Surgery, Gastroenterology and Oncology

|
|
Background: Patients with abdominal cancers may present with advanced disease either locally or metastatic and so, accurate staging help in selection of the appropriate treatment plan (curative or palliative). Up to 50% of patients diagnosed with colorectal cancers (CRC) and 40% of patients diagnosed with pancreatic cancers will have metastatic hepatic lesions during the course of the disease, where 7% of patents with CRC and 14% of patients with advanced gastric cancers will develop peritoneal metastases. Despite advances in pre-operative imaging used for diagnosis and staging as CT, MRI and PET-CT many patients are diagnosed either intra-operative or in early post-operative period with distant metastases. Aim of the work: The study aim is to assess and provide precise data for the efficacy and added value of using Indocyanine Green (ICG) in diagnostic staging laparoscopy in intra-abdominal malignancy, especially in the identification of hepatic malignancies or liver and peritoneal metastases.
Patients and Methods: This is a cross-sectional prospective study which was conducted on thirty patients with operable intra-abdominal malignancy presented to Theodor Bilhariz research institute from February 2022 to July 2023.
Results: In our study, total number was 12 hepatic lesions, 8 lesions were discovered during routine pre-operative investigation and 11 lesions were identified by intra-operative ICG florescence with histopathological confirmation of metastasis in 57%.
Conclusion: ICG is a safe and applicable tool for intra-operative assessment and detection of liver lesions with high sensitivity. It is not specific for malignant lesions rather than hypervascular lesions, affecting its specificity. Another study with larger sample size and dedicated post-operative follow up may be needed for more precise data.
INTRODUCTION
Patients with abdominal cancers may present with advanced disease either locally or metastatic and so, accurate staging help in selection of the appropriate treatment plan (curative or palliative). Although advanced pre-operative imaging as (trans-abdominal and endoscopic ultrasound, CT scan, MRI, and PET scan), many patients may have unexpected, unresectable disease at exploration (1).
Despite presence of advanced imaging modalities, the sensitivity of radiological evaluation in identifying metastatic liver and peritoneal lesions is limited. The accuracy of ultrasound was 76% and 79% for CT in detecting metastatic hepatic lesions (2).
US, CT scan and MRI are the cornerstones of detecting and staging of hepatic malignancies with high degree of accuracy and their preoperative use of them is helpful in a hepatic resection.
But one of their limitations is that they may miss focal lesions less than 0.5 cm and they provide still pictures only with a limited value as a diagnostic and guidance (3).
Peritoneal carcinomatosis (PC) is a challenge. Detection of peritoneal involvement at an early stage may help improving treatment response and final results (4). Present pre-operative diagnostic and staging techniques and imaging modalities have low sensitivity for PC less than 0.5 cm (5).
Staging laparoscopy (SL) is a safe and available tool that may help to increase accuracy of staging of abdominal malignancies by direct visualization of abdominal organs, take tissue biopsy and aspiration cytology, assist in appropriate treatment plan selection and decrease unnecessary laparotomies form 40% to 17% with less morbidity and mortality (6).
However, even with direct inspection during SL or laparotomy some small lesions could be missed, especially in if bowel mobilization is needed as in the pelvis (7).
Florescence imaging guided surgery (FIGS) and near-infrared (NIR) fluorescence is a newly developed technique that could be used to detect and visualize structures in real time intraoperative. ICG is tri-carbo-cyanine dye that has a molecular weight of 775 Da, a negatively charged, amphiphilic, water-soluble but relatively hydrophobic (8).
Advantages include high tissue penetration and low auto-fluorescence where the human eye is insensitive to its excitation light wavelength (doesn`t alter the surgical field). It binds to plasma protein, circulating in the blood then uptake occur by hepatocytes with rapid biliary excretion, with a high favorable safety and a lack of ionizing-radiation exposure (9).
Aim of the work
The study aim is to assess and provide precise data for the efficacy and added value of using ICG in diagnostic staging laparoscopy in intra-abdominal malignancy, especially in the identification of hepatic malignancies or liver and peritoneal metastases. Whether or not the ICG laparoscopic staging will alter the management plan, is yet to be seen?
PATIENTS AND METHODS
This is a cross-sectional prospective study which was conducted on thirty patients with operable intra-abdominal malignancy presented to Theodor Bilhariz research institute from February 2022 to July 2023.
Population of study and disease condition: 30 patients who were diagnosed with operable intra-abdominal malignancy presented to surgery department at Theodor Bilhriz Research Institute.
Inclusion criteria
Adult Patients (males or females) with operable intra-abdominal malignancy and fit for laparoscopic procedure.
Exclusion criteria
Age below 18 years, Inoperable malignancy or unfit for laparoscopic surgery, pregnancy or lactation, mentally disabled patients, renal failure and allergy to iodine, urografin or ICG.
Methodology in details
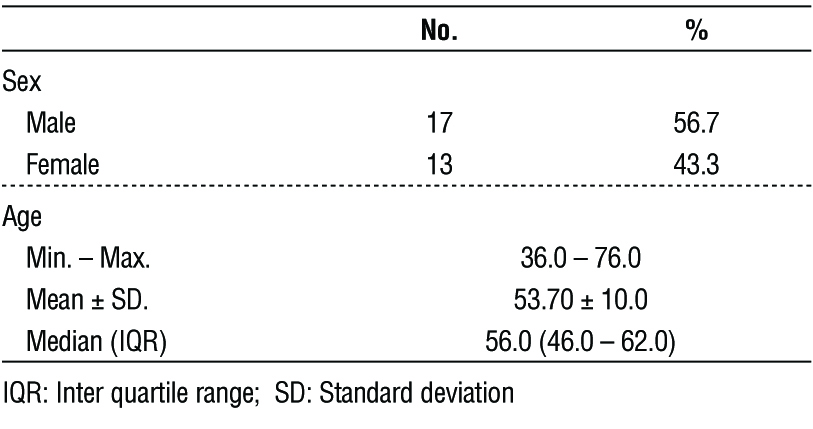
History: Detailed history including Age, Sex, presence of co-morbidities, Family history and specific history related to the complaint e.g. onset, course, duration, associations as weight loss, loss of appetite.
Examination: General: Cachexia, Jaundice.Abdominal: inspection, palpation,percussion, auscultation.
Investigations
Laboratory
Routine pre-operative labs e.g. Complete blood count (CBC), Liver functions (AST, ALT, Albumin, Bilirubin), coagulation profile (PT, INR), Kidney functions (urea, creatinine, electrolytes), Virology (HBsAg, HCV Ab, HIV Ab), Random Blood glucose level.
Tumor markers e.g.: AFP, CEA, CA 19-9, CA 125.
Imaging
For diagnosis: according to site of primary tumor: Pelvi-abdominal ultra-sound, CT scan on abdomen and pelvis with intravenous contrast ± oral, enemal contrast, MRI study for abdomen and pelvis.
For staging: Ct abdomen, chest, brain + bone Scan, or PET CT.
Endoscopic
According to the primary disease: upper GI endoscopy, colonoscopy, endoluminal ultrasound (EUS), and endoscopic retrograde cholangio-pancreaticography (ERCP).
