Surgery, Gastroenterology and Oncology

|
|
Background: Tailored axillary surgery (TAS) is a technique for clinically node-positive breast cancer (BC) that selectively removes nodal disease to convert a positive axilla to a negative, customizing surgery based on disease extent. Radiological assessment and ultrasound on the axilla have been used to identify suspicious LNs. Carbon ink injection tattooing is one popular method for axillary LNs. Objective: We evaluated the feasibility and validity of ultrasound-guided marking of metastatic axillary LNs in BC patients followed by TAS exhibiting as a method for reducing tumor burden.
Methods: This is a prospective observational cohort study that involved fifty patients with clinically node-positive (cN1) BC who underwent upfront or post-neo-adjuvant breast surgery. Participants underwent carbon particles (Black-Eye) injection in the suspicious node before surgery, aiming at TAS, followed by backup ALND.
Results: The mean age of the study participants was 54.48±9.06 years. The mean number of suspicious LNs detected by ultrasound was 2 (ranging from 1 to 3 in both upfront and post-neo-adjuvant cases). The mean number of LNs retrieved in the backup ALND was 16 (range, 7 to 31). The mean number of lymph nodes retrieved in the TAS group was 1.52 (IQR, 1 to 2) (p£0.001). while that in post-neo-adjuvant therapy cases was 1.95 (IQR, 1 to 3) (p£0.001). Backup ALND results showed metastatic deposits in 18% (9 out of 50) more than TAS. Of the 9 cases, 2 were upfront cases, and 7 were post-neo-adjuvant therapy cases (p=0.025).
Conclusion: Charcoal tattooing of ultrasound suspicious ALN is viable for clinical use, especially in resource-constrained healthcare resources. TAS selectively removed positive LNs with less radicality than ALND; however, ALND removed additional positive nodes in 18% of patients.
INTRODUCTION
Management of the axilla in breast cancer (BC) patients has shifted toward less invasive measures to reduce morbidity without compromising prognosis. Historically, patients who experienced metastatic axillary lymph nodes (LNs) had axillary lymph node dissection (ALND). The American College of Surgeons Oncology Group (ACOSOG) Z0011 trial indicated that ALND provides no survival benefit compared to sentinel lymph node dissection (SLND) (1).
The findings from this trial led to important modifications in the radiologic and surgical techniques used for assessing and managing the axilla in a specific group of BC patients (2,3).
Nevertheless, lymphatic system surgery persisted at the dogmatic level of ALND based on local control, staging information, and survival maximization until the 1990s, when the SLN technique was implemented for clinically node-negative BC. The dogma was progressively weakened, leaving only a limited number of standard indications for ALND in clinical practice (4).
A new technique called tailored axillary surgery (TAS) was developed for patients with clinically node-positive breast cancer, both during upfront surgery and after neo-adjuvant chemotherapy (NACT). TAS involves selectively removing obvious nodal disease by palpation to convert a clinically positive axilla to a negative. It also incorporates the sentinel lymph node (SLN) procedure to minimize microscopic disease. The goal of TAS is to reduce tumor load in the axilla, enabling effective control through adjuvant regional nodal irradiation (RNI) (5).
Marking of suspicious LNs is widely known for multiple types of tumors especially in BC. The application of carbon suspension tattooing has been extensively utilized for identifying lesions or tumors encompassing metastatic axillary LNs prior to neo-adjuvant chemotherapy (NACT). The use of carbon solution in this context is off-label since it is only approved for use in the gastrointestinal tract and primitive breast lesions. The modality involves injecting a charcoal suspension into the cortical of the suspicious LN under ultrasound supervision during the LN biopsy. Immediately post-injection, the charcoal particles are discernible with a hyperechoic halo around the LN (6).
Hence, we evaluated the feasibility and validity of ultrasound-guided marking of metastatic axillary LNs followed by tailored axillary surgery (TAS), in BC patients with clinically node-positive status as a method for tumor burden reduction.
PATIENTS AND METHODS
This study is a prospective observational cohort study that involved fifty female patients with clinical node-positive (cN1) BC who underwent upfront or post-neo-adjuvant breast surgery and axillary LNs dissection. The study was conducted at the Breast Surgery Unit of the General and Laparoscopic Surgery Department and Medical Oncology Department of a tertiary university-based hospital from June 2022 to March 2024.
The study included female patients aged ? 18 years with clinically node-positive BC (N1), identified via palpation or imaging at the first diagnosis. Patients were enrolled in the upfront surgical context and in instances of residual nodal disease following NACT. Patients classified as American Joint Committee on Cancer/Union for International Cancer Control (AJCC/UICC) stage II-III, including all molecular subtypes, were permitted (7).
