Surgery, Gastroenterology and Oncology

|
|
Introduction: Ductal carcinoma in situ (DCIS) is rarely diagnosed in young female patients with most studies view age as a poor prognostic factor.
Patient and Methods: Female patients who underwent surgery for DCIS of the breast (with or without microinvasion) in our department between February 2007 and February 2022 were included. They were divided into two groups: group I (<40 years old) and group II (³40 years old), aiming to evaluate and compare the clinical-epidemiological, pathological, radiological, operative, and outcome data between the two groups to test the notion that young age in DCIS patients is a poor prognostic factor that may warrant more aggressive treatment.
Results: One hundred sixty-two patients were included in the current study. The mean age was 51.8 years, and 20.1% of patients were younger than 40 years. The commonest symptom reported in 79.3% of cases was a breast lump. The final pathology was pure DCIS in 2/3 of patients, with microinvasion present in the remaining third. Recurrence occurred in 11 (6.7%) patients. Tumor size, grade, histologic subtype, ER status, pathological multicentricity, type of surgery, and recurrence were not-statistically significant (p-value=.223, .097, .68, .41, .37, 1, .23, respectively). Also, disease-free survival was not significant between the two groups (p-value = 0.3). However, the use of magnetic resonance imaging and breast reconstruction was significantly more common in the younger age group (p-value=.039 and <.001, respectively).
Conclusion: One-fifth of our patients with DCIS were less than 40 years old, with recurrence being quite uncommon. The use of Breast-conserving surgery is increasing in our community; however, it is still underutilized. Additionally, MRI and breast reconstruction were more widely used in the younger group. Outcomes were similar, not justifying the use of more aggressive treatment in younger DCIS patients.
Introduction
Ductal carcinoma in situ (DCIS) is a noninvasive pre-cancerous breast lesion that accounts for 20-25 % of annual breast cancer diagnoses (1,2). Recently, there has been a dramatic increase in its incidence from 5% to around 25% due to improved and widely used screening mammography and other imaging modalities (3).
However, DCIS is not commonly diagnosed in patients < 40 years (4), as it is mostly detected through screening mammography which is not generally applied for such a population. In previous studies, one of the poor prognosticators for both recurrence or progression and cancer mortality was young age at diagnosis (5-7). As a result, younger patients tend to receive more intensive treatment in terms of surgical extent and radiotherapy (RT) (8,9).
The complex set of biological considerations, psychosocial factors, and unique disease characteristics, combined with sparse studies and no age-specific guidelines highlights the importance of research into the characteristics, management, and outcomes of DCIS in the specific cohort of younger women compared to their older counterparts.
In this study, we aim to evaluate the clinical-epidemiological, pathological, radiological, operative, and outcome data of patients < 40 years old and compare these findings with those of their ? 40 years' old female counterparts, to test the notion that young age in DCIS patients is a poor prognostic factor that may warrant more aggressive treatment.
Patient and Methods
Type of Study
This is a retrospective, single cancer center study approved by the IRB of the Faculty of Medicine, Mansoura University (number R.22.09.1834).
Patient and Variables
We included all patients who were operated on for DCIS of the breast at the surgical oncology department, Oncology Center - Mansoura University (OCMU), between February 2007 and February 2022. The patients were divided into two groups: group I (<40 years old) and group II (? 40 years old).
Inclusion criteria
The patients with DCIS (with or without microinvasion) who underwent surgical management and subsequent follow-up at our center.
Exclusion criteria
• Invasive breast carcinoma even with intraductal component;
• Lobular carcinoma in situ;
• Intracystic papillary carcinoma;
• Non-epithelial pathology;
• Nodal positive disease;
• Missed the patient’s data or lost follow-up.
Variables
We retrieved the following data: age at diagnosis, BMI, family history, symptoms, pattern of calcifications and breast density as ACR, type of surgery, type of axillary assessment, type of breast reconstruction, morphological pattern of DCIS, tumor size, presence or absence of microinvasion, Lymphovascular emboli (LVE), estrogen receptor (ER) status, adjuvant therapies, recurrence status/pattern, date, management of recurrence, new non-breast primary cancer, and contralateral breast cancer.
A section of missed data was collected by two different investigators through phone calls with the included patients or their families, which helped minimize data loss to the lowest possible level.
Statistics
Data was analyzed on a personal computer running SPSS© for Windows (Statistical Package for Social Scientists) Release 26 for descriptive statistics of qualitative variables. The frequency distribution procedure through calculation of the number of cases and percentages was used. For descriptive statistics of quantitative variables, the mean, median, range, and standard deviation were used to describe central tendency and dispersion. A p-value of ?0.05 is considered statistically significant.
Results
One hundred sixty-two patients were recruited, with two patients having bilateral breast DCIS, thus representing 164 cases. All patients were females. Twenty-one patients (13%) had a family history of breast cancer (6 in the young group versus 15 in the old group). Additionally, 4 patients had a history of uterine cancers (3 in the old and 1 in the young group).
Table 1 - Basic epidemiologic, pathologic and outcomes data of the entire series (162 patients presented as 164 cases)
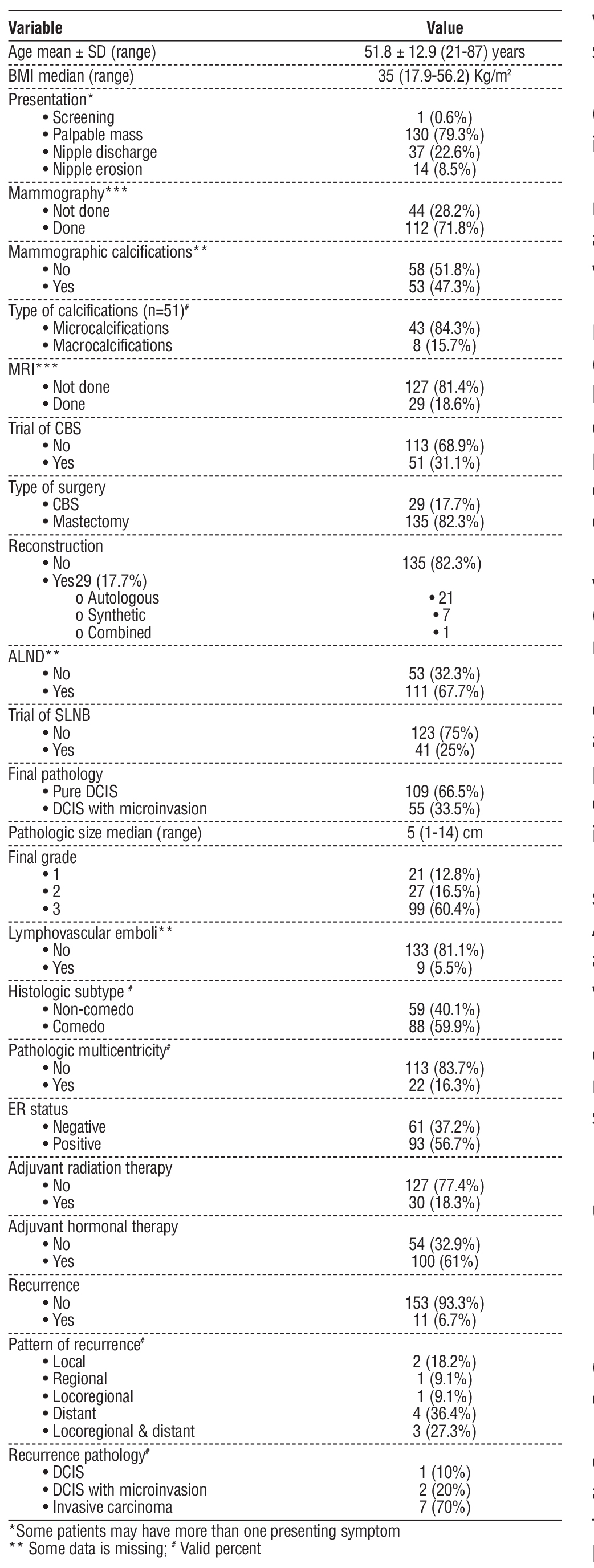
Clinico-epidemiologic, pathologic, and treatment data (table 1)
The mean age was 51.8 years (SD ± 12.9), and 20.4% of patients were younger than 40 years old. The median BMI was 35 Kg/m2 (range: 17.9-56.2).
