Surgery, Gastroenterology and Oncology

|
|
Background: Anal sphincteric injury is an uncommon but clinically significant condition associated with substantial short- and long-term morbidity, most notably anal incontinence and impaired quality of life. This study systematically compares immediate (?48 hours) versus delayed (>48 hours) repair strategies regarding postoperative morbidity, mortality, and functional outcomes, including fecal continence, infection rates, colostomy requirements, hospitalization duration, fistula formation, and reoperation rates.
Methods: This prospective study enrolled 30 patients (aged 5–60 years) with non-obstetric anal sphincter injuries involving ?50% circumferential disruption. Participants were stratified into Group I (immediate repair, ?48 hours post-injury) and Group II (delayed repair, >48 hours). All patients underwent comprehensive preoperative assessments comprising detailed clinical examination, laboratory investigations, and advanced imaging (CT, MRI, endoanal ultrasound in some cases).
Results: Demographic characteristics were comparable between immediate (Group I) and delayed (Group II) repair groups. Injury mechanisms diverged significantly (p=0.042): Group I followed high-energy trauma (motorbike 60%), presenting with bleeding (100%) and associated injuries (40%; p=0.006), while Group II comprised iatrogenic injuries (posthemorrhoidectomy 53.3%) with incontinence (100%). Immediate repairs involved larger/ multiple defects (26.7%; p<0.001 size, p=0.03 number) and universal colostomy (100% vs 26.7%; p<0.001). End-to-end repair predominated (80%), with overlap techniques more frequent in delayed cases (33.3%; p=0.17). Continence favored delayed repairs at 3 months (Wexner 2.46 vs 5; p=0.02), converging by 6 months (2.33 vs 3.6; p=0.15). Six-month continence resumption associated significantly with male sex (p=0.004), delayed timing (p=0.025), no colostomy (p=0.005), and incontinence presentation (p=0.025). Trends favored post-hemorrhoidectomy trauma (p=0.129) and single defects (p=0.073).
Conclusions: Delayed repair provided superior early continence, lower colostomy requirements, and reduced dehiscence compared to immediate repair, achieving equivalent 6-month sphincter function. Male sex, delayed timing, absence of colostomy, and incontinence presentation emerged as key predictors of continence resumption.
INTRODUCTION
Optimal management of anorectal sphincter injuries, whether traumatic, iatrogenic, or obstetric, presents formidable clinical challenges encompassing primary anatomic reconstruction, sustained continence restoration, and comprehensive psychosocial rehabilitation.
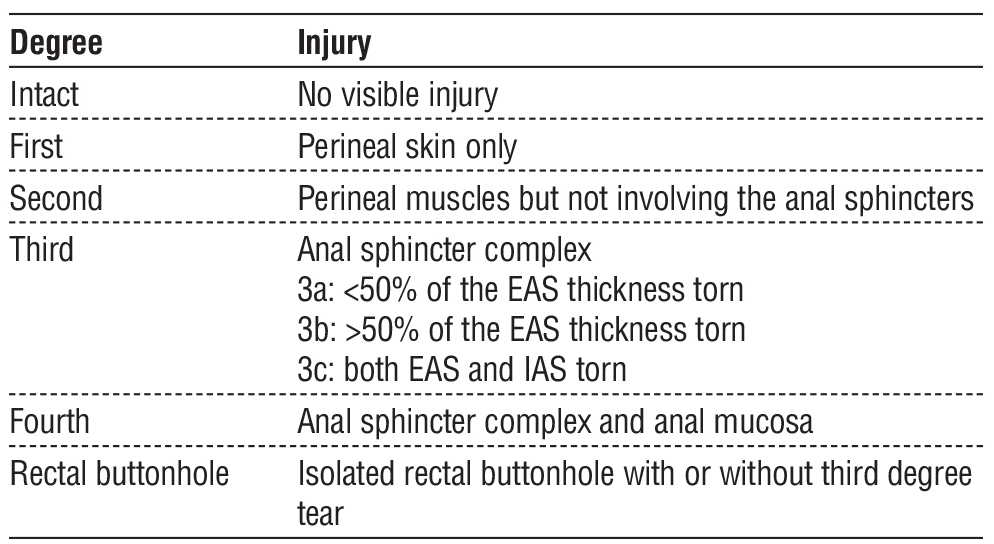
Its epidemiology spans both obstetric and non-obstetric etiologies, including vaginal delivery, blunt and penetrating perineal trauma, road traffic and motorcycle accidents, sexual trauma, and iatrogenic injury following anorectal or pelvic surgery. While obstetric anal sphincter injuries remain the most frequently reported, non-obstetric injuries account for a considerable proportion of cases, particularly among males and younger patients, and are often underdiagnosed. The reported prevalence varies widely depending on mechanism and diagnostic modality, ranging from approximately 1–11% of vaginal deliveries to up to 2–5% of patients with perineal or pelvic trauma, with higher rates detected when imaging is routinely employed (1,2). Anal sphincteric injuries may involve the internal anal sphincter, external anal sphincter, or both, and range from partial defects to complete anorectal disruption. The most widely adopted anatomical classification is proposed by the Royal College of Obstetricians and Gynaecologists, which categorizes injuries according to the extent of sphincter and rectal wall involvement and, although originally developed for obstetric trauma, is commonly applied to non-obstetric injuries to standardize diagnosis, guide management, and facilitate comparison of outcome (3) (table 1).
Table 1 - Classification of Ano-Rectal Injury
While immediate repair constitutes standard practice for anterior obstetric sphincter tears (4), technical limitations such as muscle retraction compromise outcomes by increasing suture-line tension, inducing local ischemia, and predisposing to dehiscence or repair failure (5). These complications frequently necessitate secondary interventions specifically designed to accommodate scarred, distorted anatomy and mitigate high-tension closures.
Post-traumatic fecal incontinence management centers on sphincteroplasty, mobilization and primary reapproximation of disrupted musculature, which enjoys broad literature support (6,7). When direct repair proves anatomically infeasible, augmentation strategies including dynamic graciloplasty (8) or sphincter reefing may restore continence. In acute, high-grade perineal trauma, fecal diversion via colostomy remains essential to control contamination and facilitate staged reconstruction (9).
