period after the first surgery, the RGC has not decreased. RGC following distal gastrectomy for stomach cancer, however, is becoming more common (17). The clinicopathological differences between gastric cancer and those after RGC after benign disease have been shown in various studies. In these studies, RGC was seen more at the anastomotic site in Billroth-II (B-II) reconstruction than in Billroth-I(B-I) reconstruction. The likely cause of this difference is that after B-II reconstruction, the anastomotic location experiences ongoing bile reflux, which causes inflammation and regeneration of mucosa. According to studies, the average time between the initial DG and the second surgery for RGC is 6.8–18.8 years for stomach cancer and 22–34.6 years for benign disease (8,18). The continuous stimulation caused by duodenogastric reflux is assumed to cause the benign group's prolonged RGC growth period. It has been shown that cellular proliferation is sequentially activated at the anastomotic location. Because patients are continuously monitored with endoscopic examination and already have some precancerous abnormalities, such as intestinal metaplasia and atrophic gastritis, the time between RGC and DG for gastric cancer is shorter (11).
Treatment of remnant gastric cancer
Management of primary gastric cancer in early stages, endoscopic resection techniques are frequently used, including endoscopic mucosal resection and endoscopic submucosal dissection. For endoscopic resection of gastric cancer, which solely entails excision of the primary tumor and does not need lymph node dissection, no metastases of lymph nodes are a requirement (19). A thorough analysis of the pathologic findings from surgical specimens was used to determine any recommendations for this treatment. In order to create a risk stratification method for lymph node metastases, Gotoda et al. examined the histo-pathological data of 5265 patients after gastrectomy with lymphadenectomy for early-stage gastric cancer. These findings led to the endoscopic resection of patients with a low chance of metastases to the lymph nodes. However, because of the rarity of this form of tumor, it is still not clear if endoscopic resection is practical and effective for remnant gastric cancer (20).
Additionally, endoscopic resection could be difficult because of the restricted area and fibrotic alterations near the site of the anastomosis. Recent investigations have demonstrated that endoscopic resection for RGC is possible despite these challenges. Similar guidelines for endoscopic excision of stomach cancer may also apply to remnant gastric cancer, it has been claimed. Endoscopic resection may be an option for treating remnant gastric cancer with a low risk of lymphatic metastases, especially in high-risk patients, given the high degree of difficulty associated with completing a total gastrectomy for the disease due to adhesions, fibrosis, and altered anatomy (21).
Technically complex surgery is required to remove remnant stomach cancer because of adhesions, fibrosis, and changed anatomy from previous surgery. As a result, for RGC, minimally invasive surgery (such as robotic or laparoscopic surgery) is challenging, and extensive surgical skills, experience, and a comprehensive understanding of the anatomy are essential. Nevertheless, skilled laparoscopic surgeons have attempted this improbable surgery. They have reported that it can be done safely and successfully with mortality and morbidity rates comparable to open technique (22).