Only one case was detected through screening mammography. The commonest symptom was a breast lump, reported in 130 cases (79.3%), and followed by nipple discharge in 37 cases (22.6%), of which 51.5% were bloody. The median clinical mass size was 3 cm (range: 1-12).
All cases were assessed with ultrasound; 112 cases (71.8%) also underwent mammography. MRI was used in only 29 cases (18.6%).
Preoperative ultrasound showed that BIRADS 4 was reported in 46 cases (28%), BIRADS 5 in 18 cases (11%), and BIRADS 6 in 10 cases (6.1%). The median mass size was 2.5 cm (range: 0.5-19.5).
Various biopsy techniques were used as follows: Fine needle aspiration cytology (FNAC) in 8 cases (4.9%), Tru-cut biopsy in 85 cases (51.8%), incisional biopsy in 15 cases (9.1%), and excisional biopsy in 56 cases (34.1%), of which 50 cases (30.5%) were performed as unplanned excisional biopsies without obtaining safety margins and were done outside our center.
Preoperative pathology showed that grade 1 DCIS was reported in 24 cases (14.6%), grade 2 in 26 cases (15.9%), and grade 3 in 42 cases (25.6%). Comedo necrosis was observed in 44 cases (26.8%).
Only 29 cases (17.7%) underwent successful breast conserving surgery (BCS) out of 51 attempts (31.1%) to avoid mastectomy; however, the rest of them had persistent positive margins. Additionally, another 29 cases (17.7%) underwent breast reconstruction, mostly involving the use of autologous flaps.
One hundred fifty cases (91.5%) underwent axillary surgery. Among them, 109 cases underwent upfront ALND, and 41 cases underwent SLNB, 2 of which failed and were converted to ALND. All axillary specimens were pathologically negative.
The final pathology showed pure DCIS in 2/3 of cases, 109 cases (66.5%), while microinvasion was reported in the remaining third, 55 cases (33.5%). ER status was positive in 93 cases (56.7%).
Adjuvant hormonal treatment was administered in 100 cases (61%), and all 30 cases (18.3%) who underwent BCS received adjuvant radiation therapy.
Recurrences (table 1)
The median follow-up period was 50.5 months (range: 0.5 -195), with recurrence occurred in only 11 cases (6.7%). Of these, 2 were local, 1 was regional, 1 was locoregional, 4 were distant, and 3 were combined locoregional and distant recurrence. Bone, as a single site recurrence, is the most common site for distant recurrence (in 4 patients), followed by the lung, distant lymph nodes, and combined bone and liver in 1 patient each.
We report regional nodal recurrence of 5 cases (3%) in our series, of which 4 cases were initially diagnosed with microinvasion (valid percent: 7.3%) and 1 case with pure DCIS (valid percent: 0.9%).
In patients with recurrence, 8 out of 11 (72.7%) had a microinvasive component in the primary lesion, and 8 of them (72.7%) were Grade 3 initially. The histo-pathology of the recurrence was confirmed as in situ carcinoma in 1 case, microinvasive carcinoma in 2 cases, and invasive carcinoma in 4 cases. However, biopsy was not accessible in another 3 distant metastatic sites, but they were considered invasive by definition.
Comparison of Young to Old Groups (table 2)
Family history of breast cancer showed a higher, albeit non-significant difference in favor of the old patient group (p-value = .38). Diagnostic mammography was more frequently used in the older group, with no statistical significance (p-value = .12). In contrast, MRI was significantly more commonly used in the younger group (p-value = .039).