Sphincter injuries of obstetric or iatrogenic origin often evade acute detection (10,11), with fecal incontinence emerging only during delayed phases, severely compromising quality of life and intensifying psychosocial distress (12). This study systematically compares immediate versus delayed sphincter repair, evaluating morbidity, mortality, and functional outcomes, including continence, infection rates, colostomy requirements, hospitalization duration, fistula formation, and reoperation, to inform evidence-based optimization of surgical timing and technique selection.
PATIENTS AND METHODS
This prospective clinical study evaluated 30 patients (aged 5-60 years) with nonobstetric traumatic anal sphincter injuries involving <50% of the sphincter circumference. Conducted at Assiut University Hospitals, Egypt, from March 2020 to December 2022, the study received institutional ethical approval, and informed consent was obtained from all participants. The primary objective was to assess the impact of surgical timing on postoperative morbidity and functional continence outcomes.
Patients were stratified by repair timing: Group I underwent immediate repair (?48 hours post-injury), and Group II received delayed repair (>48 hours). Patients were allocated to immediate or delayed anal sphincter repair based on predefined clinical and institutional parameters. Immediate repair (?48 hours) was performed in patients presenting with active perineal or anorectal bleeding, acute sphincter disruption with viable, non-infected tissue, or associated injuries requiring urgent surgical exploration, provided that adequate institutional resources and an experienced colorectal surgeon were available. Patients who presented early after injury with acceptable tissue conditions and no contraindications to anesthesia were also considered for immediate repair. Delayed repair (>48 hours) was selected for patients who were hemodynamically unstable requiring initial resuscitation, had significant perineal contamination, edema, infection, or tissue necrosis, presented late after injury, or when specialized colorectal surgical expertise or operating room resources were not immediately available. In such cases, initial management focused on stabilization, infection control, wound care, and, when indicated, fecal diversion, with definitive sphincter reconstruction deferred until local and systemic conditions were optimized.
All patients underwent standardized preoperative evaluation, including detailed clinical examination, advanced imaging (CT, MRI, endoanal ultrasound in some cases), and comprehensive laboratory assessment. Prophylactic antibiotics were administered universally.
Continence evaluation using the validated Wexner score was applied exclusively to the delayed group (table 2), as acute-phase assessment proved infeasible in immediate repairs due to edema and colostomy diversion. Surgical approaches were tailored to injury mechanism and anatomical disruption, encompassing both internal/external sphincters, anorectal canal, vaginal/gluteal tissues, and associated systemic trauma (thoracic, pelvic, vascular). Spinal anesthesia predominated, with general anesthesia reserved for extensive abdominal or multisystem injuries. Repair techniques comprised overlapping sphincteroplasty and end-to-end anastomosis, with defunctioning colostomy performed in all Group I patients and selectively (n=4) in Group II.
Table 2 - The Jorge-Wexner incontinence score

Perineal wounds underwent meticulous debridement of devitalized tissue to prevent necrosis and pelvic sepsis. Postoperative follow-up spanned six months, with evaluations at 1 and 3 months focusing on wound healing and subjective continence (colostomy-based assessment). At six months, objective continence was quantified using Wexner scores and digital rectal examination. Five patients (four from Group I, one from Group II) required adjunctive physiotherapy to optimize functional recovery.