Individuals with stage IV BC, clinical N3 BC, clinical N2 BC confined to the internal mammary nodes,
contralateral BC, prior axillary surgery (excluding prior sentinel node procedures for in-breast recurrence), prior regional radiotherapy (RT), and a history of hematological or primary solid tumor malignancy were excluded, unless they had been in remission for a minimum of 5 years prior to registration.
Patients were subjected to full history taking. Those who suffer from suspicious masses or are at high risk of breast malignancy proceed to further imaging procedures (Mammography, ultrasonography, and magnetic resonance imaging (MRI) if indicated).
Suppose a suspicious mass is found (with BIRADS 4-5), an ultrasound-guided biopsy is done for histopathological examination. Patients with a histopathological diagnosis of BC undergo further metastatic work-up according to the stage of presentation and immuno-histochemical study of the tumor in the form of estrogen receptor (ER), progesterone receptor (PR), human epidermal receptor (Her2neu), and proliferation index (PI) Ki67.
Figure 1 - Black eye identified in 3 lymph nodes and adjacent soft tissue (black arrow), rest of axillary lymph nodes "Level 1&2" (red arrow)
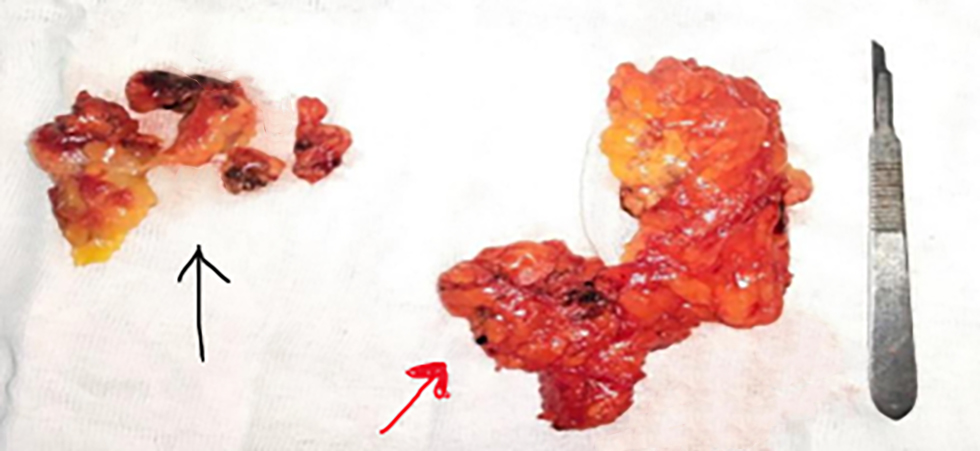
The cases were discussed among a multidisciplinary team (MDT) with members from the breast oncology, surgery, radiology, and pathology departments to determine optimal management.
All patients had their axillary LNs evaluated by clinical examination and by ultrasound performed by an experienced radiologist. Positive LNs were considered if they were firm/hard and/or matted on palpation and fulfilled abnormal node criteria on ultrasound. The sonographic characteristics of positive LNs were generalized or focal cortical thickening, globular in shape, and effacement of node fatty hilum.
Sixty patients with cN1 BC had targeted axillary ultrasonography to evaluate the number of patho-logical LNs followed by carbon particles (Black-Eye) injection in the suspicious node aiming at TAS.
Tattooing Procedure
Palpation of the axilla and target axillary high-resolution ultrasound were performed, followed immediately by injection of 0.1 to 0.3 ml of Black Eye ink into the cortex of the suspicious LN and the adjacent soft tissue.
Surgery
Then, backup axillary dissection, including levels I and II, is done, followed by a comparison of the final pathology reports.
Patients were scheduled for either breast-conserving surgery (BCS) or mastectomy. We performed targeted axillary dissection which involved identification and extraction of the inked lymph node and any palpable nodes. Then, axillary dissection, including levels I and II, was done. All dissected LNs were subjected to post-operative histopathological examination to compare the results of TAS and backup ALND with the definitive histopathological diagnosis of malignan (fig. 1).