Fluorescent guided lymphadenectomy in remnant gastric cancer
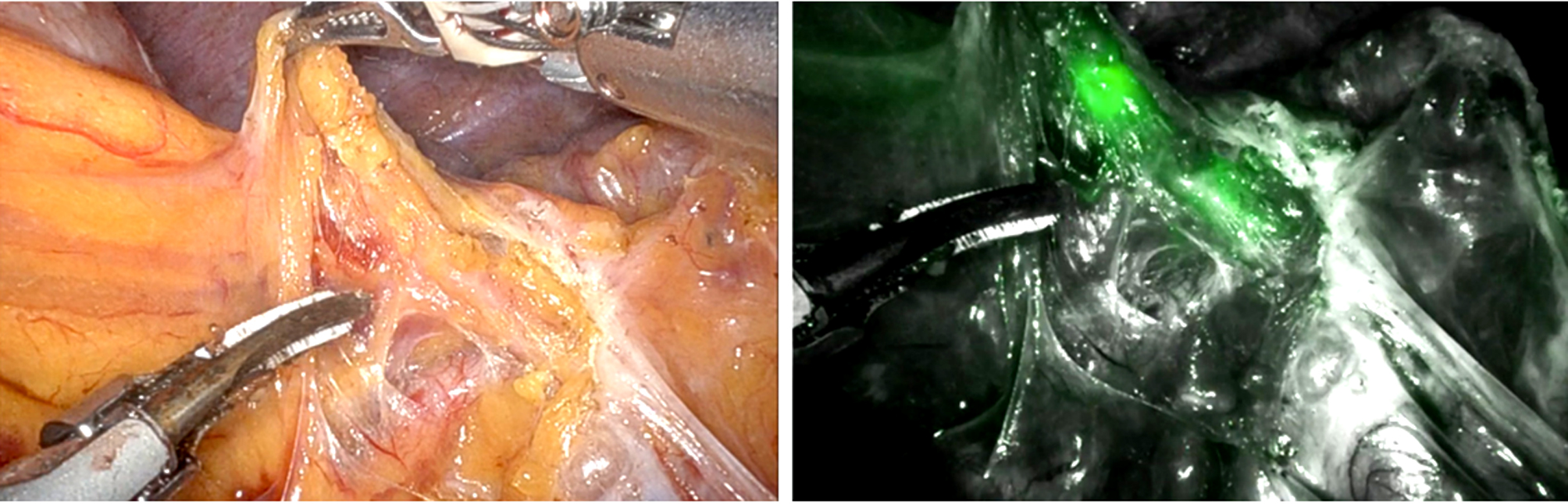
During minimally invasive gastric cancer surgery for lymphatic identification, fluorescent lymphography (FL) utilizing indocyanine green under near-infrared imaging is considered the most effective tool for lymph node identification (23). The splenic hilar area presents the most difficulties when undergoing lymphadenectomy during total gastrectomy beyond the stomach. A relatively high therapeutic index and rates of splenic hilar lymph node metastases of 14.1% and 19.2% in the RGC following partial gastrectomy for malignant and benign disease, respectively, confirm the recommendations for splenic hilar area dissection (24). Because fluorescent lymph nodes would be easily visible in the dissected area and the chance of leaving lymph nodes there would be minimized, FL allowed for the intraoperative evaluation of the lymphadenectomy's quality (25). FL is also recommended when fluorescence is present as a helpful tool to perform good-quality lymphadenectomy near the splenic hilum during complete gastrectomy for primary gastric cancer (fig. 2). FL for RGC is a potential method for completion gastrectomy because it allows for complete splenic hilar area dissection (26-27).
Figuri 2 - (a) Distal splenic artery area in remnant gastric cancer, white-light image; (b) fluorescent LNs along the distal splenic artery, fluorescent-light image
Treatment outcomes of remnant gastric cancer
Several papers have reported the outcomes of the completion of total gastrectomy for stomach cancer that was still present. Mortality rates following surgery have ranged from 0% to 12.5%, while postoperative morbidity rates have ranged from 20% to 40%. Interestingly, most of this research was in Eastern countries, including Korea, Japan, and China. The study from the Western countries found that mortality was reasonably high. Still, the data was too old to reach any conclusions about differences in mortality following completion of total gastrectomy between the East and the West. Overall, it appears that the morbidity and mortality rates following the completion of total gastrectomy for remnant gastric cancer are generally comparable to those reported for primary gastriccancer (28). A few papers have compared the outcomes results of primary vs. remnant gastric cancer surgery. In these publications, results are comparable following both types of surgery (29). It is debatable if the
prognosis of remnant stomach cancer in the upper part of the stomach is worse than that of primary stomach cancer. The results of earlier research were highly varied, according to an updated systematic review that used analysis to examine this issue—compared to the outcomes following upper-third primary stomach cancer, remnant gastric cancer had the same prognosis in the early stages 1/2. Still, it was worse in the advanced stages III/IV. The cause behind these results is still unclear. Hence, they cannot be regarded as definitive. However, they stress the significance of early gastric cancer remnant diagnosis (30).