Table 1 - Demographic distribution of the studied cases according to patient data (n = 30)
Pre-operative preparation
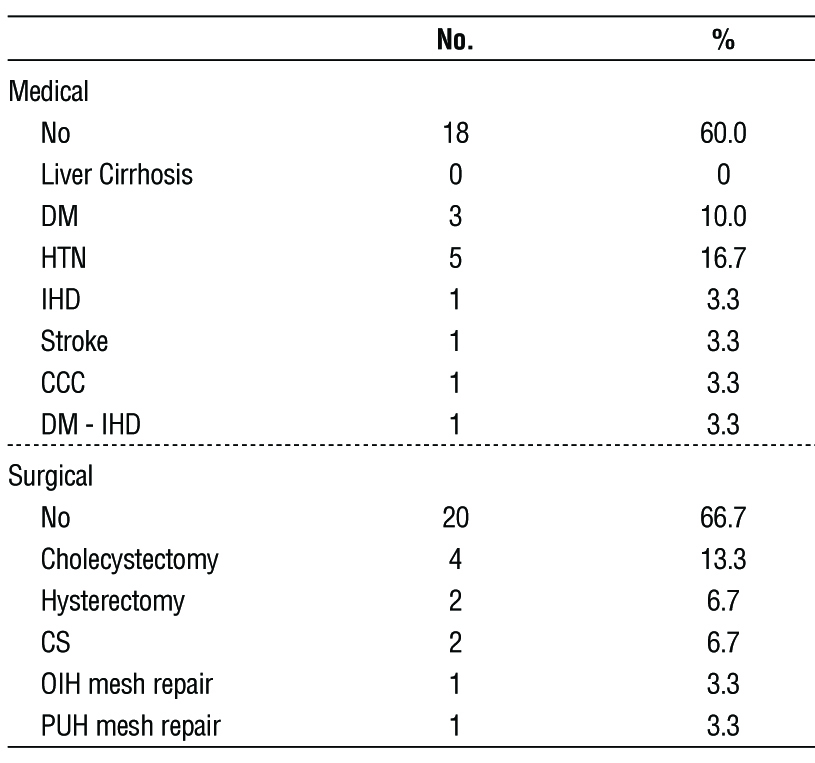
Consent: 30 patients will participate in this study. Nature and merit of the research will be explained to patients only those will accept and provide an informed written consent will be included.
In addition to standard-of-care, patients will receive an IV injection of 0.25- 0.5 mg/kg ICG 24-72 hours prior to surgery.
Intra-operative: All patients will be under general anesthesia, supine position. During surgery, patients undergo a staging laparoscopy, whereupon inspection then NIR fluorescence imaging will be performed searching for hepatic focal lesions or peritoneal
nodules. The NIR system includes light source with double filter (white and 760 nm light) and a 30° laparos-copic lens that have optical filters. The operator can easily use foot pedal to switch between usual white light and near infra-red. 10 mm trocar inserted through umbilicus and other two ports at least for allowing good exposure and completing the exploration.