Table 2 - Comparison of DCIS cases based on age as young (n = 33) versus old (n = 131)
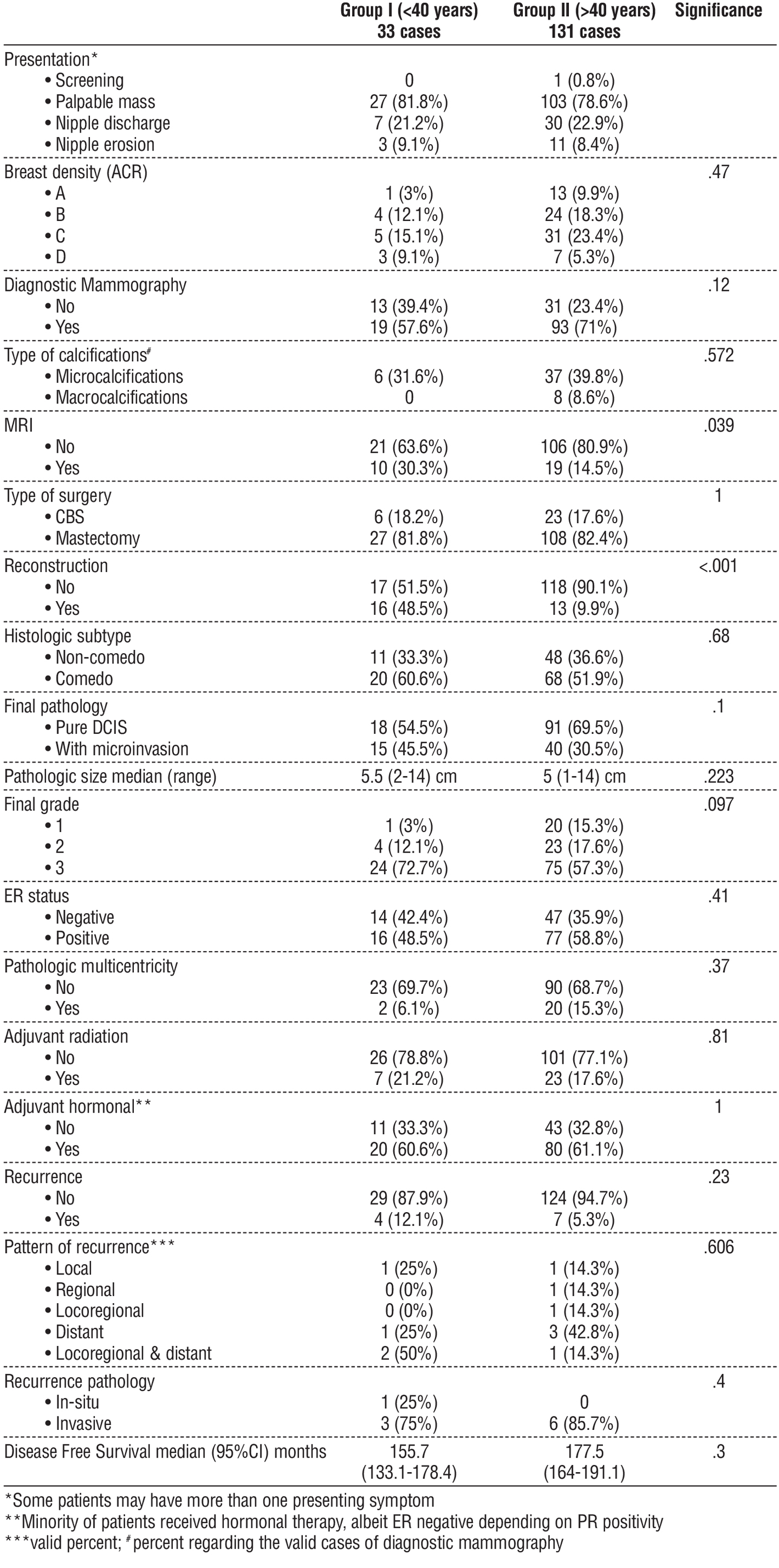
Both groups were offered the same type of surgery (mastectomy or BCS) (p-value = 1); however, breast reconstruction was significantly more frequently used in the younger group (p-value =< .001).
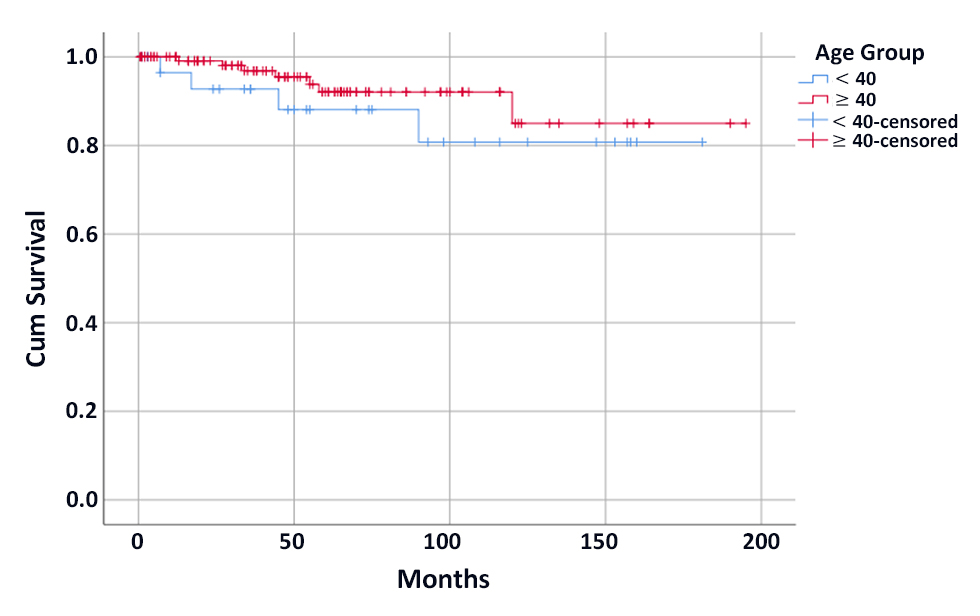
Figure 1 - Kaplan-Meier curve for disease-free survival among the two groups.
The pathologic subtype (comedo vs. non-comedo), incidence of microinvasion, final tumor grade, and ER status were similar in both groups (p-value = .68, .1, .097, and .41, respectively).
The incidence of multicentricity in the pathologic specimens was also similar between younger and older patients (p-value = .37). The young age group showed a slightly higher, though not statistically significant, median pathological tumor size (5.5 vs. 5 cm, p-value = .223).
Both groups received similar adjuvant treatments, including radiotherapy and hormonal therapy (p-value = .81 and 1, respectively).
In addition, recurrence rate, its pattern, and recurrence pathology (in-situ vs. invasive) were similar in both groups (p-value = .23, .606, and .4, respectively). However, in older patients, all distant recurrences were bony in contrast to visceral ± bone metastasis in the younger group (p-value = .072).
Four patients developed contralateral breast cancer; all were invasive. Additionally, one case each of uterine cancer, HCC, pancreatic head cancer, ovarian dysgerminoma (in a patient with Cowden syndrome), and appendiceal mucinous neoplasm were reported. All of them were > 40 years apart from the patient with Cowden syndrome.
While being lower at the young age group (155.7 vs. 177.5 months), disease-free survival (DFS) was not significant between the two groups (p-value = 0.3) (fig. 1).