Discussion
Clinically, remnant gastric cancers include a variety of malignancies that resemble primary gastric cancers, tumors brought on by bile reflux-related chronic inflammation, and additional cancers with unknown etiologies. Our present understanding of primarygastric cancer and the various causes of remnant gastric cancer results in diverse clinical responses to traditional therapies. Additionally, post-distal gastrectomy cancer in the remnant stomach has become increasingly common in recent years (31). For patients with early-stage gastric cancer, function-preserving gastrectomy (proximal gastrectomy) has recently gained popularity, and survivors who underwent this kind of surgery are increasingly common. If we consider that Korea and Japan account for two-thirds of all cases of stomach cancer, remnant gastric cancer in the lower part of the stomach following proximal gastrectomy for gastric cancer is predicted to become more common (32). Surgery; as a result, the future of remnant stomach cancer could be changed. However, it is uncertain whether this remnant gastric cancer resembles initial gastric cancer. This is yet another ambiguous matter. Despite the complexity indicated above, radical surgery is the only therapy option for residual gastric cancer that can truly cure the disease (33). However, radical surgery for stomach cancer remnants is technically challenging due to adhesions and fibrosis from the prior operation and to changes in the anatomy, including abnormalities in the lymphatics. Therefore, surgeons have to enhance their surgical expertise and skills to address the difficult condition known as remnant gastric cancer, which exists today and will in the future. Furthermore, the significance of adhering to oncologic principles for oncology surgery in all surgeries involving remnant stomach cancer cannot be overstated (34-35).
Conclusion
Clinically, gastric remnant cancer is not a very uncommon issue. RGC's clinicopathological features raise similar problems as those with primary gastric cancer. To better understand the RGC characteristics, more samples and studies are required. The best
surgical oncological approach is the completion
of total gastrectomy with fluorescence-guided
lymphadenectomy.
Conflict of interest
The author declares that they have no conflicts ofinterest.
Funding
No funding sources.
References
1.Iwu CD, Iwu-Jaja CJ. Gastric Cancer Epidemiology: Current Trend and Future Direction. Hygiene. 2023;3(3):256-268.
2.Choi Y, Kim N, Yoon H, Shin CM, Park YS, Lee DH, et al. The Incidence and Risk Factors for Metachronous Gastric Cancer in the Remnant Stomach after Gastric Cancer Surgery. Gut Liver. 2022;16(3):366-374.
3.Rawla P, Barsouk A. Epidemiology of gastric cancer: global trends, risk factors and prevention. Prz Gastroenterol. 2019;14(1):26-38.
4.Vaz Safatle-Ribeiro A, Ribeiro U, Sakai P, Iriya K, Ishioka S, Gama-Rodrigues J. Gastric stump mucosa: is there a risk for carcinoma? Arq Gastroenterol. 2001; 38(4):227-31.
5. Shimada H, Fukagawa T, Haga Y, Oba K (2016) Does remnant gastric cancer really differ from primary gastric cancer? A systematic review of the literature by the Task Force of Japanese Gastric Cancer Association. Gastric Cancer. 2016;19(2):339-349.
6.Dhir M. Gastric Remnant Cancer: Is it different From Primary Gastric Cancer? Insights Into a Unique Clinical Entity. Ann Surg Oncol. 2020;27(11):4079-4081.
7.Mak TK, Guan B, Peng J, Chong TH, Wang C, Huang S, et al. Prevalence and characteristics of gastric remnant cancer: a systematic review and meta-analysis. Asian J Surg. 2021;44(1): 11-17.
8.Park YE, Kim SW. Clinicopathologic features of remnant gastric cancer after curative distal gastrectomy according to previous reconstruction method: A retrospective cohort study. World J Surg Oncol. 2019;17(1):203.
9.Ohira M, Toyokawa T, Sakurai K, Kubo N, Tanaka H, Muguruma K, et al. Current status in remnant gastric cancer after distal gastrectomy. World J Gastroenterol. 2016;22(8):2424-33.
10.Chen D, Tang C, He F, Yang F, Woraikat S, Qian K, et al. Comparison of Billroth II with Braun and Roux-en-Y reconstructions after distal gastrectomy for gastric cancer: a meta-analysis. MedComm-Oncology. 2023;2:e48.