The whole abdomen is visualized and inspected for suspected lesions, including the liver (right and left lobe), the parietal and visceral peritoneum, greater omentum, transverse mesocolon and the pelvis. Any suspected lesions are sampled and sent to histopathological assessment.
If no metastases are identified, the procedure will continue as planned. If metastases are suspected, the decision to resume primary cancer resection or convert to palliative or abort is up to the surgeon.
Table 2 - Distribution of the studied cases according to past history (n = 30)
Statistical analysis
The data will be analyzed using Microsoft Excel 2016 and statistical package for social science ‘IBM SPSS Statistics for Windows, version 26 (IBM Corp., Armonk, N.Y., USA)'. Continuous normally distributed variables will be represented as mean ±SD. with 95% confidence interval, while non-normal variables will be summarized as median with 25 and 75 percentiles and using the frequencies and percentage for categorical variables; a p value < 0.05 will be considered statistically significant. To compare the means of normally distributed variables between groups, the Student’s t test will be performing, and Mann-whitney U test will be used in non-normal variables. c2 test or Fisher’s exact test will be used to determine the distribution of categorical variables between groups. Spearman's rank correlation coefficient (r) will be done to show the correlation between different parameters in this study, in addition to regression analysis to evaluate the risk assessment (33).
RESULTS
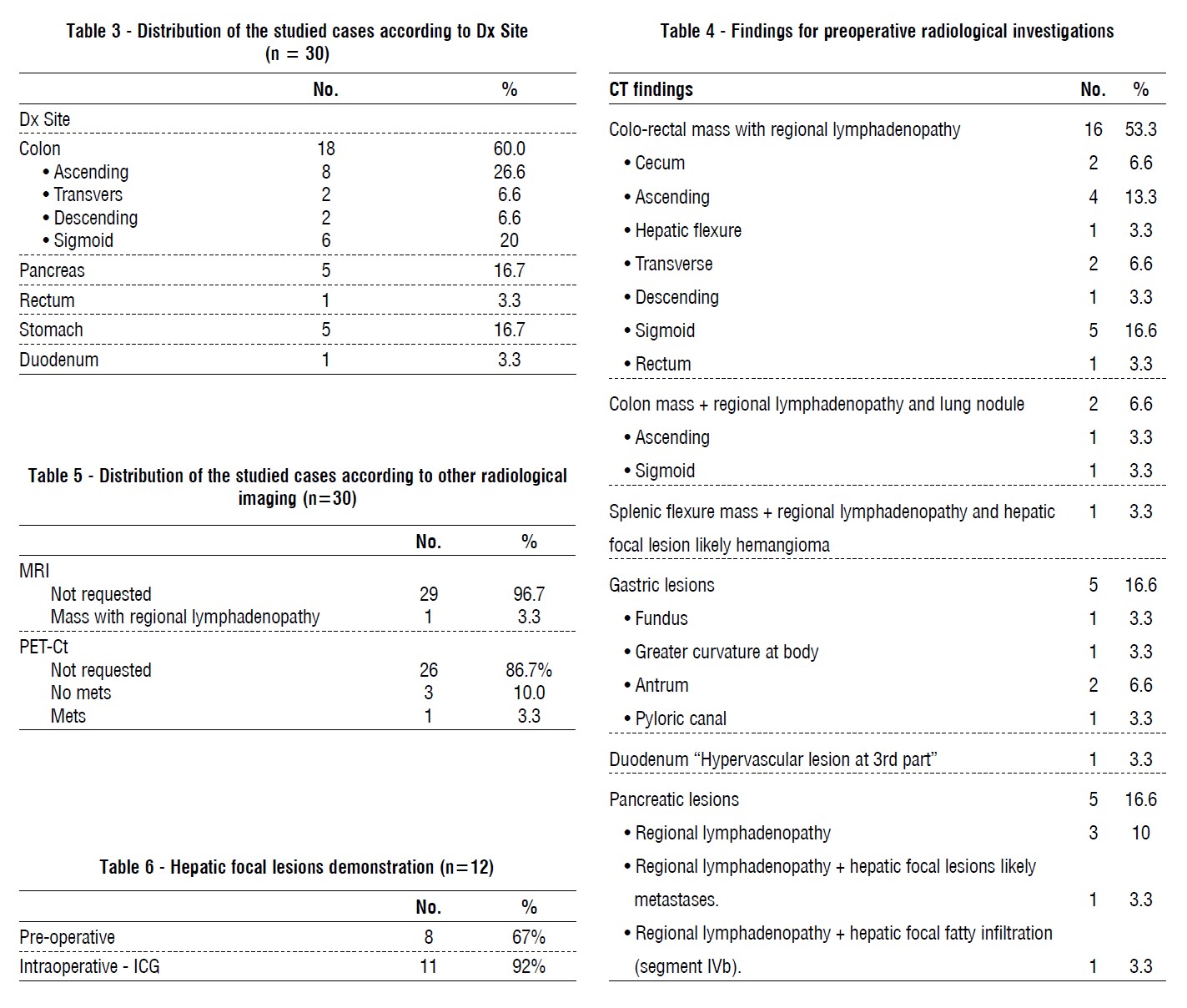
Figure 1 - Hepatic focal lesions (n = 12)
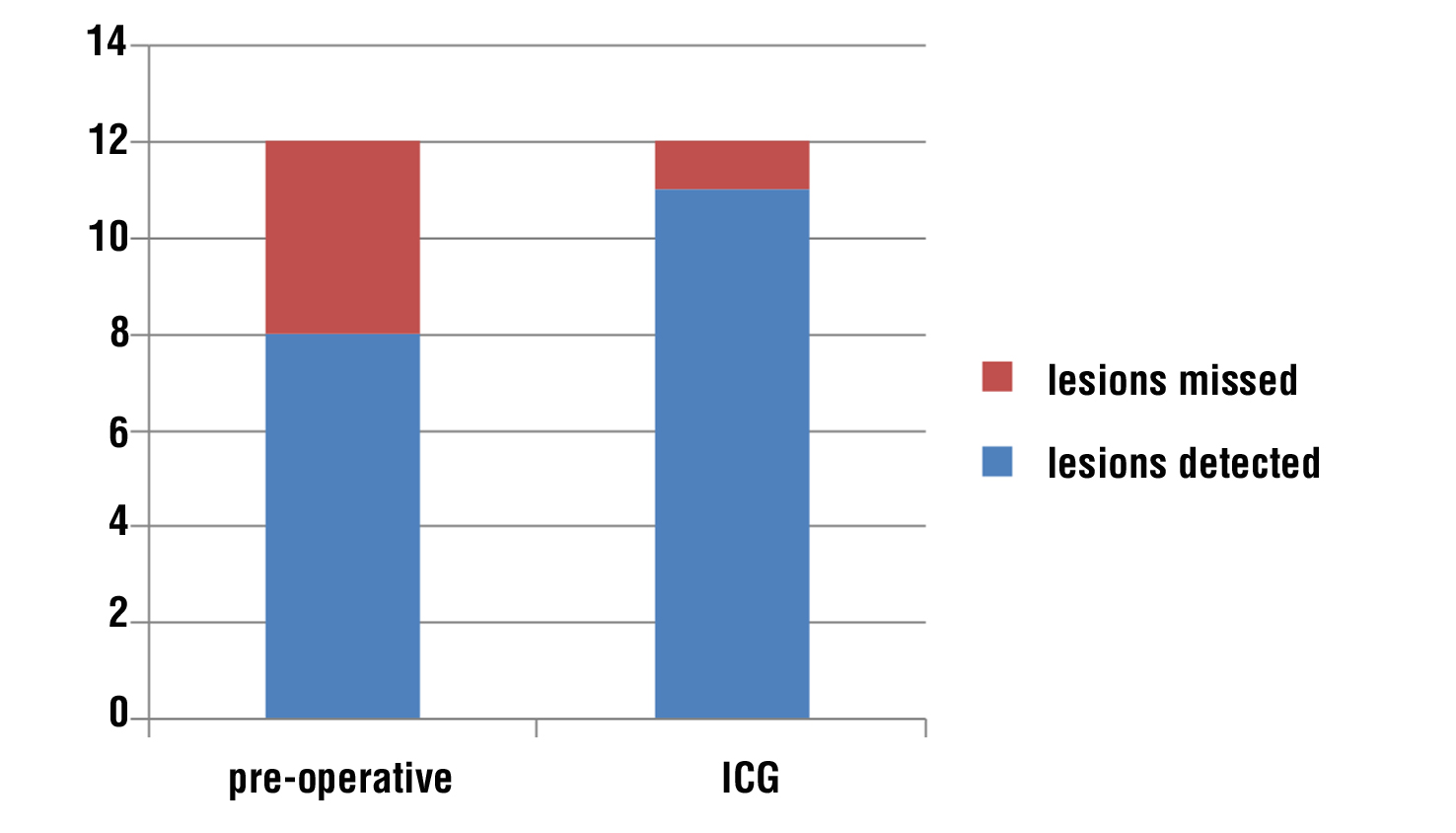
Figure 2 - Photo shows hepatic focal lesions (segment IVb) as seen in (a) CT image, (b) naked eye during staging laparoscopy and by (c) ICG auto-fluorescence
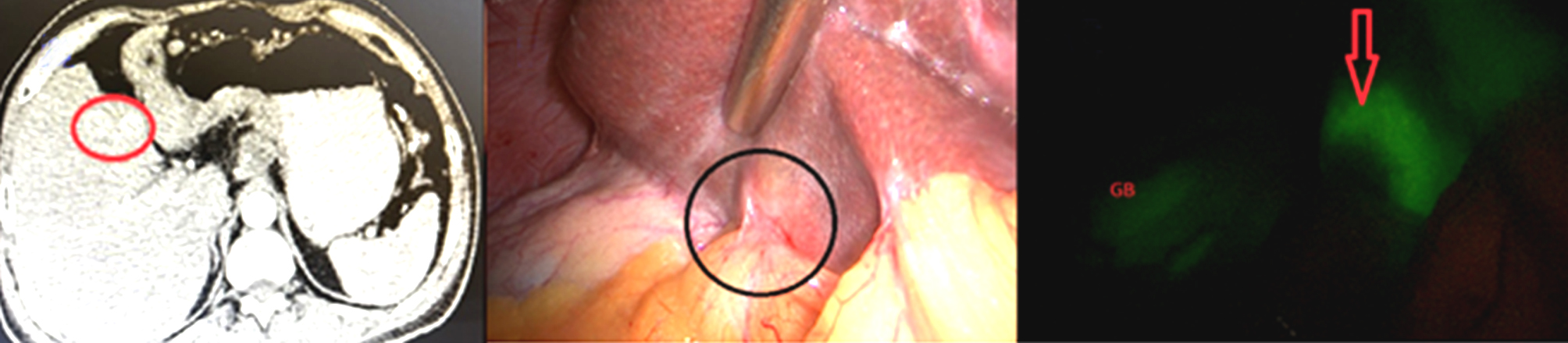
Figure 3 - Photo show segment III in (A) CT image shows only one focal lesion, while (B) naked eye during staging laparoscopy shows two focal lesions and in (C) ICG auto-fluorescence shows two hepatic focal lesions
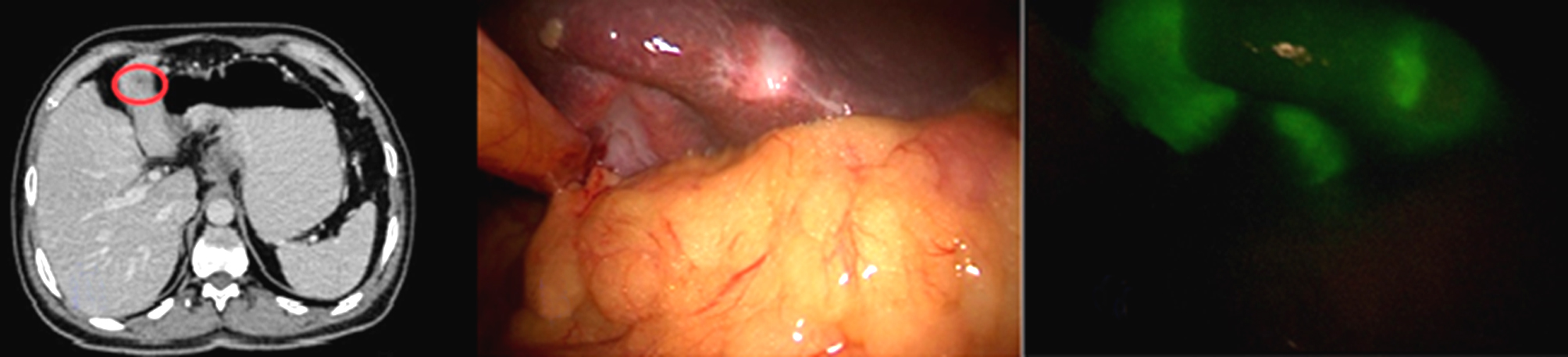
Figure 4 - Photo shows (a) segment III focal lesion by naked eye and (b) by ICG (c) segment IVb focal lesions by naked eye and (d) by ICG
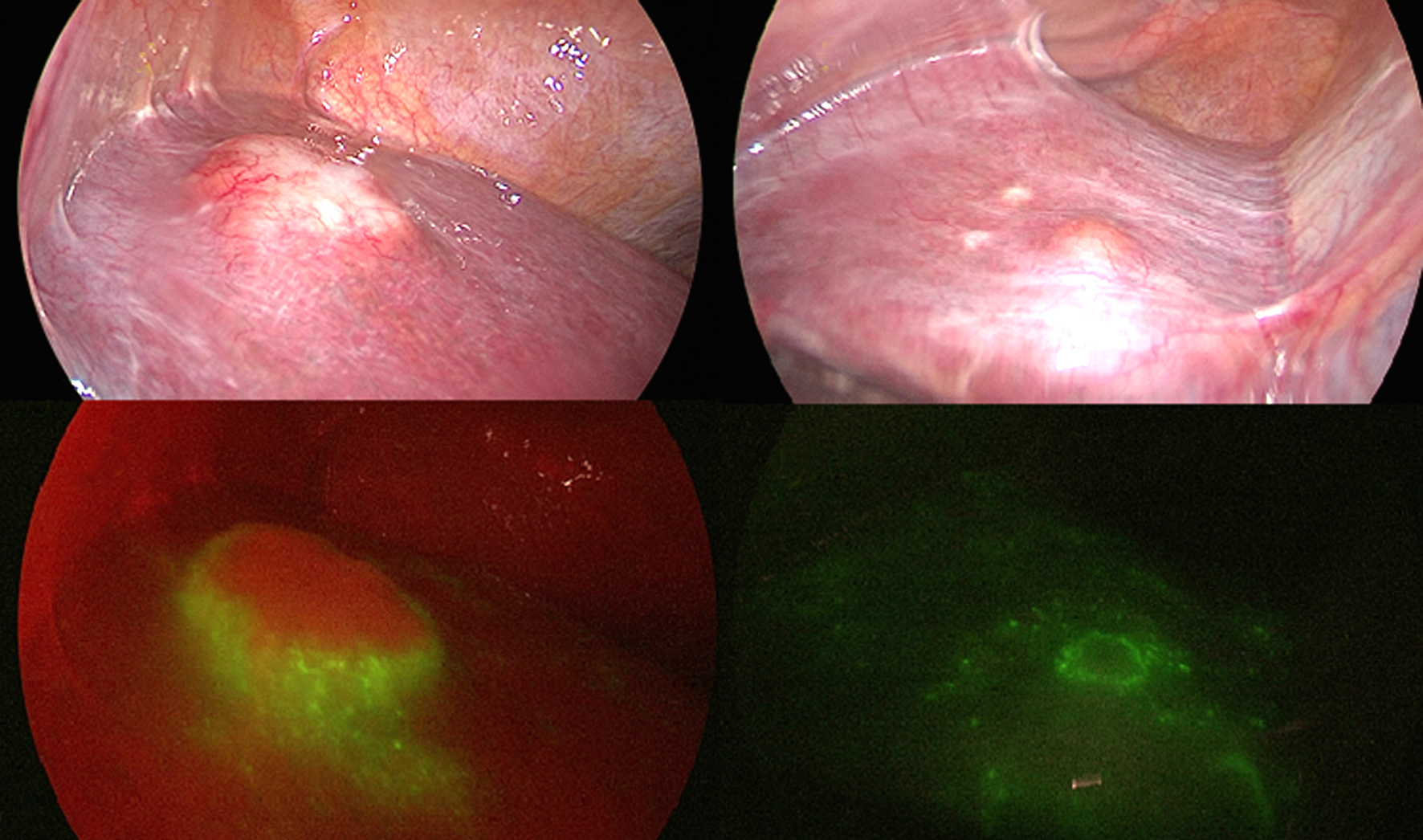