Statistical Analysis
Statistical analyses were conducted using SPSS version 22.0 (IBM Corp., Armonk, NY, USA). Continuous data were summarized as means ± standard deviations (SD) or medians (interquartile ranges) based on normality assessment via Shapiro-Wilk tests. Categorical variables were expressed as frequencies and percentages.
Comparisons between immediate and delayed repair groups employed independent-samples t-tests or Mann-Whitney U tests for continuous outcomes (e.g., age, Wexner scores, hospital stay) and Pearson chi-square or Fisher's exact tests for categorical variables (e.g., sex, trauma type, complications), with exact tests applied when expected cell counts were <5. Associations between predictors (e.g., timing, colo-stomy) and continence resumption at 6 months used chi-square tests (Pearson or Fisher’s exact as appropriate). Statistical significance was defined as p<0.05 (two-tailed).
RESULTS
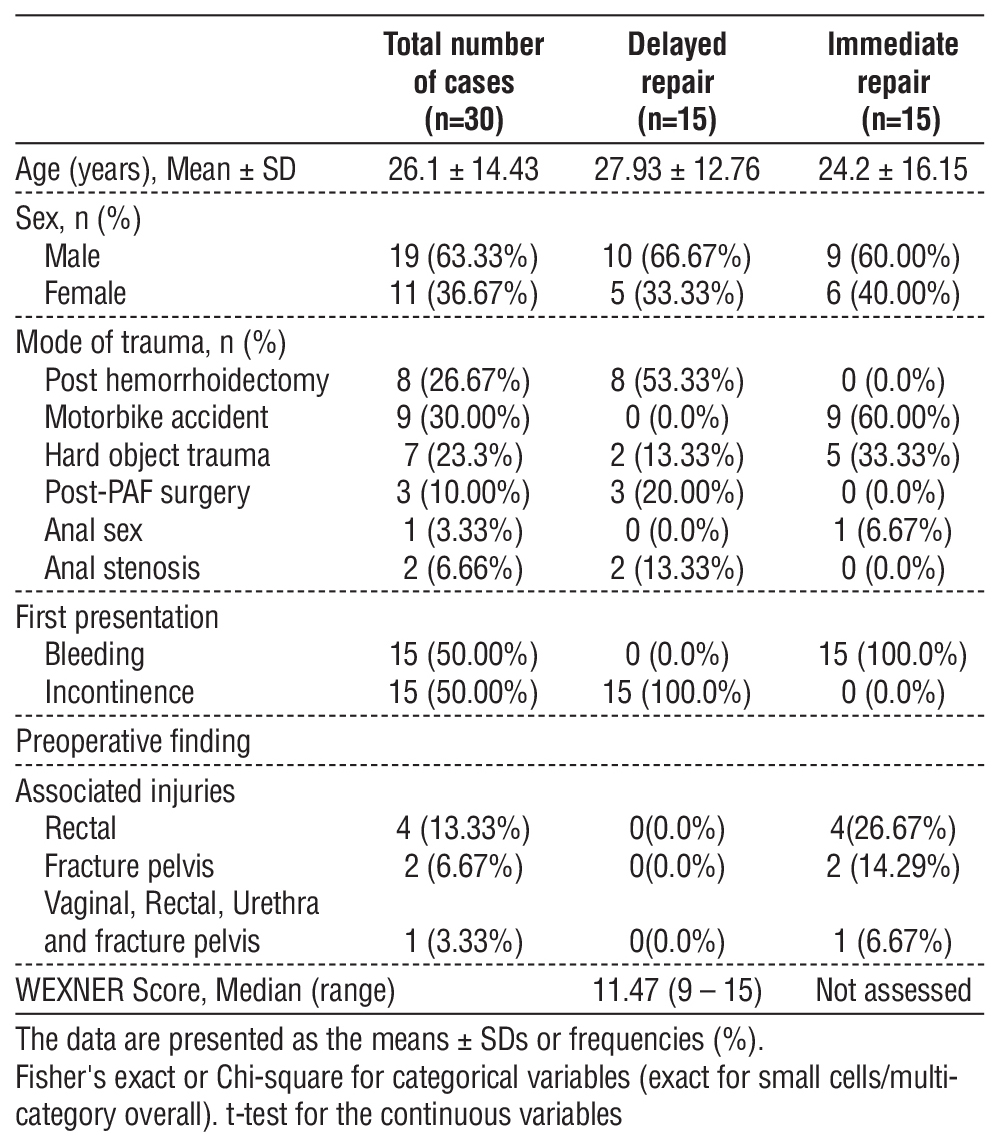
The demographic characteristics and preoperative findings of patients undergoing immediate versus delayed repair for traumatic anal sphincteric injury are summarized in table 3. The delayed group demonstrated a slightly higher mean age (27.93±12.76 years vs. 24.2±16.15 years), but this difference was not significant (p=0.54). Males predominated in both cohorts (66.67% vs. 60%), with no significant sex difference (p=0.89). Significant differences existed in mode of trauma (p=0.042): post-hemorrhoidectomy complications (53.33%) led in delayed repairs, whereas motorbike accidents were predominant in immediate cases (60%), followed by hard object trauma (33.33%). Anal stenosis occurred exclusively in delayed repairs (13.33%). Presentation differed significantly (p<0.001): active hemorrhage characterized all immediate cases (100%), while fecal incontinence defined all delayed presentations (100%). Associated injuries differed significantly (p=0.006), occurring only in immediate repairs [rectal trauma (26.67%), pelvic fractures (14.29%), and one complex case involving combined pelvic, rectal, and urethral injury (6.67%)]. Preoperative Wexner scores in the delayed group showed a median of 11.47 (range 9–15), reflecting continence loss.
Table 3 - Demographic data and preoperative findings are presented in this table.
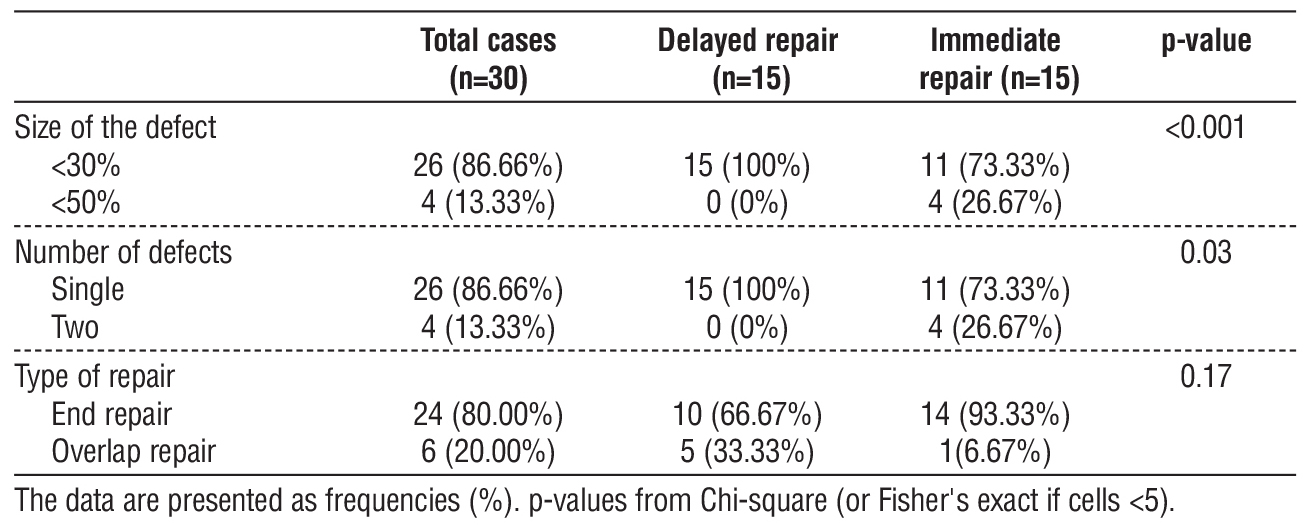
Intraoperative findings are detailed in table 4. Defect size differed significantly (p<0.001): defects <30% dominated both groups, but immediate repairs involved larger defects (<50%) in 26.67% of cases versus 0% in delayed repairs. Number of defects also differed significantly (p=0.03): single defects were universal in delayed repairs (100%) versus 73.33% in immediate cases, with multiple defects exclusive to immediate repairs (26.67%). End-to-end repair predominated overall (80.00%), especially in immediate cases (93.33%) versus delayed cases (66.67%), but type of repair showed no significant difference (p=0.17); overlap techniques trended higher in delayed cases (33.33% vs 6.67%).
Table 4 - Intraoperative data
Postoperative outcomes are presented in table 5. At 1 month, the delayed group showed rapid continence improvement (median Wexner: 2.18, range 1–4). Immediate repair patients had longer hospital stays (median 7 vs. 5 days), but this was not significant (p=0.12). Colostomy use differed significantly (p<0.001), being universal in immediate repairs (100%) versus only 26.67% in delayed cases. Complications were comparable: superficial infection affected 26.67% of delayed versus 20.00% of immediate cases (p=0.48), and suture dehiscence occurred only in immediate repairs (13.33% vs 0%; p not significant).