11.Kondo K. Duodenogastric reflux and gastric stump carcinoma. Gastric Cancer. 2002;5(1):16-22.
12.Xu ZY, Hu C, Chen S, Du YA, Huang L, Yu PF, et al. Evaluation of D2-plus radical resection for gastric cancer with pyloric invasion. BMC Surg. 2019; 19(1):172.
13.Kawamoto A, Kimura K, Hirose K, Izumi T, Taniguchi D, Yano H, et al (2022) Synchronous gallbladder metastasis originating from residual gastric cancer: a case report and the review of literature. Surg Case Rep. 2022;8(1):88.
14.Takahashi T, Inaki N, Saito H, Sakimura Y, Hayashi K, Tsuji T, et al. Laparoscopic Completion total Gastrectomy as an Alternative Procedure for Gastric Stump Cancer: A Case Control Study. Research Square 1-13.
15.Paragomi P, Dabo B, Pelucchi C, Bonzi R, Bako AT, Sanusi NM, et al. The Association between Peptic Ulcer Disease and Gastric Cancer: Results from the Stomach Cancer Pooling (StoP) Project Consortium. Cancers (Basel). 2022;14(19):4905.
16.Hwang JJ, Lee DH, Lee AR, Yoon H, Shin CM, Park YS, et al. Characteristics of gastric cancer in peptic ulcer patients with Helicobacter pylori infection. World J Gastroenterol. 2015;21(16): 4954-60.
17.Lee CW, Sarosi GA, Jr. Emergency ulcer surgery. Surg Clin North Am. 2011;91(5):1001-13.
18.Parthasarathy S, Radhakrishna K, Kalawat T, B. V. Phaneendra3, Venkatarami Reddy Vutukuru1. Prospective randomised study comparing Billroth II with Braun anastomosis versus Roux-en-Y reconstruction after radical distal gastrectomy for gastric cancer. J Clin Sci Res. 2020;9(3):150–154.
19.Vasconcelos AC, Dinis-Ribeiro M, Libânio D. Endoscopic Resection of Early Gastric Cancer and Pre-Malignant Gastric Lesions. Cancers (Basel). 2023;15(12):3084.
20.Abdelfatah MM, Barakat M, Lee H, Kim JJ, Uedo N, Grimm I, et al. The incidence of lymph node metastasis in early gastric cancer according to the expanded criteria in comparison with the absolute criteria of the Japanese Gastric Cancer Association: a systematic review of the literature and meta-analysis. Gastrointest Endosc. 2018;87(2):338-347.
21.Ko WJ, Song GW, Kim WH, Hong SP, Cho JY. Endoscopic resection of early gastric cancer: Current status and new approaches. Transl Gastroenterol Hepatol. 2016;1:24. eCollection 2016.
22.Parisi A, Reim D, Borghi F, Nguyen NT, Qi F, Coratti A, et al. Minimally invasive surgery for gastric cancer: a comparison between robotic, laparoscopic and open surgery. World J Gastroenterol. 2017; 23(13):2376-2384.
23.Jung MK, Cho M, Roh CK, Seo WJ, Choi S, Son T, et al (2021) Assessment of diagnostic value of fluorescent lymphography-guided lymphadenectomy for gastric cancer. Gastric Cancer. 2021; 24(2):515-525.
24.Alrashidi N, Kim KY, Park SH, Lee S, Cho M, Kim YM, et al. Fluorescent Lymphography-Guided Lymphadenectomy during Minimally Invasive Completion Total Gastrectomy for Remnant Gastric Cancer Patients. Cancers (Basel). 2022;14(20):5037.
25.Liu M, Xing J, Xu K, Yuan P, Cui M, Zhang C, et al. Application of near-infrared fluorescence imaging with indocyanine green in totally laparoscopic distal gastrectomy. J Gastric Cancer. 2020;20(3):
290-299.