Figure 5 - Photo show (A) left liver lobe with no obvious lesions (B) left lobe of liver with no micro-lesions
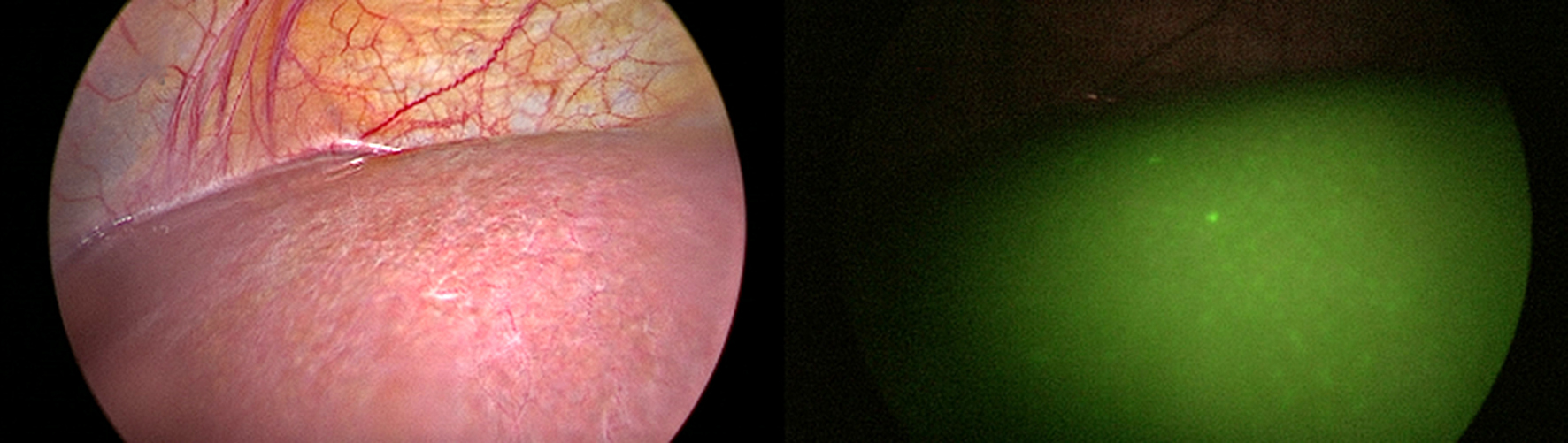
Figure 6 - Photo show (a) segment III with hepatic focal lesion and (b) the same lesion by ICG autoflorescence
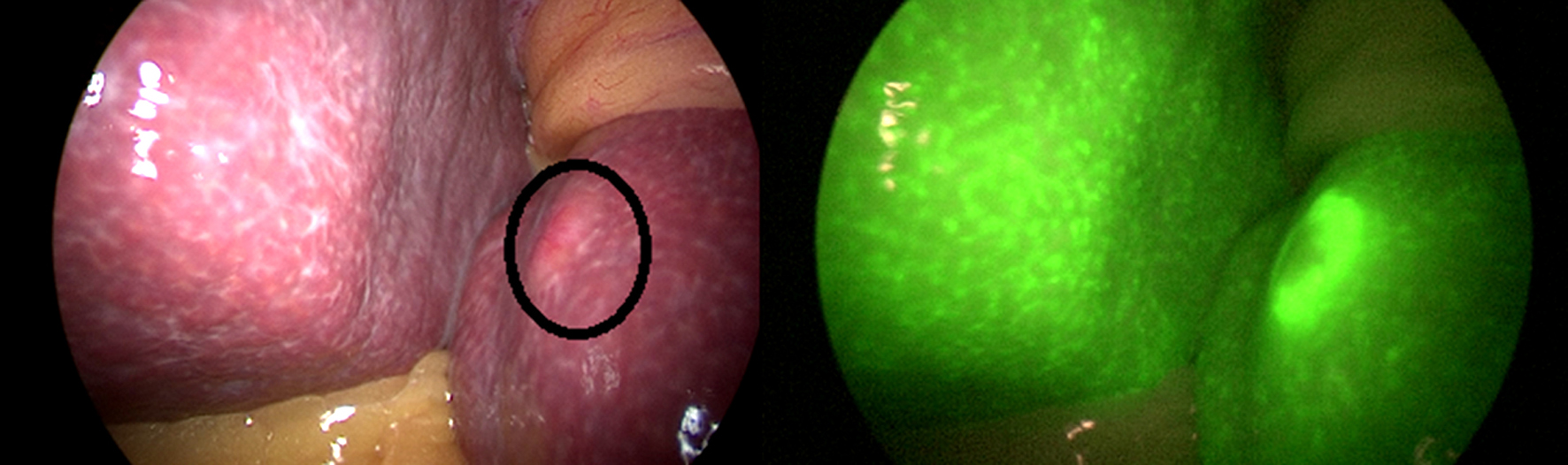
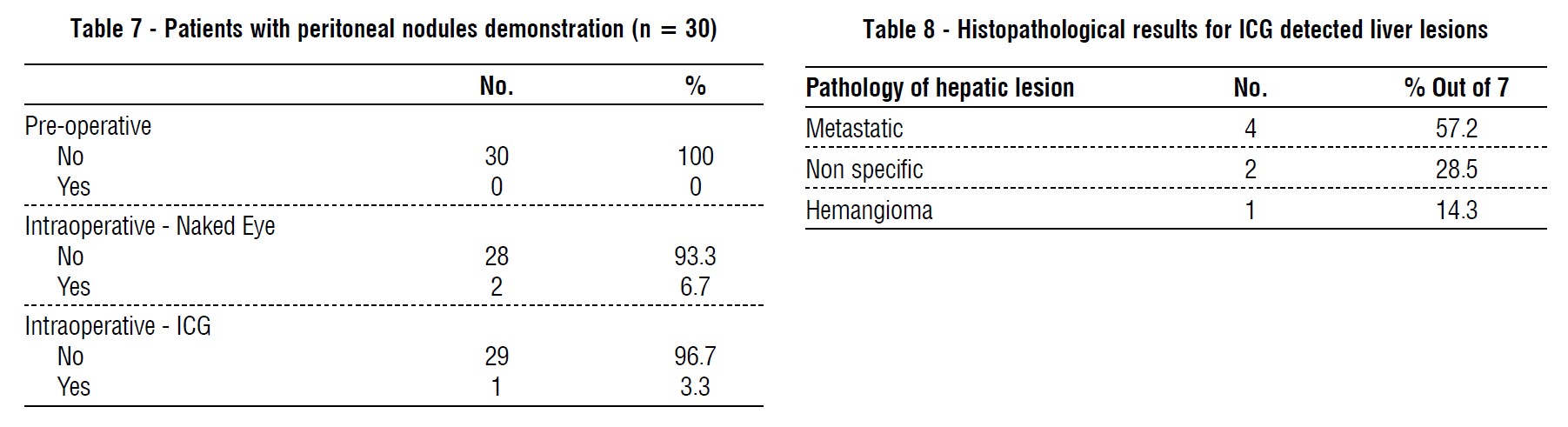
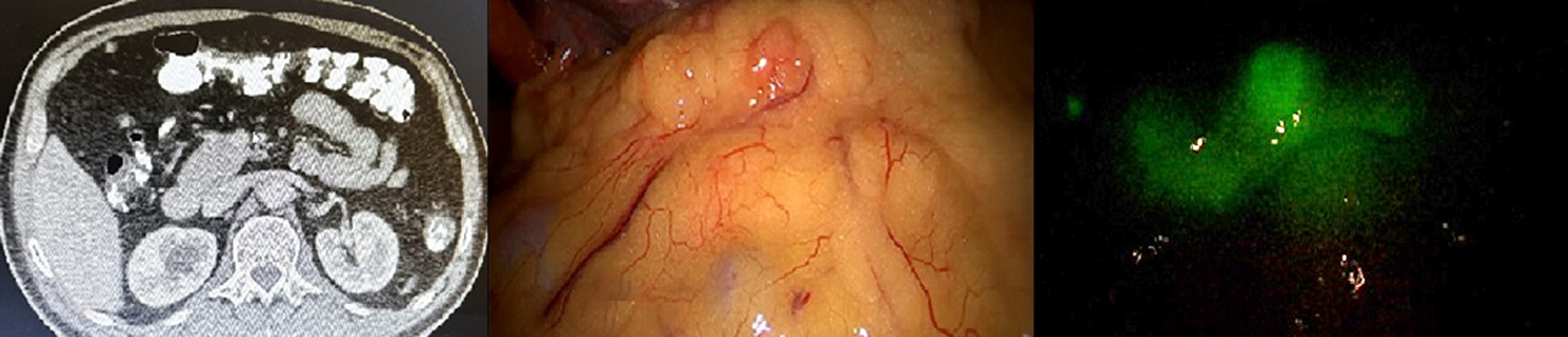
Figure 7 - Photo shows peritoneal (Omentum) nodules as seen in (a) CT image,(b) naked eye during staging laparoscopy and (c) by ICG auto-fluorescence
DISCUSSION
Patients with abdominal cancers may present with advanced disease either locally or metastatic and so, accurate staging help in selection of the appropriate treatment plan (curative or palliative) (1). Metastatic spread is the most common cause of morbidity and mortality in malignant patient, resemble about 90% (10). According to seed and soil theory liver is the commonest site for metastatic spread especially from gastrointestinal tumors up to about 30-50% of patients with CRC will have metastatic hepatic lesions during the course of the disease and about 35% in patients with pancreatic cancers (11).