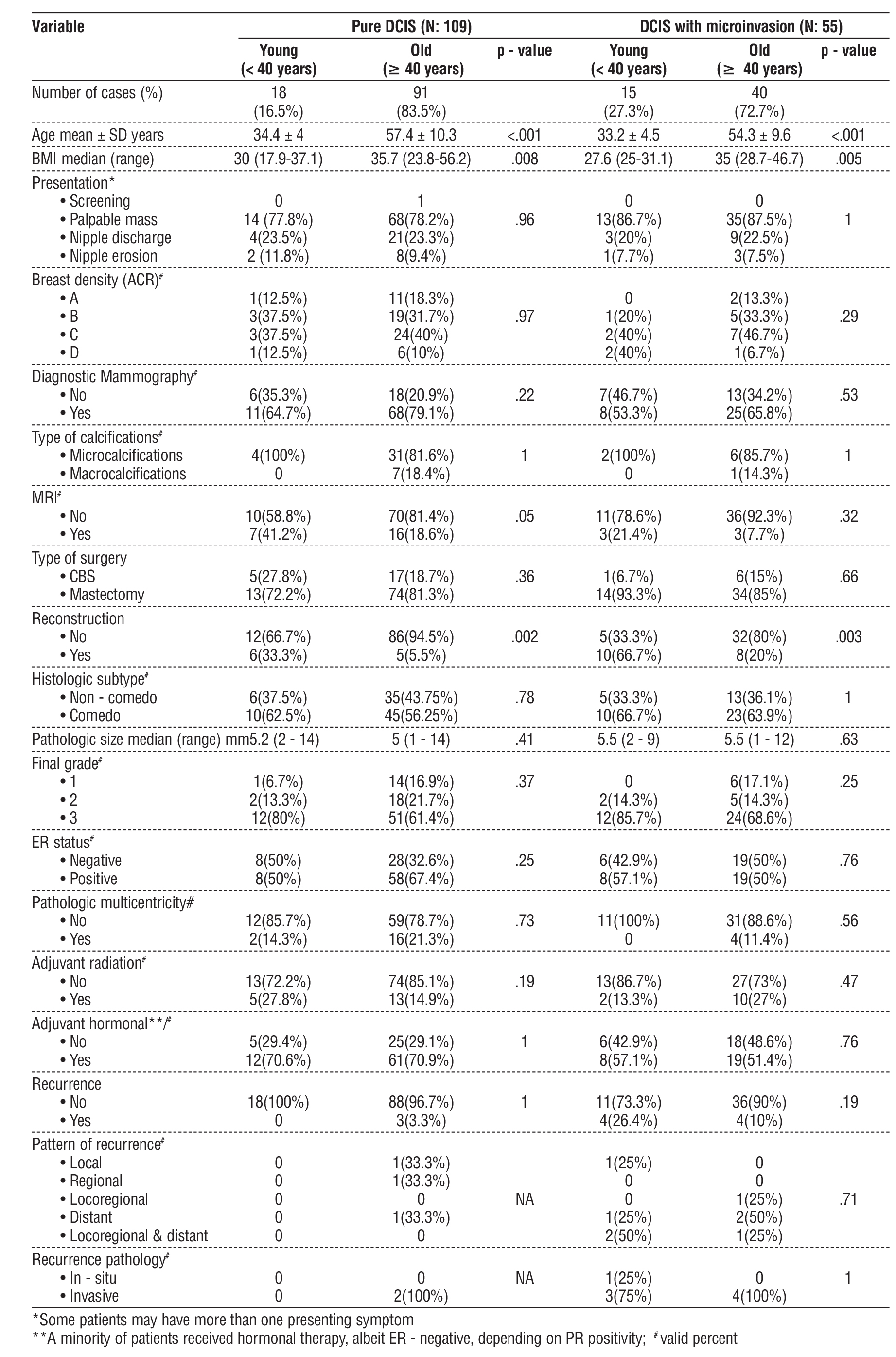
Subgroup Analysis (table 3)
In the subgroup analysis, comparisons between the two (young vs. old) groups in patients with pure DCIS only and in those with DCIS with microinvasion showed significant differences in both BMI (p-value= .008 and .005) and reconstruction (p-value= .002 and .003), where younger patients were slimmer and opted for more breast reconstructive procedures, respectively.
Table 3 - Subgroup analysis (the two groups compared according to the result of the final pathology)
Discussion
In the USA, Female patients < 40 years old showed a nearly 4-fold increase in the incidence of DCIS in the period from 1975 to 2015 (10), however, in 2017, Nguyen et al. (11) reported an incidence of 2.9% from 2004 to 2014. In 2019, this patient category represented only 2% of newly diagnosed cases, making this population particularly challenging to study due to low number of cases (12).
Over 15 years of our practice, young female patients represented only one-fifth of the cases diagnosed with DCIS. However, Alvarado et al. (13) reported an incidence of 6.5%, Jhingran et al. (14) reporting 13%, and Vicini et al. (5) reported 21% of patients were < 45 years old.
Presentations of DCIS vary between clinical and radiological, or both, with different orders among publications. For example, Fisher et al. (15) reported that the most common presentation was imaging-only calcifications without a mass in 77.7% of patients, followed by breast mass ± calcifications in 15.6%. Jhingran et al. (14) reported mass lumps in only 25% of their patients. In 2013, Vicini et al. (5) reported on 30 cases < 45 compared with 115 cases > 45 years old, showed that screening mammography was the most common mode of detection and was used nearly equally in both groups (83.3% in young vs. 90.4% in old patients). Mammographic calcifications were reported in 84.8% of cases (86.7% in young vs. 84.3% in old), with the young group showing a higher rate of clinical signs compared to old (16.7% vs. 9.6%), albeit with no statistically significance regarding those parameters.
Tesch et al. (4) reported that only 36.8% of their 98 cases showed symptoms on diagnosis, with palpable breast lump observed in 22.4%, and radiologic only features in 52% of cases.
On the other hand, Alvarado et al. (13) reported different patterns of presentation with statistically significance differences between groups, as they showed higher clinical versus radiological presentation (56.1% vs. 43.9%) in <40 years old, with breast mass being the most common finding. Conversely, radiological presentation was higher in the older age group (86% vs. 14%).
Regarding our cohort, a breast lump was reported in more than three-quarters of patients, with only one patient detected through screening. This can be explained by the fact that screening mammography is generally not applied to <40 years old females, (4) resulting in most young age patients showing more clinically advanced DCIS (13). Additionally, the delayed implementation of the breast screening mammography campaign in Egypt until 2019 could be the culprit. However, diagnostic mammography was performed in 71.8 % of our cohort and in 57.6% of our younger group, with no statistically significant difference compared with the old group (p-value = .12). All of our young patients presented with symptoms, and a breast lump was reported in 81.8% of them. Mammographic calcifications were reported in 47.3% of cases, with the majority (84.3%) being microcalcifications, again with no significant difference among groups (p-value = .572).
Similarly, the same differences among publications can be found regarding tumor biological characteristics. Alvarado et al. (13) reported significantly higher tumor grade, ER-positive status, multicentricity, and multi-focality with no statistically significant difference regarding size either, mammographically or pathologically, when comparing young versus old patients. These unfavorable tumor characteristics in the younger group may be explained by either genetic predisposition or the more advanced clinical presentation, given the fact that this group is generally below the age threshold for mammographic breast cancer screening, as previously mentioned. Also, Halasz et al. (16) reported significantly larger size and higher grade tumors in young patients <30 years old. In contrast, Vicini et al (5) reported no statistically significant differences regarding pathologic calcifications, histologic subtype, and nuclear grade. Similarly, our study showed no significance differences between groups in such parameters, which may be explained, as in Vicini et al. (5), by the relatively low number of young patients in our cohort.
Most patients in both age groups in all of these publications (5,13,16) as well as in our study, there was a trend toward a higher percentage of grade 3 tumors. However, Tesch et al. (4) reported a higher proportion of grade 2 tumors. In fact, it is well known that only 9-12% of DCIS are low grade (17-21).