Table 5 - The postoperative evaluation results
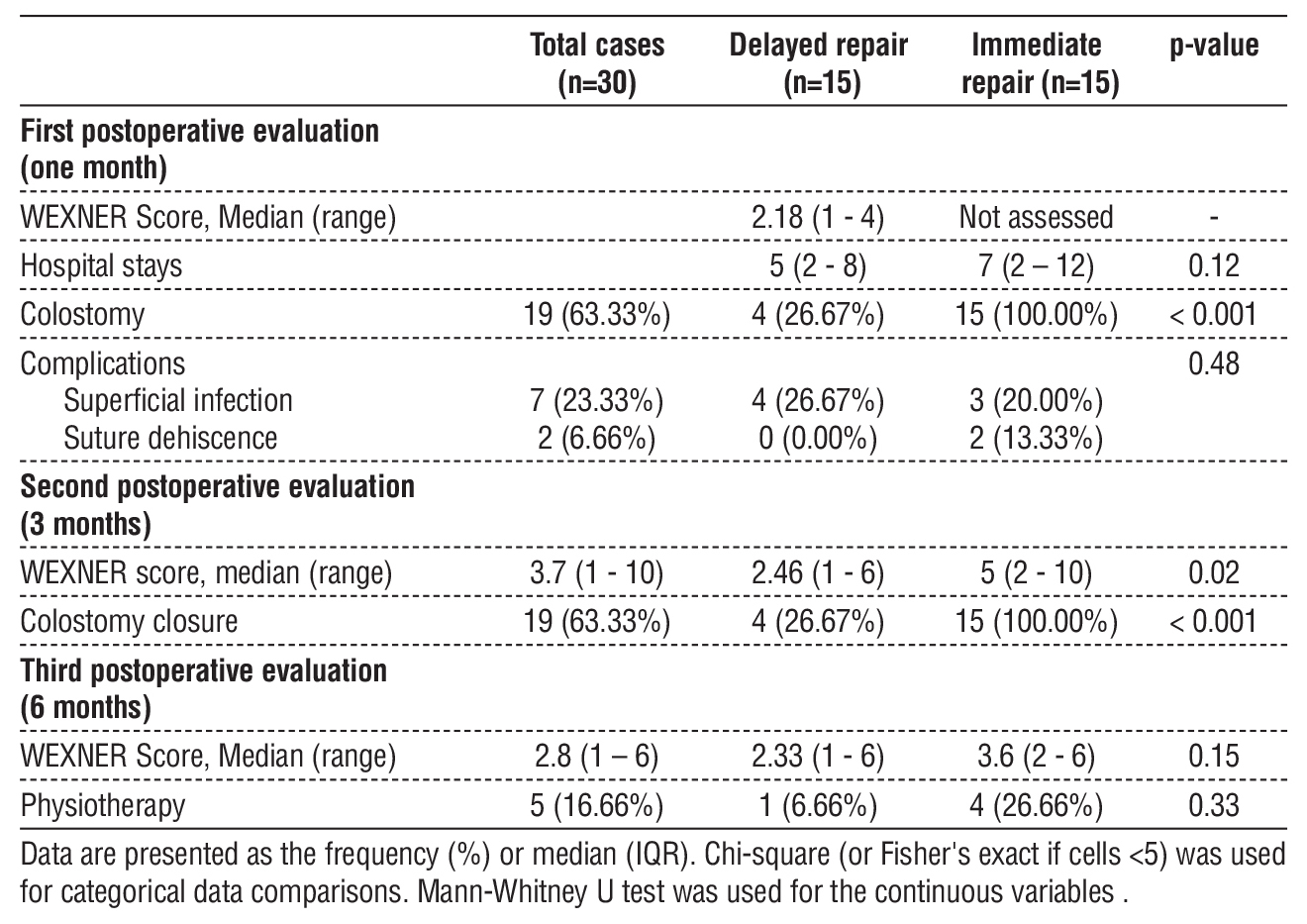
At 3 months, continence differed significantly (p=0.02), favoring delayed repairs (median Wexner: 2.46 vs. 5), with colostomy closure in 63.33% overall. By 6 months, scores converged (2.33 vs. 3.6; p=0.15, not significant). Physiotherapy trended higher in immediate repairs (26.66% vs 6.66%), but this was not significant (p=0.33).
Factors associated with resumption of sphincteric continence at 6 months are detailed in tables 6-8.
Table 6 showed male sex was significantly associated with continence (p=0.004): 91.7% of continent patients were male versus 38.9% of incontinent patients, while females comprised 61.1% of incontinent versus only 8.3% of continent cases. Presentation was significantly associated with continence (p=0.025): incontinence at presentation showed higher continence rates (75.0%) compared to bleeding (25.0%). Age was higher in continent patients (mean 30.0±15.9 years vs. 23.4±13.1 years), but this difference was not significant (p=0.229).
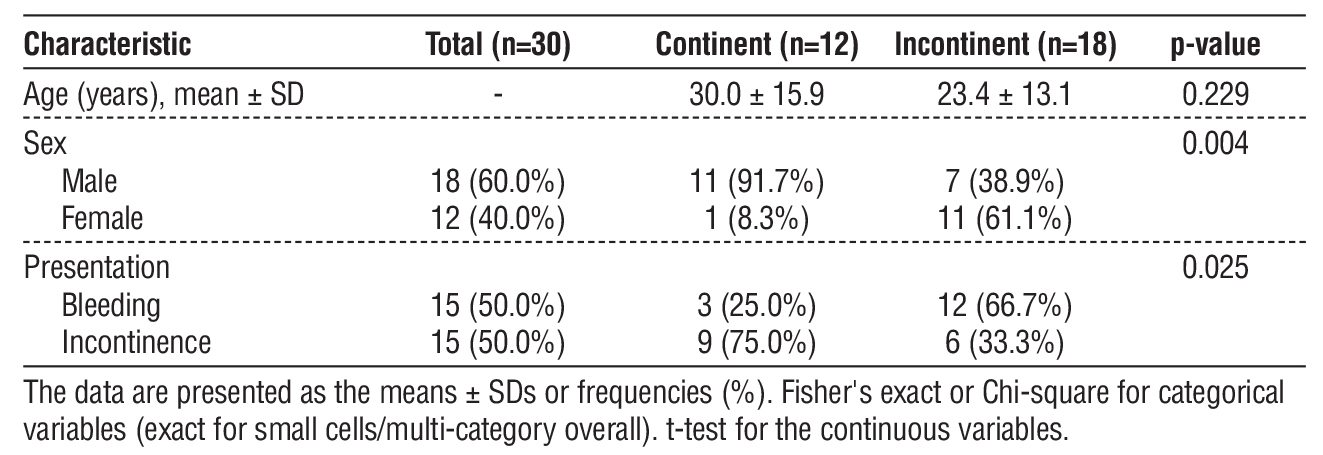
Table 6 - Baseline characteristics associated with resumption of sphincteric continence at 6 months.