Transcoelomic mechanism through shedding of malignant cells is the theory suggested for peritoneal carcinomatosis and metastatic nodules, where 7% of patents with CRC and 14% of patients with advanced gastric cancers will develop peritoneal metastases (12). Despite advances in preoperative imaging used for diagnosis and staging as U/S, CT scan, MRI, and PET-CT scan, many patients are diagnosed either intra-operative or in early post-operative period with distant metastases (2).
Diagnostic laparoscopy (DL) is a less invasive technique help in intra-abdominal lesions diagnosis. It enables visualization and examination of large surface areas of intraabdominal organs and deep parts that cannot be easily reached by laparotomies. Facilitate biopsy specimen taking, use laparoscopic ultrasound, fluorescence imaging and may be therapeutic. It’s associated with less post-operative pain, less morbidity and decreases the need of unnecessary laparotomy from 40 to 17% (13). However, even with direct inspection during SL or laparotomy some small lesions could be missed, especially in if bowel mobilization is needed as in the pelvis (7).
Fluorescence image is used as a color code for delineation of malignant tissues, confirm sentinel lymph nodes, detecting early peritoneal disease and metastasis, decrease recurrence rate (14). ICG is a negatively charged tri-carbocyanine dye which has a high safety profile, with very low risk for anaphylaxis and cross-reactions with iodine sensitivity (1:10000). It is excreted unchanged in bile and not absorbed by intestinal mucosa diminishing risk of toxicity (15).
ICG binds to plasma proteins which extravasate through vascular neogenesis and hyper permeability of malignant vasculature and then illuminate by a light with an excitation wavelength range from 750 to 800 nm with high tissue penetration (millimeters to centimeters) where the human eye cannot detect so, it does not alter the surgical field. ICG device need a couple of filters, a special camera and a light source. The pattern of ICG florescence of tumors is classified into three types: total, partial and rim fluorescence (16).
This study was conducted in order to assess and provide precise data for the efficacy and added value of using ICG in diagnostic staging laparoscopy in intra-abdominal malignancy, especially in the identification of hepatic malignancies or liver and peritoneal metastases. Thirty patients with operable intra-abdominal malignancy presented to Theodor Bilhariz research institute from February 2022 to July 2023 were included in this study. In addition to standard of care, patients received an intravenous injection of 0.25-0.5 mg/kg ICG 24-72 hours prior to surgery. During surgery, patients underwent a staging laparoscopy; where upon inspection then near infra-red (NIR) fluorescence imaging was performed searching for hepatic focal lesions or peritoneal nodules.
In our study, the primary cancer was in the colon in 18 patients (60%), pancreas in 5 patients (16.7%), rectum in 1 patient (3.3%), stomach in 5 patients (16.7%) and duodenum in 1 patient (3.3%). In another study conducted by Lim, et al., ICG ability to localize liver tumor (17). Shirakawa, et al., and Handgraaf, et al., used indocyanine green to detect hepatic micro-metastases in patients with pancreatic cancer (13,18).
In another studies conducted by Filippello et al., and Barabino et al., assessed the affinity of Indocyanine green in the detection of peritoneal carcinomatosis (PC) in colorectal cancers (19,20). Satou et al., study search for PC in hepato-cellular carcinoma (HCC) patients Satou et al. (21), Ikoma et al., search for PC in gastric cancers Ikoma et al. (22) and Shirakawa, et al., search for PC in pancreatic cancers (18).
In our study, 0.25-0.5 mg/kg of ICG 24-72 hours pre-operative was injected. Kose, et al., inject ICG 24-48 hours pre-operative (23). Yokoyama, et al., inject 25 mg IV 24 hour before surgery (24).
No adverse reactions were recorded in any of our patients. This is concomitant to the results reported by Lim et al. (17) and Kose et al. (23).
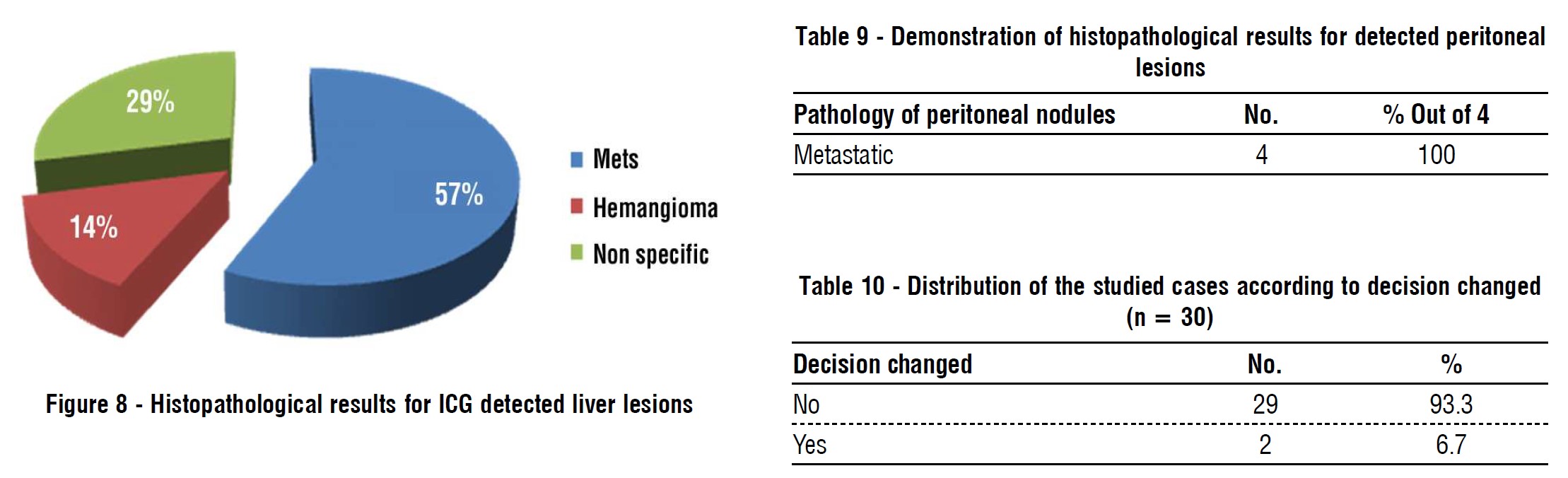
In our study, total numbers of patients who had hepatic focal lesions were 6 patients, one patient had fatty infiltration, so, he was excluded (n=5). 4 patients (80%) discovered with contrast computed tomography (CT) which was concomitant with the study conducted by Shah et al. (25), 4 patients (80%) were discovered by near infra-red indocyanine green auto-florescence. Total numbers of hepatic focal lesions were 12 lesions (n=12); 8 lesions (67%) were detected by contrast computed tomography (CT), while 11 lesions (92%) were detected by near infra-red indocyanine green auto-florescence, (p value) this is almost similar to the result conducted by Piccolo, et al., reported sensitivity up to (100%) in detection of liver metastasis by ICG (26). Lim et al., reported (82.7%) successful ICG detection (17). Boogerd reported experience in a series of patients with multiple hepatic tumors where ICG allowed detection of 3 other lesions with overall sensitivity (94%) Boogerd et al. (27), but these were not concomitant with the results reported by Kose et al., who reported only (43%) accuracy in detection of liver lesions by ICG versus (74%) for pre-operative imaging (23).