The comedo architectural subtype was the commonest in our cohort, reported in 59.9% of cases, with no significant difference between groups (60.6% in young vs. 51.9% in old, p-value=.68). Vicini et al. (5) reported a lower incidence of the comedo pattern in 23.6% of cases, with the cribriform pattern being the commonest subtype in 51.4% of cases. The comedo pattern was more frequent in the young age group (30% vs. 20.9%); in contrast the cribriform pattern was higher in the old age group (53.9% vs. 33.3%). However, no statistically significant difference was reported between groups regarding the pattern. Regarding the central necrosis, it was reported in 57.2% of cases, with a higher though non-statistically significant difference in younger versus old patients (73.3% vs. 61%). Alvarado et al (13) also reported the same trend regarding necrosis (44.7% in young vs. 37.2% in old patients).
However, Tesch et al. (4) reported a much lower rate of the comedo pattern in only 3.1% of patients, with the commonest pattern being solid (54.1%), followed by cribriform (25.5%). Comedo necrosis was observed in 35.7 % of patients.
Tesch et al. (4) reported multifocality in 10.2% of their cohort, while Alvarado et al. (13) found that women <40 years had significantly higher rates of both multicentric and multifocal disease than the older women, 29.3 % and 30.1 %, respectively. In our study, multicentricity was reported in 16.3% of cases with no statistically significant difference between the groups (p-value = .37).
DCIS is characterized by its heterogeneous nature and the potentiality for both recurrence and/or progression to invasive cancer (4), reported in up to 20-30% of patients if not being treated (22). It also
carries a higher risk for breast cancer related mortality (23,24).
Considering the longer life expectancy of younger patients and, therefore, the increased benefit of risk reduction, multimodality therapy is being pursued, including surgery, adjuvant radiotherapy, and hormonal therapy (25,26). Additionally, the active surveillance strategy is another option that is being investigated through many trials questioning both its validity and the qualified cohort of patients for such an approach (27-31).
The younger population shows some unique considerations in the management of breast cancer, such as increased concerns for possible lower ipsilateral breast cosmesis, overall bilateral breast symmetry, post-treatment quality of life, and the side effects of long periods of hormonal therapy on both fertility and pregnancy (32-34). Also, the high levels of anxiety and the false notion of lower survival benefit following breast conserving surgery (BCS) lead many patients to opt for mastectomy instead (35,36).
However, with the superior cosmetic results, BCS has become the standard of care for DCIS treatment (25,37).
M. Worni et al. (22) found a significant shift in loco-regional therapies for DCIS, with the use of lumpectomy combined with adjuvant radiotherapy increased in their cohort by almost 100%, whereas the use of unilateral mastectomy dropped nearly by two-thirds in comparison with past cohorts.
However, Byun et al. (38) reported an increased frequency of bilateral mastectomy in female patients < 40 years old with larger and higher grade pure DCIS tumors from 2004 to 2016, even surpassing BCS since 2010, despite showing no OS benefit. The same trend of increasing bilateral mastectomy use in younger patients was also reported by Halasz et al (16).
No significant difference was noted among our patients regarding type of surgery (p-value = 1), with less than a fifth (17.7 %) having a successful BCS. This may be attributed to an increased number of cases referred from rural hospitals, where 30.5% of patients presented after unplanned excisional biopsies without adequate safety margins. Also, the high percentage (79.3%) of patients presented with palpable breast lumps makes it sometimes difficult to preserve the breast. Most tumors were high grade, and another possible cause till few years ago was the concern of a substantial percentage of patients about the safety of BCS in terms of recurrence and survival outcomes, added to that the fear to undergo another surgical intervention (39).
Over-treatment in the form of axillary lymph node dissection (ALND) was performed in the majority of our patients, which was a common practice in our department until recent years, when a shift towards sentinel lymph node biopsy (SLNB) was commenced (40).
Regarding adjuvant hormonal therapy, it acts as chemoprophylaxis, resulting in risk reduction in both ipsilateral recurrence and contralateral invasive or in situ breast cancer (18,41-45). Therefore, the National Comprehensive Cancer Network (NCCN) guidelines recommend that it can be considered for risk reduction in cases of ER-positive DCIS (46). In our cohort, nearly two-thirds of patients received adjuvant hormonal therapy; however, no significant difference was noted among the age groups (p-value = 1).
Many clinical trials have confirmed the local control benefit of adjuvant radiotherapy (RT) use after BCS for DCIS, despite showing no effect on overall survival (15,18,47-52) In our cohort, all of our patients who underwent successful BCS, regardless of age group (p-value = .81), received adjuvant RT.
It is worth noting that both the proportional and absolute risk reductions in ipsilateral breast recurrence following the use of adjuvant RT was reported to be lower in younger compared with older patients, highlighting the influence of age at DCIS diagnosis, an effect which was not confounded by known prognostic factors (37,53). Moreover, there is evidence that adjuvant RT is more likely to reduce mortality rate in younger than in older women (8,54).
In previous studies, young age at diagnosis, mostly proposed as < 40 or < 50 years old, was one of the negative risk factors for disease recurrence or progression (5,6). For example, Kong et al. (55) found significantly higher local recurrence (LR) rates in Women <45 years, with significantly lower 5- and 10-year actuarial LRFS 84 vs. 90% and 79% vs. 86%, respectively compared to those 45-50 years. However, there was no statistically significant difference in invasive local recurrence among groups. Similarly, Alvarado et al. (13) reported 5-year rates of LR of 10.1 % in <40 years compared to 3.2 % in older women. Also, the 10-year breast tumor recurrence risk was reported as 11.2–31% versus 3–9%. (5,14,56,57).
In our study, recurrence was observed in 6.7% of the entire cohort, with distant recurrence being the commonest pattern, followed by combined loco-regional-distant, and then by local recurrence. Comparing the two groups, recurrence was more frequent in the young age group (12.1% vs. 5.3%); however, it was not statistically significant (p-value = .23). Moreover, there was no statistically significant difference regarding the pattern of recurrence (p-value = .606). Also, DFS was not significant between the two groups (p-value = 0.3) (fig. 1).
The low incidence of local recurrence in our cohort may be attributed to the high percentage (82.3%) of mastectomies performed. Stuart et al. (25) reported in a meta-analysis of 9,404 DCIS cases with 10-year follow-up that the adjusted meta-regression local recurrence rates were 2.6% for mastectomy, 13.6% for BCS with RT, 25.5% for BCS without RT, and 27.8% for biopsy-only (residual predominately low-grade DCIS following inadequate excision).
Even after BCS, <45 years women diagnosed with DCIS have a greater risk of local recurrence with different patterns of failure, which is independent of other previously defined risk factors, that are most notable within 10 years of diagnosis (5,58).
Tunon-de-Lara et al. (59) showed that comedo-carcinoma subtype, histological size >10 mm, necrosis, and positive safety margin were statistically significant predictive factors for recurrence after BCS in young patients.
Young age has also been associated with a higher breast cancer-specific mortality at 20 years, with the hazard ratio of 2.58 for < 35 years patients (7).