Statistical Analysis
Data were encoded and input with the Statistical Package for Social Sciences (SPSS) version 28 (IBM Corp., Armonk, NY, USA). Data was summarized utilizing mean, standard deviation (SD), median, minimum, maximum, and interquartile range (IQR) for quantitative data, alongside frequency (count) and relative frequency (%) for categorical data. Quantitative factors were compared utilizing the non-parametric Kruskal-Wallis and Mann-Whitney tests (8). A Chi-square test was used to compare categorical data. The Exact test was employed when the anticipated frequency fell below 5 (9). The Spearman correlation coefficient was used to assess the correlation among quantitative variables (10).
P-values below 0.05 were deemed statistically significant.
RESULTS
This study successfully comprised sixty individuals with node-positive N1 BC. Ten patients were excluded from the analysis, 4 patients had more than 3 suspicious pathological lymph nodes, 3 patients had indeterminate lymph nodes, and FNA results from the suspicious lymph nodes turned out to be free of malignancy, and those patients underwent SLNB, and finally 3 patients were excluded due to failure of ink identification during surgery. We ended up with 50 evaluable patients for analysis. All patients had backup ALND.
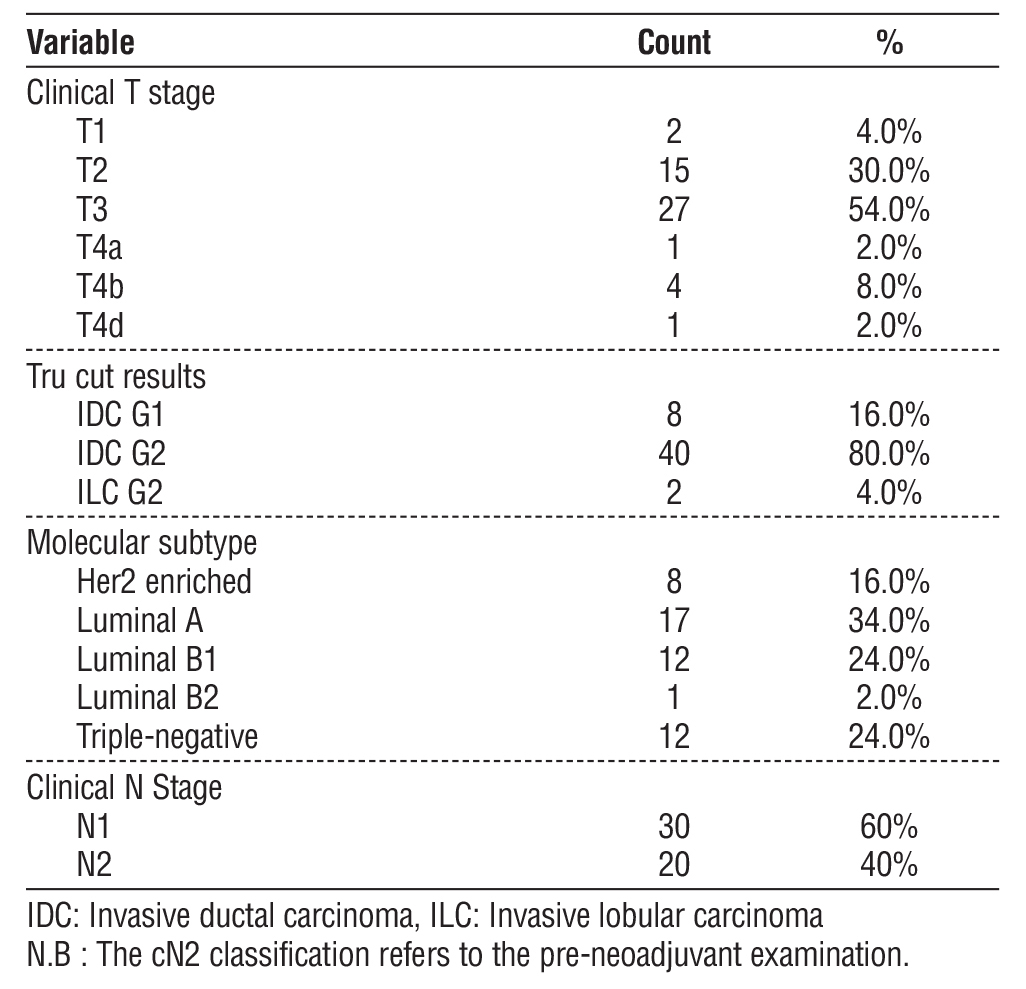
The mean age of the patients at diagnosis was 54.48±9.06 years. The mean size of the primary tumor at presentation was 5.77±1.88 cm (range, 1.60 to 9 cm). Twenty-nine patients underwent upfront surgery (58%), and twenty-one underwent post-neo-adjuvant therapy surgery (42%). Twenty-seven patients underwent oncoplastic breast surgery (54%), and twenty-three patients underwent modified radical mastectomy (46%) (table 1).