26.Chen QY, Xie JW, Zhong Q, Wang JB, Lin JX, Lu J, et al. Safety and Efficacy of Indocyanine Green Tracer-Guided Lymph Node Dissection during Laparoscopic Radical Gastrectomy in Patients with Gastric Cancer: A Randomized Clinical Trial. JAMA Surg. 2020;155(4):300-311.
27.Cianchi F, Indennitate G, Paoli B, Ortolani M, Lami G, Manetti N, et al. The Clinical Value of Fluorescent Lymphography with Indocyanine Green During Robotic Surgery for Gastric Cancer: a Matched Cohort Study. J Gastrointest Surg. 2020;24(10):2197-2203.
28.Degiuli M, Sasako M, Calgaro M, Garino M, Rebecchi F, Mineccia M, et al (2004) Morbidity and mortality after D1 and D2 gastrectomy for cancer: Interim analysis of the Italian Gastric Cancer Study Group (IGCSG) randomised surgical trial. Eur J Surg Oncol. 2004;30(3): 303-8.
29.Huang H, Wang W, Chen Z, Jin JJ, Long ZW, Cai H, et al. Prognostic factors and survival in patients with gastric stump cancer. World J Gastroenterol. 2015;21(6):1865-71.
30.Li F, Zhang R, Liang H, Quan J, Liu H, Zhang H. Gastric remnant cancer patients had a better prognosis than upper-third gastric cancer patients in a case-control study after surgical treatment. Tumori. 2013;99(4):510-5.
31.Lee HC, Huang KH, Yeh TS, Chi CW. Somatic alterations in mitochondrial DNA and mitochondrial dysfunction in gastric cancer progression. World J Gastroenterol. 2014;20(14):3950-9.
32.Hiramatsu Y, Kikuchi H, Takeuchi H. Function-Preserving Gastrectomy for Early Gastric Cancer. Cancers (Basel). 2021;13(24): 6223.
33.Japanese Gastric Cancer Association (2021) Japanese gastric cancer treatment guidelines 2018 (5th edition). Gastric Cancer. 2021; 24(1):1-21.
34. Beyer K. Surgery Matters: Progress in Surgical Management of Gastric Cancer. Curr Treat Options Oncol. 2023;24(2):108-129.
35.Choi YY, Noh SH. Surgery for Remnant Gastric Cancer: Open Surgery. In: Noh S, Hyung W (eds). Surgery for Gastric Cancer. Berlin, Heidelberg: Springer; 2019.
Full Text Sources:
Abstract:
Views: 5127

For Authors
Journal Subscriptions

Dec 2025
Supplements
Instructions for authors
Online submission
Contact
e-ISSN: 2601 - 1700 (online)
ISSN-L: 2559 - 723X
Journal Abbreviation: Surg. Gastroenterol. Oncol.
Surgery, Gastroenterology and Oncology (SGO) is indexed in:
- SCOPUS
- EBSCO
- DOI/Crossref
- Google Scholar
- SCImago
- Harvard Library
- Open Academic Journals Index (OAJI)
Surgery, Gastroenterology and Oncology (SGO) is an open-access, peer-reviewed online journal published by Celsius Publishing House. The journal allows readers to read, download, copy, distribute, print, search, or link to the full text of its articles.
Time to first editorial decision: 25 days
Rejection rate: 61%
CiteScore: 0.2

Meetings and Courses in 2025
Meetings and Courses in 2024
Meetings and Courses in 2023
Meetings and Courses in 2022
Meetings and Courses in 2021
Meetings and Courses in 2020
Meetings and Courses in 2019
Verona expert meeting 2019

Surgery, Gastroenterology and Oncology applies the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits readers to copy and redistribute the material in any medium or format, remix, adapt, build upon the published works non-commercially, and license the derivative works on different terms, provided the original material is properly cited and the use is non-commercial. Please see: https://creativecommons.org/licenses/by-nc/4.0/
Publisher’s Note:
The opinions, statements, and data contained in article are solely those of the authors and not of Surgery, Gastroenterology and Oncology journal or the editors. Publisher and the editors disclaim responsibility for any damage resulting from any ideas, instructions, methods, or products referred to in the content.
 IASGO Society News
IASGO Society News