The 20-year risk of breast cancer related death following diagnosis of DCIS was reported to be 3 fold higher than the general population of the same age, with the standardized mortality ratio for < 40 years women being 11.95, which was higher than that for older age groups (26). It is worth noting that, not all women who die of cancer following DCIS, experience a local recurrence, suggesting that the current treatment focus on preventing invasive recurrence may be insufficient to eliminate all deaths from breast cancer after DCIS.
As a result of the previously discussed data, young DCIS patients tended to receive more intensive treatment, especially in terms of surgical extent and RT (8,9).
Still, there is a paucity of published articles describing treatment options and outcomes in young patients with DCIS (58,60).
In our study, despite some parameters being higher in one group compared to the other, no statistically significant differences were detected, except for MRI use and breast reconstruction, which were statistically significant for the young age patients who were more motivated to preserve their breasts.
Strengths and Limitations
Our study has frequent limitations. As a retrospective study conducted in a single tertiary center with a relatively small number of cases, mainly in the younger group, over an extended period, there is a possibility of missed important data, heterogeneity in assessment and treatment approaches, possible over-treatment, and loss to follow up. These limitations may have weakened the statistical power of some of our results; therefore, they may not represent the true impact of age on the larger population. Moreover, our results slightly contradict findings from other publications, so caution is mandatory while interpreting our results within the context of existing literature. However, this is the first publication from our locality to address this rare issue with such a thorough analysis. Despite limitations, our results stand against pursuing more aggressive treatment in younger patients such as, mastectomy in patients candidate for BCS or ALND instead of SLNB. We recommend adherence to the current guidelines with an individualized treatment approach for DCIS patients, regardless of age.
Conclusion
One-fifth of our patients with DCIS were less than 40 years old, with recurrence being quite uncommon. The use of Breast-conserving surgery is increasing in our community; however, it is still underutilized. Additionally, MRI and breast reconstruction were more widely used in the younger group. Outcomes were similar, not justifying, the use of more aggressive treatment in younger DCIS patients.
Competing Interests
Authors disclose no potential conflicts of interest.
Funding
Authors disclose no funding sources.
References
1. Vatovec C, Erten MZ, Kolodinsky J, Brown P, Wood M, James T, et al. Ductal carcinoma in situ: a brief review of treatment variation and impacts on patients and society. Critical reviews in eukaryotic gene expression. 2014;24(4):281-6.
2. Chootipongchaivat S, van Ravesteyn NT, Li X, Huang H, Weedon-Fekjær H, Ryser MD, et al. Modeling the natural history of ductal carcinoma in situ based on population data. Breast Cancer Res. 2020;22(1):53.
3. Toss MS, Pinder SE, Green AR, Thomas J, Morgan DA. Breast conservation in ductal carcinoma in situ (DCIS): what defines optimal margins? 2017;70(5):681-92.
4. Tesch ME, Rosenberg SM, Collins LC, Wong JS, Dominici L, Ruddy KJ, et al. Clinicopathologic Features, Treatment Patterns, and Disease Outcomes in a Modern, Prospective Cohort of Young Women Diagnosed with Ductal Carcinoma In Situ. Ann Surg Oncol. 2022.
5. Vicini FA, Shaitelman S, Wilkinson JB, Shah C, Ye H, Kestin LL, et al. Long-Term Impact of Young Age at Diagnosis on Treatment Outcome and Patterns of Failure in Patients with Ductal Carcinoma In Situ Treated with Breast-Conserving Therapy. The Breast Journal. 2013;19(4):365-73.
6. Mamtani A, Nakhlis F, Downs-Canner S, Zabor EC, Morrow M, King TA, et al. Impact of Age on Locoregional and Distant Recurrence After Mastectomy for Ductal Carcinoma In Situ With or Without Microinvasion. Annals of Surgical Oncology. 2019;26(13):4264-71.
7. Narod SA, Iqbal J, Giannakeas V, Sopik V, Sun P. Breast Cancer Mortality After a Diagnosis of Ductal Carcinoma In Situ. JAMA Oncology. 2015;1(7):888-96.
8. Sagara Y, Freedman RA, Vaz-Luis I, Mallory MA, Wong SM, Aydogan F, et al. Patient Prognostic Score and Associations With Survival Improvement Offered by Radiotherapy After Breast-Conserving Surgery for Ductal Carcinoma In Situ: A Population-Based Longitudinal Cohort Study. J Clin Oncol. 2016;34(11):1190-6.
9. Park HL, Chang J, Lal G, Lal K, Ziogas A, Anton-Culver H. Trends in Treatment Patterns and Clinical Outcomes in Young Women Diagnosed With Ductal Carcinoma In Situ. Clinical Breast Cancer. 2018;18(2):e179-e85.
10. Guo F, Kuo Y-f, Shih YCT, Giordano SH, Berenson AB. Trends in breast cancer mortality by stage at diagnosis among young women in the United States. Cancer. 2018;124(17):3500-9.
11. Nguyen TT, Hoskin TL, Day CN, Habermann EB, Goetz MP, Boughey JC. Factors Influencing Use of Hormone Therapy for Ductal Carcinoma In Situ: A National Cancer Database Study. Annals of
surgical oncology. 2017;24(10):2989-98.
12. DeSantis CE, Ma J, Gaudet MM, Newman LA, Miller KD, Goding Sauer A, et al. Breast cancer statistics, 2019. CA: A Cancer Journal for Clinicians. 2019;69(6):438-51.
13. Alvarado R, Lari SA, Roses RE, Smith BD, Yang W, Mittendorf EA, et al. Biology, treatment, and outcome in very young and older women with DCIS. Annals of surgical oncology. 2012;19(12):3777-84.
14. Jhingran A, Kim JS, Buchholz TA, Katz A, Strom EA, Hunt KK, et al. Age as a predictor of outcome for women with DCIS treated with breast-conserving surgery and radiation: The University of Texas M. D. Anderson Cancer Center experience. International Journal of Radiation Oncology*Biology*Physics. 2002;54(3):804-9.
15. Fisher B, Dignam J, Wolmark N, Mamounas E, Costantino J, Poller W, et al. Lumpectomy and radiation therapy for the treatment of intraductal breast cancer: findings from National Surgical Adjuvant Breast and Bowel Project B-17. J Clin Oncol. 1998;16(2):441-52.
16. Halasz SR, O'Keefe T, Wallace AM, Blair SL. Ductal carcinoma in situ in patients younger than 30 years: differences in adjuvant endocrine therapy and outcomes. Breast cancer research and treatment. 2021;186(2):551-9.
17. van Maaren MC, Lagendijk M, Tilanus-Linthorst MMA, de Munck L, Pijnappel RM, Schmidt MK, et al. Breast cancer-related deaths according to grade in ductal carcinoma in situ: A Dutch population-based study on patients diagnosed between 1999 and 2012. European journal of cancer (Oxford, England : 1990). 2018;101: 134-42.