In our study, hepatic focal lesions which were positive for metastases were seen intraoperative by near infra-red technique as a rim of florescence with dark center. The area for florescence was larger than the actual size of the metastatic lesion (margins were pathologically negative) and this was concomitant with the study conducted by Naoyuki, et al., who said the area of the fluorescence was larger than the actual tumor size, indicating that the metastatic cells themselves did not fluorescence (28).
All of other lesions detected by indocyanine green auto-florescence were superficial “< 1cm in depth” and the one missed by ICG was a hemangioma as it was impeded in liver parenchyma. These are concomitant with the study conducted by Lim et al., reported
accuracy (84.1%) for lesions at depth <1cm and less than (50%) for lesions at depth >1cm from liver surface Lim et al. (17) and Kose et al., study findings accuracy of ICG were (95%) for superficial lesions and (4%) for deep lesions (23). Ishizawa et al., and Boogerd et al., studies found limited of depth of detection (8 mm) from the surface (27,29).
One out of five of patients presented with pancreatic malignancy (20%) had 2 micro-lesions not seen in triphasic CT imaging or during diagnostic staging laparoscopy, which are concomitant with the study done by Piccolo et al. Indocyanine green allow detection of hidden micro lesions that were missed by preoperative imaging (26). Yokoyama et al. demonstrated that ICG is able to detect additional micro nodules in the liver in (16%) of the patients with pancreatic cancer (24). Yokoyama and Otani in another study reported presence of liver micro-metastases by ICG in 4 out of 26 patients (15.4%) who previously diagnosed with liver metastases in colorectal cancer (CRCLM) (24).
In our study, 7 hepatic lesions were biopsied for histopathological assessment, 4 lesions were positive for metastasis (57.2%) and one was identified as hemangioma (14.3%) and the last 2 lesions (micro-lesions) showed non specific findings (28.5%). In the study conducted by Yokoyama et al., 31 liver biopsy had been taken and 17 (55%) were positive for malignancy and the rest of specimens were false positive and in CT examination follow up 6 months post-operative, the patients with liver micro metastatic nodules developed liver metastases more than the patients without micro metatatic nodules(24).
In our study, by conventional preoperative investigations no peritoneal nodules were identified, while two patients were discovered to have multiple peritoneal nodules during diagnostic laparoscopy. ICG auto-florescence imaging helped in identification of peritoneal nodules in one patient only (over all sensitivity 50%). Lieto, et al., study reported that the ICG florescence allowed identifying of 16 nodules undiagnosed from conventional procedures, adding a 25% diagnostic improvement (30). In contrast to Kose, et al., study reported that peritoneal (omental and diaphragmatic) metastatic nodules did not show fluorescence (23).
In our study, all histopathological assessment of peritoneal (omentum and diaphragmatic) nodules are positive for malignancy.
In our study, the surgical decision had been changed in 2 out of 30 patient (6.7%) and this is concomitant with the study conducted by Uchiyama et al. who used contrast enhanced intra-operative ultrasound plus ICG and they changed the surgical strategy in 6% of patients (31). Liberale et al. study results showed that intra-operative ICG florescence is helpful in the detection of peritoneal metastases of (CRC), resulting in surgery modifications in 38% of patients (32).
Limitation
The main limitation for the usage of indocyanine green is the cost and availability of laparoscopic near infra-red auto-florescence system. A major drawback of ICG in detection of hepatic lesions is the limited penetration power; hence, only superficial lesions could be seen.
CONCLUSION
ICG is a safe and applicable tool for intra-operative assessment and detection of liver lesions with high sensitivity. It is not specific for malignant lesions rather than hyper-vascular lesions, affecting its specificity. There is a discrepancy between sensitivity of ICG for detection of liver lesions and detection of peritoneal nodules, which may be affected by dose or time of pre-operative injection. Another study with larger sample size and dedicated post-operative follow up may be needed for more precise data.
Conflicts of interest and source of funding
No conflicts of interest, no source of funding.
Ethics approval
The procedures were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Committee for Human Rights.
REFERENCES
1. Muntean V, Oniu T, Lungoci C, Fabian O, Munteanu D, Molnar G, et al. Staging laparoscopy in digestive cancers. J Gastrointestin Liver Dis. 2009;18(4):4617.
2. Callisia C and Brian B. Surgery for Cancers of the Gastrointestinal Tract A Step-by-Step Approach. https:// www. amazon. com/ Surgery-Cancers-Gastrointestinal-Tract Step/dp/1493918923 2016.
3. Peloso A, Franchi E, Canepa MC, Barbieri L, Briani L, Ferrario J, et al. Combined use of intraoperative ultrasound and indocyanine green fluorescence imaging to detect liver metastases from colorectal
cancer. HPB (Oxford). 2013;15(12):928-34.
4. Huang Y, Alzahrani NA, Chua TC, Liauw W, Morris DL. Impacts of peritoneal cancer index on the survival outcomes of patients with colorectal peritoneal carcinomatosis. Int J Surg. 2016;32:65–70.
5. Low RN, Barone RM and Lucero J. Comparison of MRI and CT for predicting the peritoneal Cancer index (PCI) preoperatively in patients being considered for cytoreductive surgical procedures. Ann Surg Oncol. 2015;22(5):1708-15.
6. Machairas N, Charalampoudis P, Molmenti EP, Kykalos S, Tsaparas P, Stamopoulos P, et al. The value of staging laparoscopy in gastric cancer. Ann Gastroenterol. 2017;30(3):287-294.
7. Baiocchi GL, Gheza F, Molfino S, Arru L, Vaira M, Giacopuzzi S. Indocyanine green fluorescence-guided intraoperative detection of peritoneal carcinomatosis: systematic review. BMC Surg. 2020;20(1):158.
8. Schols RM, Connell NJ, Stassen LPS. Near-infrared fluorescence imaging for real-time intraoperative anatomical guidance in
minimally invasive surgery: a systematic review of the literature. World J Surg. 2015;39(5):1069-79.
9. Handa T, Katare RG, Nishimori H, Wariishi S, Fukutomi T, Yamamoto M, et al. New device for intraoperative graft assessment: HyperEye charge-coupled device camera system. Gen Thorac Cardiovasc Surg. 2010;58(2):68-77.
10. Guan X. Cancer metastases: challenges and opportunities. Acta Pharm Sin B. 2015;5(5):402-18.
11. Horn SR, Stoltzfus KC, Lehrer EJ, Dawson LA, Tchelebi L, Gusani NJ, et al. Epidemiology of liver metastases. Cancer Epidemiol. 2020; 67:101760.
12. Miku?a-Pietrasik J, Uruski P, Tykarski A, Ksi??ek K. The peritoneal "soil" for a cancerous "seed": a comprehensive review of the pathogenesis of intraperitoneal cancer metastases. Cell Mol Life Sci. 2018; 75(3):509-525.