Table 7 - Trauma and defect characteristics associated with resumption of sphincteric continence at 6 months.
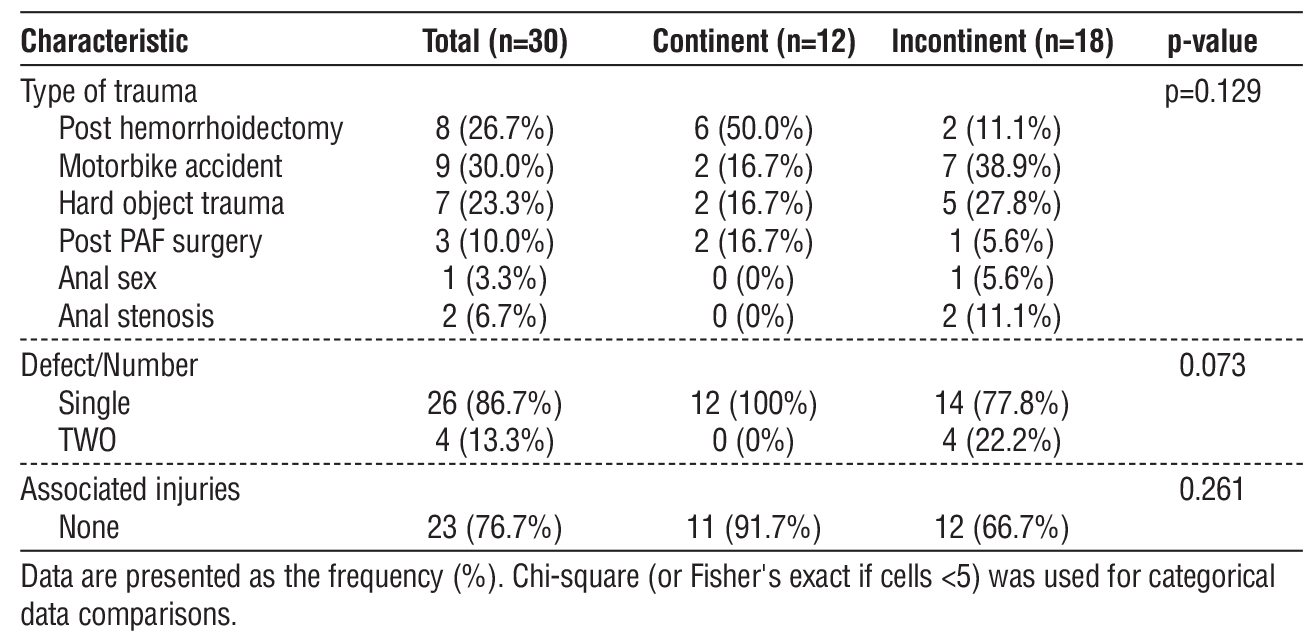
Table 8 identified significant procedure-related predictors of continence. Delayed repair was significantly associated with continence (p=0.025): 75.0% of continent patients had delayed versus 33.3% of incontinent patients, while immediate repair predominated in incontinent cases (66.7%). Absence of colo-stomy was significantly associated with continence (p=0.005): 66.7% of continent patients had no colo-stomy versus only 16.7% of incontinent patients.
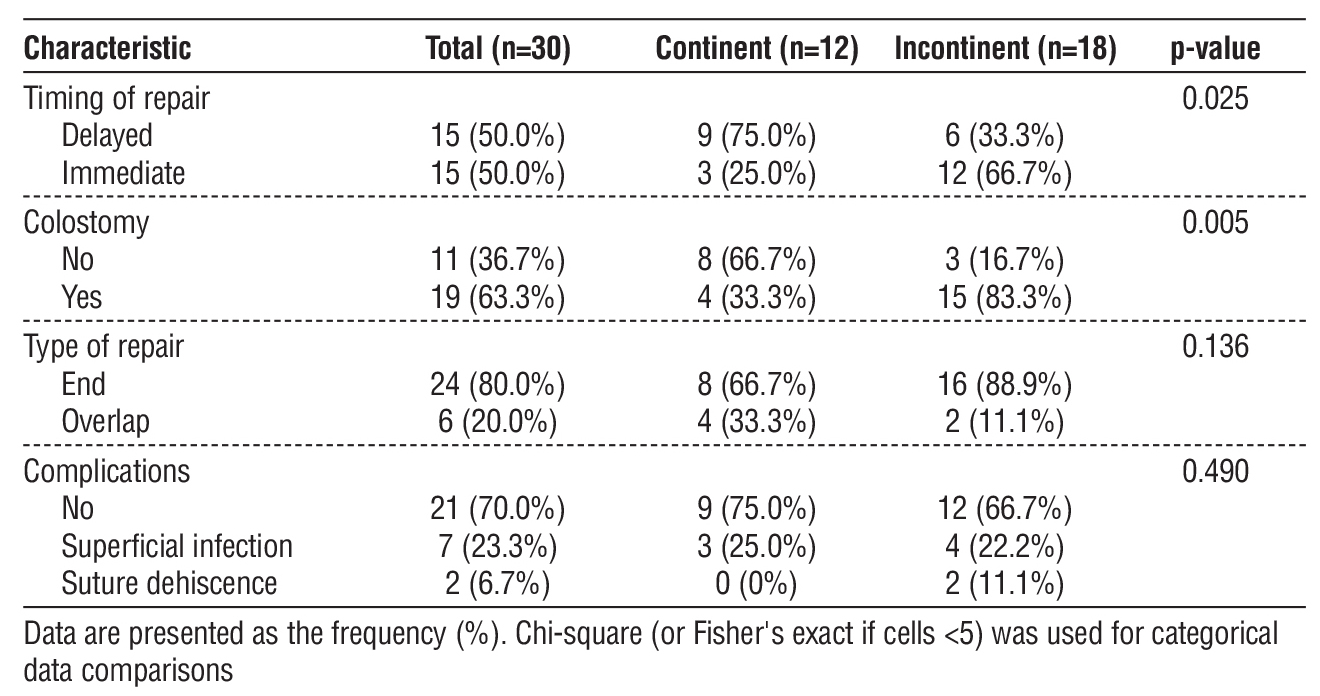
Type of repair showed overlap repairs with higher continence (33.3% vs 11.1%), but this was not significant (p=0.136). Complications showed no significant association with continence (p=0.490).