18. Thompson AM, Clements K, Cheung S, Pinder SE, Lawrence G, Sawyer E, et al. Management and 5-year outcomes in 9938 women with screen-detected ductal carcinoma in situ: the UK Sloane Project. European journal of cancer (Oxford, England : 1990). 2018;101:210-9.
19. Rakovitch E, Nofech-Mozes S, Hanna W, Baehner FL, Saskin R, Butler SM, et al. A population-based validation study of the DCIS Score predicting recurrence risk in individuals treated by breast-
conserving surgery alone. Breast cancer research and treatment. 2015;152(2):389-98.
20. Punglia RS, Jiang W, Lipsitz SR, Hughes ME, Schnitt SJ, Hassett MJ, et al. Clinical risk score to predict likelihood of recurrence after ductal carcinoma in situ treated with breast-conserving surgery. Breast cancer research and treatment. 2018;167(3):751-9.
21. van Dooijeweert C, van Diest PJ. Significant inter- and intra-laboratory variation in grading of ductal carcinoma in situ of the breast: a nationwide study of 4901 patients in the Netherlands. 2019;174(2):479-88.
22. Worni M, Akushevich I, Greenup R, Sarma D, Ryser MD, Myers ER, et al. Trends in Treatment Patterns and Outcomes for Ductal Carcinoma In Situ. Journal of the National Cancer Institute. 2015; 107(12):djv263.
23. Eusebi V, Foschini MP, Cook MG, Berrino F, Azzopardi JG. Long-term follow-up of in situ carcinoma of the breast with special emphasis on clinging carcinoma. Seminars in diagnostic pathology. 1989;6(2):165-73.
24. Eusebi V, Feudale E, Foschini MP, Micheli A, Conti A, Riva C, et al. Long-term follow-up of in situ carcinoma of the breast. Semin Diagn Pathol. 1994;11(3):223-35.
25. Stuart KE, Houssami N, Taylor R, Hayen A, Boyages J. Long-term outcomes of ductal carcinoma in situ of the breast: a systematic review, meta-analysis and meta-regression analysis. BMC Cancer. 2015;15:890.
26. Giannakeas V, Sopik V, Narod SA. Association of a Diagnosis of Ductal Carcinoma In Situ With Death From Breast Cancer. JAMA Network Open. 2020;3(9):e2017124-e.
27. Francis A, Thomas J, Fallowfield L, Wallis M, Bartlett JM, Brookes C, et al. Addressing overtreatment of screen detected DCIS; the LORIS trial. European journal of cancer (Oxford, England: 1990). 2015; 51(16):2296-303.
28. Elshof LE, Tryfonidis K, Slaets L, van Leeuwen-Stok AE, Skinner VP, Dif N, et al. Feasibility of a prospective, randomised, open-label, international multicentre, phase III, non-inferiority trial to assess the safety of active surveillance for low risk ductal carcinoma in situ - The LORD study. European journal of cancer (Oxford, England : 1990). 2015;51(12):1497-510.
29. Lippey J, Spillane A, Saunders C. Not all ductal carcinoma in situ is created equal: can we avoid surgery for low-risk ductal carcinoma in situ? ANZ journal of surgery. 2016;86(11):859-60.
30. Hwang ES, Hyslop T, Lynch T, Frank E, Pinto D, Basila D, et al. The COMET (Comparison of Operative versus Monitoring and Endocrine Therapy) trial: a phase III randomised controlled clinical trial for low-risk ductal carcinoma in situ (DCIS). BMJ open. 2019;9(3):e026797.
31. Van Bockstal MR, Agahozo MC, Koppert LB, van Deurzen CHM. A retrospective alternative for active surveillance trials for ductal carcinoma in situ of the breast. International journal of cancer. 2020; 146(5): 1189-97.
32. Rosenberg SM, Partridge AH. Management of breast cancer in very young women. Breast. 2015;24 Suppl 2:S154-8.
33. Llarena NC, Estevez SL, Tucker SL, Jeruss JS. Impact of Fertility Concerns on Tamoxifen Initiation and Persistence. Journal of the National Cancer Institute. 2015;107(10).
34. Voci A, Bandera B, Ho E, Lee J, Goldfarb M, DiNome M. Variations in cancer care for adolescents and young adults (AYAs) with ductal carcinoma in situ. The breast journal. 2018;24(4):555-60.
35. Hawley ST, Griggs JJ, Hamilton AS, Graff JJ, Janz NK, Morrow M, et al. Decision involvement and receipt of mastectomy among racially and ethnically diverse breast cancer patients. J Natl Cancer Inst. 2009;101(19):1337-47.
36. Jagsi R, Hawley ST, Griffith KA, Janz NK, Kurian AW, Ward KC, et al. Contralateral Prophylactic Mastectomy Decisions in a Population-Based Sample of Patients With Early-Stage Breast Cancer. JAMA Surg. 2017;152(3):274-82.
37. Correa C, McGale P, Taylor C, Wang Y, Clarke M, Davies C, et al. Overview of the randomized trials of radiotherapy in ductal carcinoma in situ of the breast. Journal of the National Cancer Institute Monographs. 2010;2010(41):162-77.
38. Byun DJ, Wu SP, Nagar H, Gerber NK. Ductal Carcinoma in Situ in Young Women: Increasing Rates of Mastectomy and Variability in Endocrine Therapy Use. Annals of surgical oncology. 2021;28(11): 6083-96.
39. Abdelwahab K, Ibrahim N, Hamdy O, Abdallah A, Zaid AM, Shetiwy M. Factors Affecting Shared Decision-Making in Breast Cancer Surgeries: Egyptian Perspective. International Journal of Cancer and Biomedical Research. 2021;5(1):165-70.
40. Abdallah A, Farouk O, Metwally IH, Zuhdy M, Hamdy M, Attia E, et al. Predictors and effectiveness of different biopsy techniques in spotting invasion in the clinically diagnosed breast ductal carcinoma in situ. Revista de Senología y Patología Mamaria. 2022; 35(2):73-82.
41. Cuzick J, Sestak I, Pinder SE, Ellis IO, Forsyth S, Bundred NJ, et al. Effect of tamoxifen and radiotherapy in women with locally excised ductal carcinoma in situ: long-term results from the UK/ANZ DCIS trial. The Lancet Oncology. 2011;12(1):21-9.
42. Wapnir IL, Dignam JJ, Fisher B, Mamounas EP, Anderson SJ, Julian TB, et al. Long-term outcomes of invasive ipsilateral breast tumor recurrences after lumpectomy in NSABP B-17 and B-24 randomized clinical trials for DCIS. Journal of the National Cancer Institute. 2011;103(6):478-88.
43. Staley H, McCallum I, Bruce J. Postoperative tamoxifen for ductal carcinoma in situ. The Cochrane database of systematic reviews. 2012;10:CD007847.