13. Handgraaf HJ, Sibinga Mulder BG, Shahbazi Feshtali S, Boogerd LS, van der Valk MJ, Fariña Sarasqueta A, et al. Staging laparoscopy with ultrasound and near-infrared fluorescence imaging to detect occult metastases of pancreatic and periampullary cancer. PLoS One. 2018;13(11):e0205960.
14. Olson MT, Ly QP, Mohs AM. Fluorescence guidance in surgical oncology: challenges, opportunities, and translation. Mol Imaging Biol. 2019;21(2):200-218.
15. Alander JT, Kaartinen I, Laakso A, Pätilä T, Spillmann T, Tuchin VV, et al. A review of indocyanine green fluorescent imaging in surgery. Int J Biomed Imaging. 2012:2012:940585.
16. Ishizawa T, Masuda K, Urano Y, Kawaguchi Y, Satou S, Kaneko J, et al. Mechanistic background and clinical applications of indocyanine green fluorescence imaging of hepatocellular carcinoma. Ann Surg Oncol. 2014;21(2):440-8.
17. Lim HJ, Chiow AK, Lee LS, San Tan S, Goh BK, Koh YX, et al., Novel method of intraoperative liver tumour localisation with indocyanine green and near-infrared imaging. Singapore Med J. 2021;62(4):
182-189.
18. Shirakawa S, Toyama H, Kido M, Fukumoto T. A prospective single-center protocol for using near-infrared fluorescence imaging with indocyanine green during staging laparoscopy to detect small metastasis from pancreatic cancer. BMC Surg. 2019;19(1):165.
19. Filippello A, Porcheron J, Klein JP, Cottier M, Barabino G. Affinity of indocyanine green in the detection of colorectal peritoneal carcinomatosis: the role of enhanced permeability and retention effect. Surg Innov. 2017;24(2):103-108.
20. Barabino G, Klein JP, Porcheron J, Grichine A, Coll JL, Cottier M. Intraoperative near-infrared fluorescence imaging using indocyanine green in colorectal carcinomatosis surgery: proof of concept. Eur J Surg Oncol. 2016;42(12):1931-7.
21. Satou S, Ishizawa T, Masuda K, Kaneko J, Aoki T, Sakamoto Y, Hasegawa K, Sugawara Y, et al., Indocyanine green fluorescent imaging for detecting extrahepatic metastasis of hepatocellular
carcinoma. J Gastroenterol. 2013;48(10):1136-43.
22. Ikoma N, Blum M, Chiang YJ, Estrella JS, Roy-Chowdhuri S, Fournier K, et al. Yield of staging laparoscopy and lavage cytology for radiologically occult peritoneal carcinomatosis of gastric cancer. Ann Surg Oncol. 2016;23(13):4332-7.
23. Kose E, Kahramangil B, Aydin H, Donmez M, Takahashi H, Acevedo-Moreno LA, et al. A comparison of indocyanine green fluorescence and laparoscopic ultrasound for detection of liver tumors. HPB. 2020;22(5):764-9.
24. Yokoyama N, Otani T, Hashidate H, Maeda C, Katada T, Sudo N, et al. Real-time detection of hepatic micrometastases from pancreatic cancer by intraoperative fluorescence imaging: preliminary results of a prospective study. Cancer. 2012;118(11):2813-9.
25. Shah AJ, Callaway M, Thomas MG, Finch-Jones. Contrast-enhanced intraoperative ultrasound improves detection of liver metastases during surgery for primary colorectal cancer. HPB. 2010;12(3):
181-7.
26. Piccolo G, Barabino M, Pesce A, Diana M, Lecchi F, Santambrogio R, et al. Role of indocyanine green fluorescence imaging in minimally invasive resection of colorectal liver metastases. Surg Laparosc Endosc Percutan Tech. 2022;32(2):259-265.
27. Boogerd LS, Handgraaf HJ, Lam HD, Huurman VA, Farina-Sarasqueta A, Frangioni JV, et al. Laparoscopic detection and
resection of occult liver tumors of multiple cancer types using real-time near-infrared fluorescence guidance. Surg Endosc. 2017;31(2): 952-961.
28. Tateiwa N, Haga Y, Matsuda TD, Fisk Z. Magnetic measurements at pressures above 10 GPa in a miniature ceramic anvil cell for a
superconducting quantum interference device magnetometer. Rev Sci Instrum. 2012;83(5):053906.
29. Ishizawa T, Fukushima N, Shibahara J, Masuda K, Tamura S, Aoki T, et al., Real-time identification of liver cancers by using indocyanine green fluorescent imaging. Cancer. 2009;115(11):2491-504.
30. Lieto E, Auricchio A, Cardella F, Mabilia A, Basile N, Castellano P, et al. Fluorescence-guided surgery in the combined treatment of peritoneal carcinomatosis from colorectal cancer: preliminary results and considerations. World J Surg. 2018;42(4):1154-1160.
31. Uchiyama K, Ueno M, Ozawa S, Kiriyama S, Shigekawa Y, Yamaue H. Combined use of contrast-enhanced intraoperative ultrasono-
graphy and a fluorescence navigation system for identifying hepatic metastases. World J Surg. 2010;34(12):2953-9.
32. Liberale G, Vankerckhove S, Gomez Caldon M, Ahmed B, Moreau M, El Nakadi I, et al. Fluorescence Imaging After Indocyanine Green Injection for Detection of Peritoneal Metastases in Patients Undergoing Cytoreductive Surgery for Peritoneal Carcinomatosis From Colorectal Cancer: A Pilot Study. Ann Surg. 2016;264(6):1110-1115.
33. McLeod S. Z-score definition, calculation and interpretation. Simply Psychology. https://www.simplypsychology.org/z-score.html (2019, May 17).
Full Text Sources:
Abstract:
Views: 3201

For Authors
Journal Subscriptions

Dec 2025
Supplements
Instructions for authors
Online submission
Contact
e-ISSN: 2601 - 1700 (online)
ISSN-L: 2559 - 723X
Journal Abbreviation: Surg. Gastroenterol. Oncol.
Surgery, Gastroenterology and Oncology (SGO) is indexed in:
- SCOPUS
- EBSCO
- DOI/Crossref
- Google Scholar
- SCImago
- Harvard Library
- Open Academic Journals Index (OAJI)
Surgery, Gastroenterology and Oncology (SGO) is an open-access, peer-reviewed online journal published by Celsius Publishing House. The journal allows readers to read, download, copy, distribute, print, search, or link to the full text of its articles.
Time to first editorial decision: 25 days
Rejection rate: 61%
CiteScore: 0.2

Meetings and Courses in 2025
Meetings and Courses in 2024
Meetings and Courses in 2023
Meetings and Courses in 2022
Meetings and Courses in 2021
Meetings and Courses in 2020
Meetings and Courses in 2019
Verona expert meeting 2019

Surgery, Gastroenterology and Oncology applies the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits readers to copy and redistribute the material in any medium or format, remix, adapt, build upon the published works non-commercially, and license the derivative works on different terms, provided the original material is properly cited and the use is non-commercial. Please see: https://creativecommons.org/licenses/by-nc/4.0/
Publisher’s Note:
The opinions, statements, and data contained in article are solely those of the authors and not of Surgery, Gastroenterology and Oncology journal or the editors. Publisher and the editors disclaim responsibility for any damage resulting from any ideas, instructions, methods, or products referred to in the content.
 IASGO Society News
IASGO Society News