Table 1 - Tumor details at initial presentation among study participants.
None of the patients had complications related to LN Black Eye ink injection. The mean delay between ink injection and surgery in days is 3.5 days (1 to 7 days) this was due to scheduling conflicts related to the
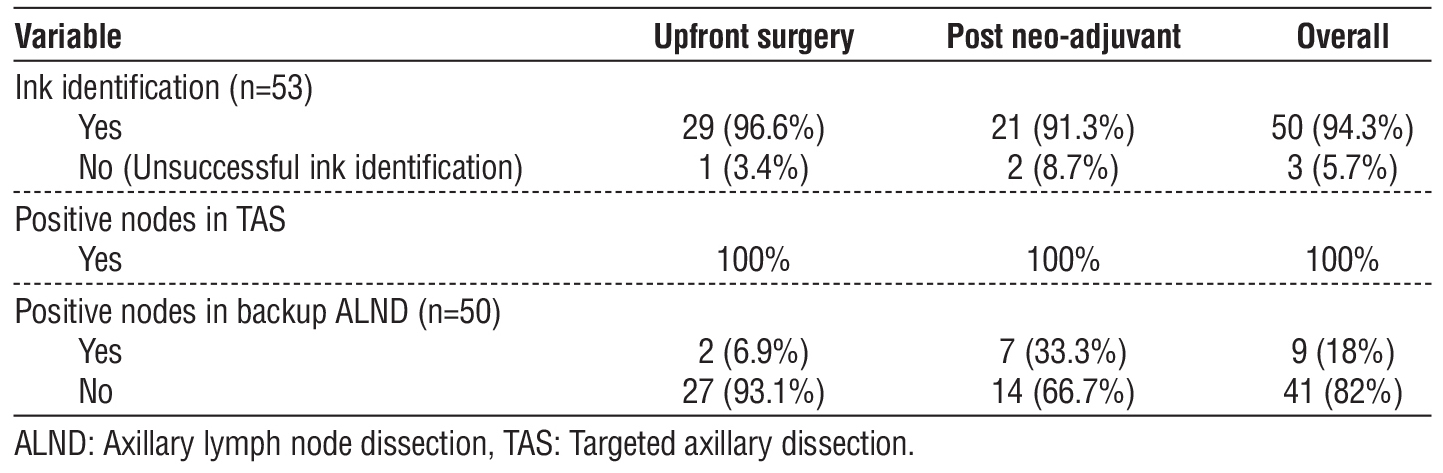
availability of the interventional radiologist and the patient’s position in the operating list. The mean number of suspicious LNs detected by ultrasound in upfront and post-neo-adjuvant cases was 2 LNs (1.76 and 2.10 respectively). The mean suspicious LN cortical thickness in upfront cases was 7.34±1.01 mm, while in post-neo-adjuvant therapy cases, it was 7.48±0.7 mm (p=0.633). It is worth mentioning that the ink injection technique was successful in 50 out of 53 initially recruited cases (94.3%).
The mean number of LNs retrieved in the backup axillary dissection was 16 (range, 7 to 31). Backup ALND results showed metastatic deposits in 18% (9 out of 50) which were not detected during TAS. Of the 9 cases, 2 were upfront cases (6.9% of the 29 upfront cases), and 7 were post-neo-adjuvant therapy cases (33.3% of the 21 post-neo-adjuvant cases), showing a statistically significant difference between the two groups (p=0.025).
Ink-tattooed nodes were excised and labeled by surgeons at a median of 2 nodes (IQR 1-3), which corresponded to a median of 4 nodes (IQR 1-7) identified by the pathologists, two of which were positive (IQR 1–3). The median number of positive LNs in backup ALND was 3 LNs (IQR 1–12). The number of LNs injected with ink compared to those found positive in TAS and backup ALND is demonstrated in table 2. TAS revealed a 100% positive LNs.
Table 2 - Comparison between ink-injected lymph nodes and positive lymph nodes during TAS and backup ALND among study participants.