44. Allred DC, Anderson SJ, Paik S, Wickerham DL, Nagtegaal ID, Swain SM, et al. Adjuvant tamoxifen reduces subsequent breast cancer in women with estrogen receptor-positive ductal carcinoma in situ: a study based on NSABP protocol B-24. Journal of clinical oncology : official journal of the American Society of Clinical Oncology. 2012;30(12):1268-73.
45. Lo AC, Truong PT, Wai ES, Nichol A, Weir L, Speers C, et al. Population-based analysis of the impact and generalizability of the NSABP-B24 study on endocrine therapy for patients with ductal carcinoma in situ of the breast. Annals of oncology : official journal of the European Society for Medical Oncology. 2015;26(9):1898-903.
46. NCCN Clinical Practice Guidelines in Oncology. Breast cancer (version 2.2026). 2026.
47. Bijker N, Meijnen P, Peterse JL, Bogaerts J, Van Hoorebeeck I, Julien J-p, et al. Breast-conserving treatment with or without radiotherapy in ductal carcinoma-in-situ: ten-year results of European Organisation for Research and Treatment of Cancer randomized phase III trial 10853—a study by the EORTC Breast Cancer Cooperative Group and EORTC Radiotherapy Group. Journal of clinical oncology. 2006;24(21):3381-7.
48. Cuzick J, Sestak I, Pinder SE, Ellis IO, Forsyth S, Bundred NJ, et al. Effect of tamoxifen and radiotherapy in women with locally excised ductal carcinoma in situ: long-term results from the UK/ANZ DCIS trial. The Lancet Oncology. 2011;12(1):21-9.
49. Donker M, Litière S, Werutsky G, Julien JP, Fentiman IS, Agresti R, et al. Breast-conserving treatment with or without radiotherapy in ductal carcinoma In Situ: 15-year recurrence rates and outcome after a recurrence, from the EORTC 10853 randomized phase III trial. Journal of clinical oncology : official journal of the American Society of Clinical Oncology. 2013;31(32):4054-9.
50. Goodwin A, Parker S, Ghersi D, Wilcken N. Post-operative radiotherapy for ductal carcinoma in situ of the breast. The Cochrane database of systematic reviews. 2013(11):CD000563.
51. Wärnberg F, Garmo H, Emdin S, Hedberg V, Adwall L, Sandelin K, et al. Effect of radiotherapy after breast-conserving surgery for ductal carcinoma in situ: 20 years follow-up in the randomized SweDCIS Trial. J Clin Oncol. 2014;32(32):3613-8.
52. Chien JC, Liu WS, Huang WT, Shih LC, Liu WC, Chen YC, et al. Local treatment options for young women with ductal carcinoma in situ: A systematic review and meta-analysis comparing breast conserving surgery with or without adjuvant radiotherapy, and mastectomy. Breast. 2022;63:29-36.
53. Holmberg L, Garmo H, Granstrand B, Ringberg A, Arnesson LG, Sandelin K, et al. Absolute risk reductions for local recurrence after postoperative radiotherapy after sector resection for ductal carcinoma in situ of the breast. J Clin Oncol. 2008;26(8):1247-52.
54. Giannakeas V, Sopik V, Narod SA. Association of Radiotherapy With Survival in Women Treated for Ductal Carcinoma In Situ With Lumpectomy or Mastectomy. JAMA Network Open. 2018;1(4): e181100-e.
55. Kong I, Paszat L, Saskin R, Taylor C, Nofech-Moses S, Hanna W, et al. Outcomes of Young Women with Ductal Carcinoma In Situ Treated with Breast-conserving Surgery and Radiotherapy: A Population-based Analysis. International Journal of Radiation Oncology Biology Physics. 2010;78(3):S99.
56. Fisher B, Costantino J, Redmond C, Fisher E, Margolese R, Dimitrov N, et al. Lumpectomy compared with lumpectomy and radiation therapy for the treatment of intraductal breast cancer. N Engl J Med. 1993;328(22):1581-6.
57. Solin LJ, Kurtz J, Fourquet A, Amalric R, Recht A, Bornstein BA, et al. Fifteen-year results of breast-conserving surgery and definitive breast irradiation for the treatment of ductal carcinoma in situ of the breast. J Clin Oncol. 1996;14(3):754-63.
58. Vicini FA, Kestin LL, Goldstein NS, Chen PY, Pettinga J, Frazier RC, et al. Impact of young age on outcome in patients with ductal carcinoma-in-situ treated with breast-conserving therapy. Journal of clinical oncology : official journal of the American Society of Clinical Oncology. 2000;18(2):296-306.
59. Tunon-de-Lara C, Lemanski C, Cohen-Solal-Le-Nir C, de Lafontan B, Charra-Brunaud C, Gonzague-Casabianca L, et al. Ductal carcinoma in situ of the breast in younger women: a subgroup of patients at high risk. European journal of surgical oncology : the journal of the European Society of Surgical Oncology and the British Association of Surgical Oncology. 2010;36(12):1165-71.
60. Cronin PA, Olcese C, Patil S, Morrow M, Van Zee KJ. Impact of Age on Risk of Recurrence of Ductal Carcinoma In Situ: Outcomes of 2996 Women Treated with Breast-Conserving Surgery Over 30 Years. Annals of surgical oncology. 2016;23(9):2816-24.
Full Text Sources:
Abstract:
Views: 91

For Authors
Journal Subscriptions

Dec 2025
Supplements
Instructions for authors
Online submission
Contact
e-ISSN: 2601 - 1700 (online)
ISSN-L: 2559 - 723X
Journal Abbreviation: Surg. Gastroenterol. Oncol.
Surgery, Gastroenterology and Oncology (SGO) is indexed in:
- SCOPUS
- EBSCO
- DOI/Crossref
- Google Scholar
- SCImago
- Harvard Library
- Open Academic Journals Index (OAJI)
Surgery, Gastroenterology and Oncology (SGO) is an open-access, peer-reviewed online journal published by Celsius Publishing House. The journal allows readers to read, download, copy, distribute, print, search, or link to the full text of its articles.
Time to first editorial decision: 25 days
Rejection rate: 61%
CiteScore: 0.2

Meetings and Courses in 2025
Meetings and Courses in 2024
Meetings and Courses in 2023
Meetings and Courses in 2022
Meetings and Courses in 2021
Meetings and Courses in 2020
Meetings and Courses in 2019
Verona expert meeting 2019

Surgery, Gastroenterology and Oncology applies the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits readers to copy and redistribute the material in any medium or format, remix, adapt, build upon the published works non-commercially, and license the derivative works on different terms, provided the original material is properly cited and the use is non-commercial. Please see: https://creativecommons.org/licenses/by-nc/4.0/
Publisher’s Note:
The opinions, statements, and data contained in article are solely those of the authors and not of Surgery, Gastroenterology and Oncology journal or the editors. Publisher and the editors disclaim responsibility for any damage resulting from any ideas, instructions, methods, or products referred to in the content.
 IASGO Society News
IASGO Society News