DISCUSSION
Traumatic anal sphincter injury (ASI) represents a significant clinical challenge, profoundly impacting anorectal continence, psychosocial well-being, and quality of life. These injuries commonly arise from obstetric complications, iatrogenic procedures, or high-energy trauma, often accompanied by concomitant pelvic or systemic injuries (13). Surgical restoration of sphincter integrity constitutes the cornerstone of management, yet remains technically demanding due to edema, tissue loss, contamination, and patient comorbidities. Considerable controversy persists regarding optimal repair timing, with outcomes shaped by injury severity, surgeon experience, institutional resources, and individual patient factors (13).
Immediate repair, typically performed within 24-48 hours, promotes anatomic restoration, minimizes fibrosis, and reduces infection risk (14,15). However, it poses technical challenges from intraoperative edema and bleeding, requiring specialized colorectal expertise and substantial resources (16). Conversely, delayed repair, generally executed 6-12 weeks post-injury (7,17), offers improved tissue visualization and handling but risks fistula formation, persistent pain, prolonged recovery, and psychological burden (12).
This study classified repairs as immediate (?48 hours) or delayed (>48 hours), guided by hemodynamic stability, tissue condition, and resource availability. As illustrated in fig. 1, motorbike accidents dominated immediate repairs (60.0%), while iatrogenic injuries —particularly post-hemorrhoidectomy complications (53.3%)—prevailed in delayed cases. Immediate presentations universally featured active hemorrhage (100%), whereas delayed cases manifested exclusively as fecal incontinence (100%), reflecting progressive sphincter dysfunction (18).
Figure 1 - A case of female patient 36 years old, with delayed repair for post haemorrhoidectomy anal sphincteric injury. (a) intraoperative; (b) postoperative.
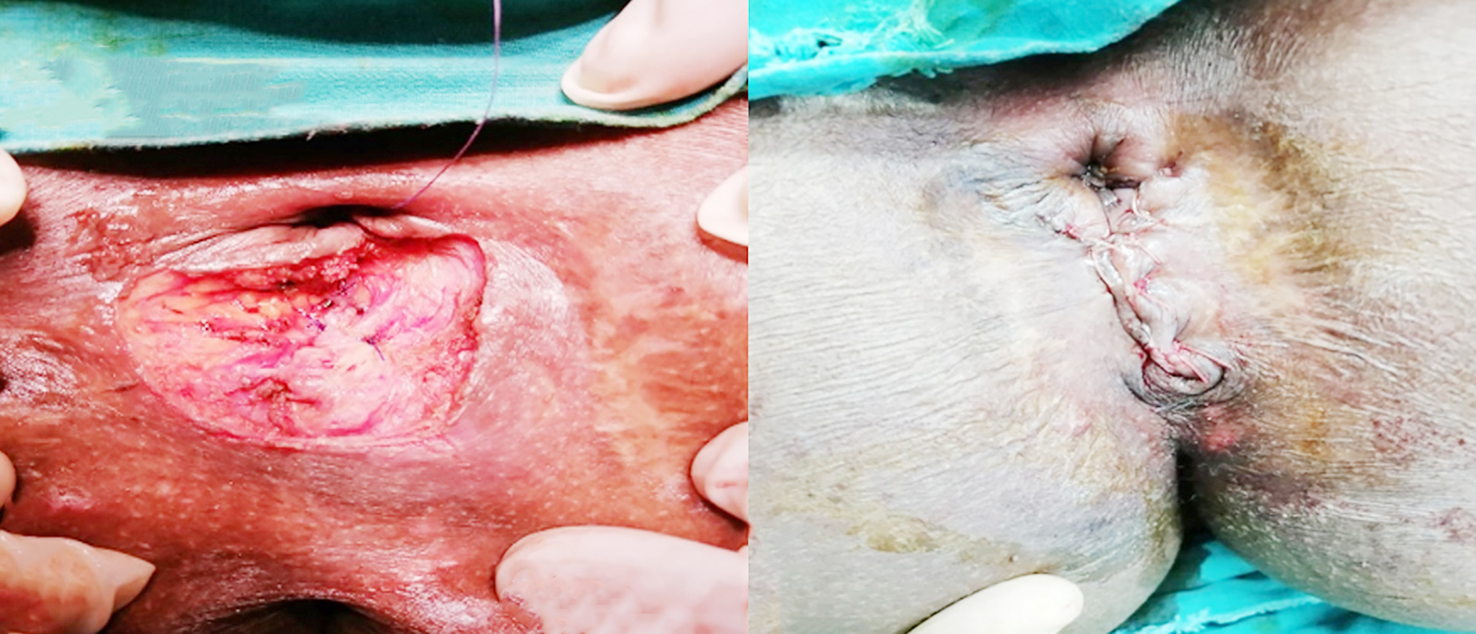
Figure 2 - A case of female child patient 8 years old with immediate repair for post hard object traumatic anal sphincteric injurt. (a) preoperative, (b, c) intraoperative, and (d) postoperative.
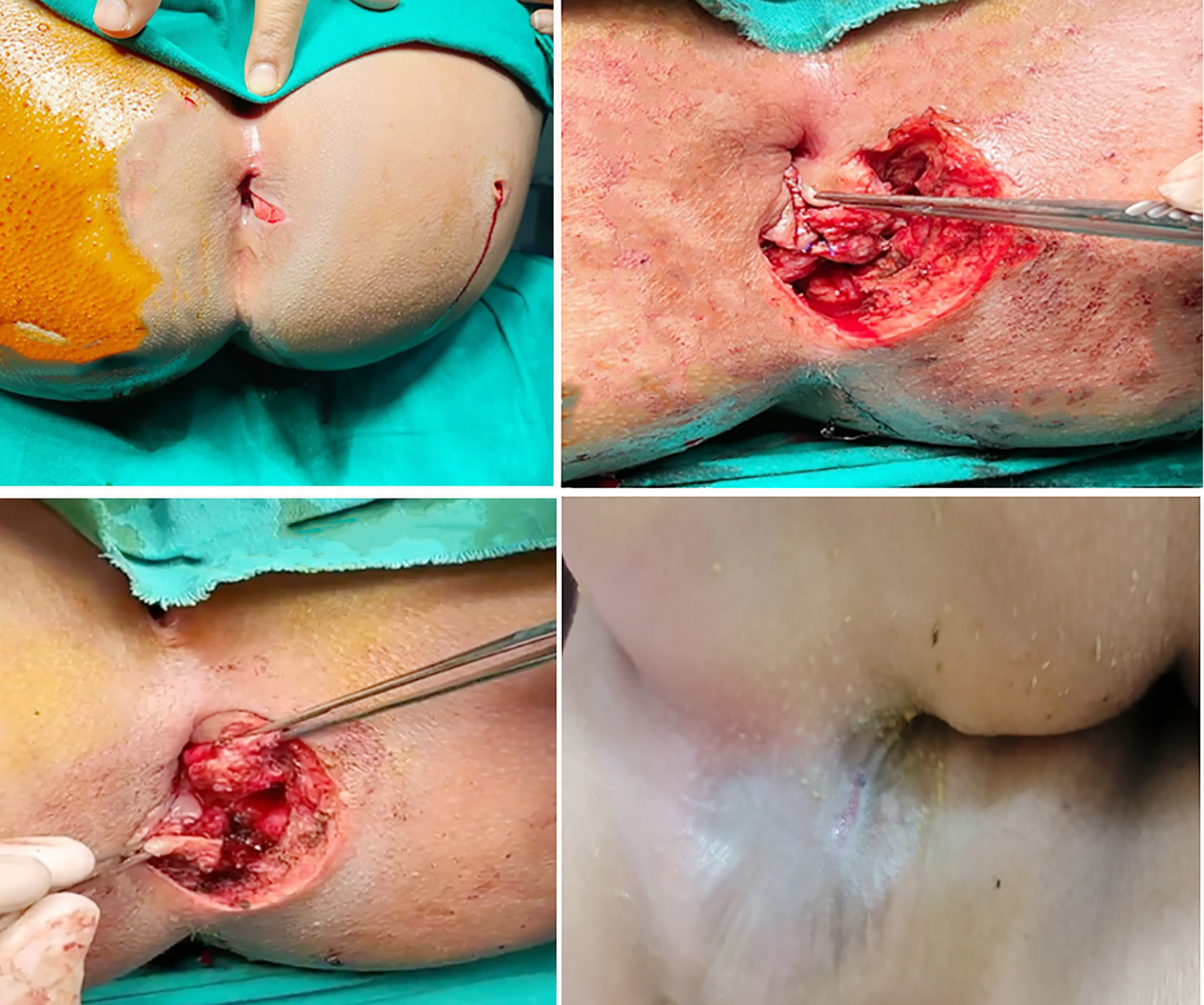
Functional assessment employed the validated Wexner continence score, applicable only to delayed cases due to acute-phase constraints. Preoperative scores indicated severe impairment (median 11.47, range 9-15). At 3 months, delayed repairs demonstrated superior continence (median Wexner 2.46 vs. 5; p=0.02), though scores converged by 6 months (2.33 vs. 3.6; p=0.15), consistent with findings from Nordenstam et al. (14) and Barisic et al. (19), despite reports of progressive functional decline in other cohorts.
Surgical technique selection reflected timing and tissue characteristics. End-to-end repair predominated in immediate cases (93.3%) due to acute tissue constraints, while overlap sphincteroplasty trended higher in delayed repairs (33.3% vs. 6.7%; p=0.17). Although overlap techniques demonstrate superior long-term continence in meta-analyses (17,19,20), no significant differences emerged here.
Colostomy requirements differed starkly: universal in immediate repairs (100% vs. 26.7%; p<0.001), underscoring acute contamination risks and supporting selective diversion strategies (16,18). Postoperative physiotherapy proved more frequent in immediate cases (26.7% vs. 6.7%; p=0.33), emphasizing structured rehabilitation needs in complex trauma.
Complication profiles varied predictably. Immediate repairs experienced suture dehiscence (13.3% vs. 0%) and comparable infection rates (20.0% vs. 26.7%; p=0.48), while delayed repairs avoided dehiscence entirely (21,22). Hospital stay duration favored delayed intervention (median 5 vs. 7 days; p=0.12), aligning with literature documenting extended acute-phase hospitalizations (16).
CONCLUSIONS
Both immediate and delayed sphincter repairs achieved satisfactory 6-month continence restoration. Delayed repair offered early advantages, superior 3-month continence, reduced colostomy requirements, and no dehiscence, while immediate repair remains essential for patients with active hemorrhage. Optimal outcomes necessitate individualized assessment, multidisciplinary care, and sustained follow-up, consistent with contemporary guidelines.
Acknowledgment
I would like to extend my heartfelt gratitude to Professor Dr-Ahmed Abdel Aziz Abou-Zeid for his invaluable participation and support throughout this work. His insights, feedback, and collaboration greatly enriched this study, and his contributions are deeply appreciated.
Author’s Contributions
Mahmoud Refaat Shehata: Supervision, Formal analysis, Revision, Review & editing manuscript.
Abd El-Moniem El-Khateeb: Supervision, Formal analysis, Review&editing manuscript.
Ahmed Youssef Mohamed Ali: Conceptualization, Methodology, Follow-up, Investigation, Statistical analysis of the results and Writing – review editing original draft.
Conflict of Interest
The authors declares no conflict of interests.
Source of Funding
No funding was received for this study.
Ethical Approval
Study approved by Assuit University, faculty of medicine institutional review board (IRB approval
number 17101619) at assuit university, Egypt and was conducted in the general surgery department at assuit university hospitals.
Consent to Participate
Written informed consent from all patients included in the study.
Availability of Data and Material
The corresponding author will provide the datasets used or analyzed during the current work upon reasonable requests.
REFERENCES
1. Dudding TC, Vaizey CJ, Kamm MA. Obstetric and traumatic anal sphincter injury: incidence, risk factors, and management. Ann Surg. 2008;247(2):224-37.
2. Spinelli A, Laurenti V, Carrano FM, Gonzalez-Díaz E, Borycka-Kiciak K. Diagnosis and treatment of anal sphincter injuries. J Clin Med. 2021;10(15):3261.
3. Royal College of Obstetricians and Gynaecologists (RCOG). Management of third- and fourth-degree perineal tears (Green-top Guideline No. 29). RCOG Press. [Updated 2020]. 2015.
4. Fernando RJ, Sultan AH, Kettle C, Radley S, Jones P, O’Brien PM. Repair techniques for obstetric anal sphincter injuries: a randomized controlled trial. Obstet Gynecol. 2006;107(6):1261–8.
5. Londono-Schimmer EE, Garcia-Duperly R, Nicholls RJ, Ritchie JK, Hawley PR, Thomson JPS. Overlapping anal sphincter repair for fecal incontinence due to sphincter trauma: five year follow-up
functional results. Int J Colorectal Dis. 1994;9(2):110–3.
6. Laalim SA, Hrora A, Raiss M, Ibnmejdoub K, Toughai I, Ahallat M, et al. Direct sphincter repair: technical issues, indications and results. Pan Afr Med J. 2013:14:11. French
7. Malouf AJ, Norton CS, Engel AF, Nicholls RJ, Kamm MA. Long-term results of overlapping anterior anal-sphincter repair for obstetric trauma. Lancet. 2000;355(9200):260-5.
8. Baeten CGMI, Geerdes BP, Adang EMM, Heineman E, Konsten J, Engel GL, et al. Anal Dynamic Graciloplasty in the Treatment of Intractable Fecal Incontinence. N Engl J Med. 1995;332(24):1600–5.
9. Steele SR, Hull TL, Hyman N, Maykel JA, Read TE, Whitlow CB. The ASCRS textbook of colon and rectal surgery. Springer Nature; 2022.
10. Andrews V, Sultan AH, Thakar R, Jones PW. Occult anal sphincter injuries - myth or reality? BJOG. 2006;113(2):195-200.
11. Sultan AH, Kamm MA, Hudson CN, Thomas JM, Bartram CI. Anal-Sphincter Disruption during Vaginal Delivery. N Engl J Med. 1993;329(26):1905–11.
12. Bartlett L, Nowak M, Ho YH. Impact of fecal incontinence on quality of life. World J Gastroenterol. 2009;15(26):3276-82.
13. Saad DA, Elhiny AA. Outcomes of Anal Sphincter Injury Repair; Minia University Hospital Experience Prospective Study. Minia J Med Res. 2020;31(4):110–21.
14. Nordenstam J, Mellgren A, Altman D, López A, Johansson C, Anzén B, et al. Immediate or Delayed Repair of Obstetric Anal Sphincter Tears - A Randomized Controlled Trial. Obstet Gynecol Surv. 2008; 63(11):686–7.
15. Ko?ci?ski T. Obstetric damage to the perineum and the anal sphincters. Reconstruction surgery. Ginekol Pol. 2014;85(8): 624-8.
16. Mohamed AN, Tohamy TA, Saad Kamel DA, Allah MK. Perineal trauma & Anal Sphincter Injuries: Treatment Outcomes in Minia University Emergency Unit. Minia J Med Res. 2022;33(2):62–72.
17. Fernando RJ. Management of third-and fourth-degree perineal tears following vaginal delivery. Royal College of Obstetricians and Gynecologists; 2001.
18. Di?çi E, Peksöz R, Y?ld?z M, Y?ld?rgan ?, Fakirullaho?lu M, Albayrak Y, et al. Management and outcomes of anal sphincter injuries: A retrospective cohort study. J Surg Med. 2022;6(3):254–7.
19. Barisic GI, Krivokapic ZV, Markovic VA, Popovic MA. Outcome of overlapping anal sphincter repair after 3 months and after a mean of 80 months. Int J Colorectal Dis. 2006 Jan;21(1):52–6.
20. Chase S, Mittal R, Jesudason MR, Nayak S, Perakath B. Anal sphincter repair for fecal incontinence: experience from a tertiary care center. Indian J Gastroenterol. 2010 Jul;29(4):162–5.
21. Okeahialam NA, Wong KW, Thakar R, Sultan AH. The incidence of wound complications following primary repair of obstetric anal sphincter injury: a systematic review and meta-analysis. Am J Obstet Gynecol. 2022;227(2):182–91.
22. Barbosa M, Glavind?Kristensen M, Moller Soerensen M, Christensen P. Secondary sphincter repair for anal incontinence following obstetric sphincter injury: functional outcome and quality of life at 18 years of follow?up. Colorectal Dis. 2020;22(1):71–9.
Full Text Sources:
Abstract:
Views: 1569

For Authors
Journal Subscriptions

Dec 2025
Supplements
Instructions for authors
Online submission
Contact
e-ISSN: 2601 - 1700 (online)
ISSN-L: 2559 - 723X
Journal Abbreviation: Surg. Gastroenterol. Oncol.
Surgery, Gastroenterology and Oncology (SGO) is indexed in:
- SCOPUS
- EBSCO
- DOI/Crossref
- Google Scholar
- SCImago
- Harvard Library
- Open Academic Journals Index (OAJI)
Surgery, Gastroenterology and Oncology (SGO) is an open-access, peer-reviewed online journal published by Celsius Publishing House. The journal allows readers to read, download, copy, distribute, print, search, or link to the full text of its articles.
Time to first editorial decision: 25 days
Rejection rate: 61%
CiteScore: 0.2

Meetings and Courses in 2025
Meetings and Courses in 2024
Meetings and Courses in 2023
Meetings and Courses in 2022
Meetings and Courses in 2021
Meetings and Courses in 2020
Meetings and Courses in 2019
Verona expert meeting 2019

Surgery, Gastroenterology and Oncology applies the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits readers to copy and redistribute the material in any medium or format, remix, adapt, build upon the published works non-commercially, and license the derivative works on different terms, provided the original material is properly cited and the use is non-commercial. Please see: https://creativecommons.org/licenses/by-nc/4.0/
Publisher’s Note:
The opinions, statements, and data contained in article are solely those of the authors and not of Surgery, Gastroenterology and Oncology journal or the editors. Publisher and the editors disclaim responsibility for any damage resulting from any ideas, instructions, methods, or products referred to in the content.
 IASGO Society News
IASGO Society News