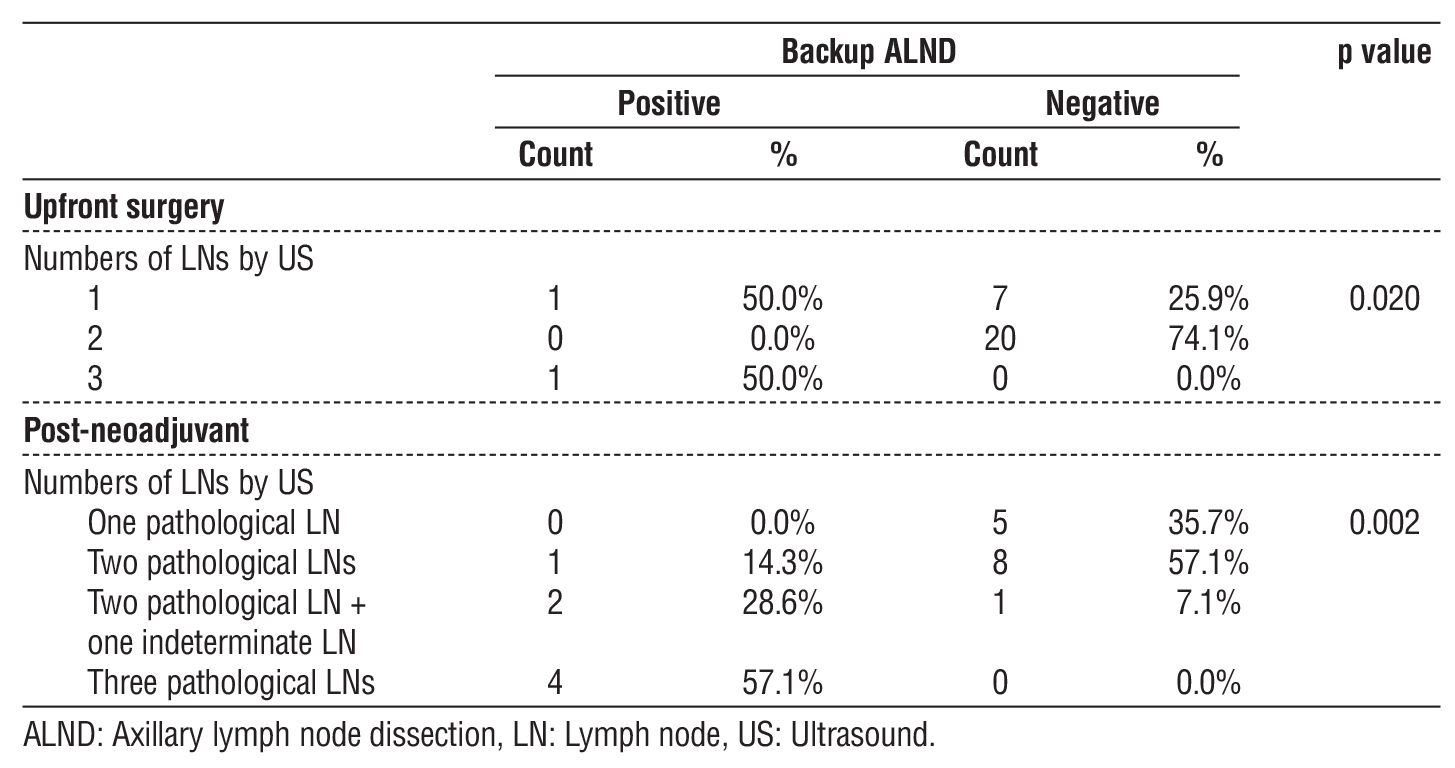
In upfront cases, one (50%) patient with one suspicious LN and one (50%) patient with three suspicious LNs had positive backup ALND with a significant P-value of 0.020. In post-NACT, one (14.3%) patient with two pathological LNs, two (28.6%) patients with two pathological and one indeterminate LN, and Four (57.1%) patients with three pathological LNs had positive ALND, with a significant difference between the number of LNs and positive backup ALND nodes (p=0.002) (table 3).
Table 3 - Correlation between numbers of LNs by US and ALND among study participants.
Radiological Features of Suspicious LNs
Thirty-four cases (68%) had diffuse cortical thickening, thirteen cases (26%) had focal cortical thickening, and three cases (6%) had combined diffuse and focal cortical thickening. The mean cortical thickness measurement in cases of positive backup ALND was 7.11 mm (IQR, 6 to 8), while in cases of negative ALND was 7.46 mm (IQR, 6 to 9) [p=0.306]. Four (44.4%) patients with diffuse cortical thickening LNs, three (33.3%) patients with focal cortical thickening LNs, and two (22.2%) with both diffuse and focal cortical thickening LNs had positive backup ALND. These findings show a significant difference (p=0.048) (table 4).
Table 4 - Correlation between radiological features of affected LNs and backup ALND among study participants.
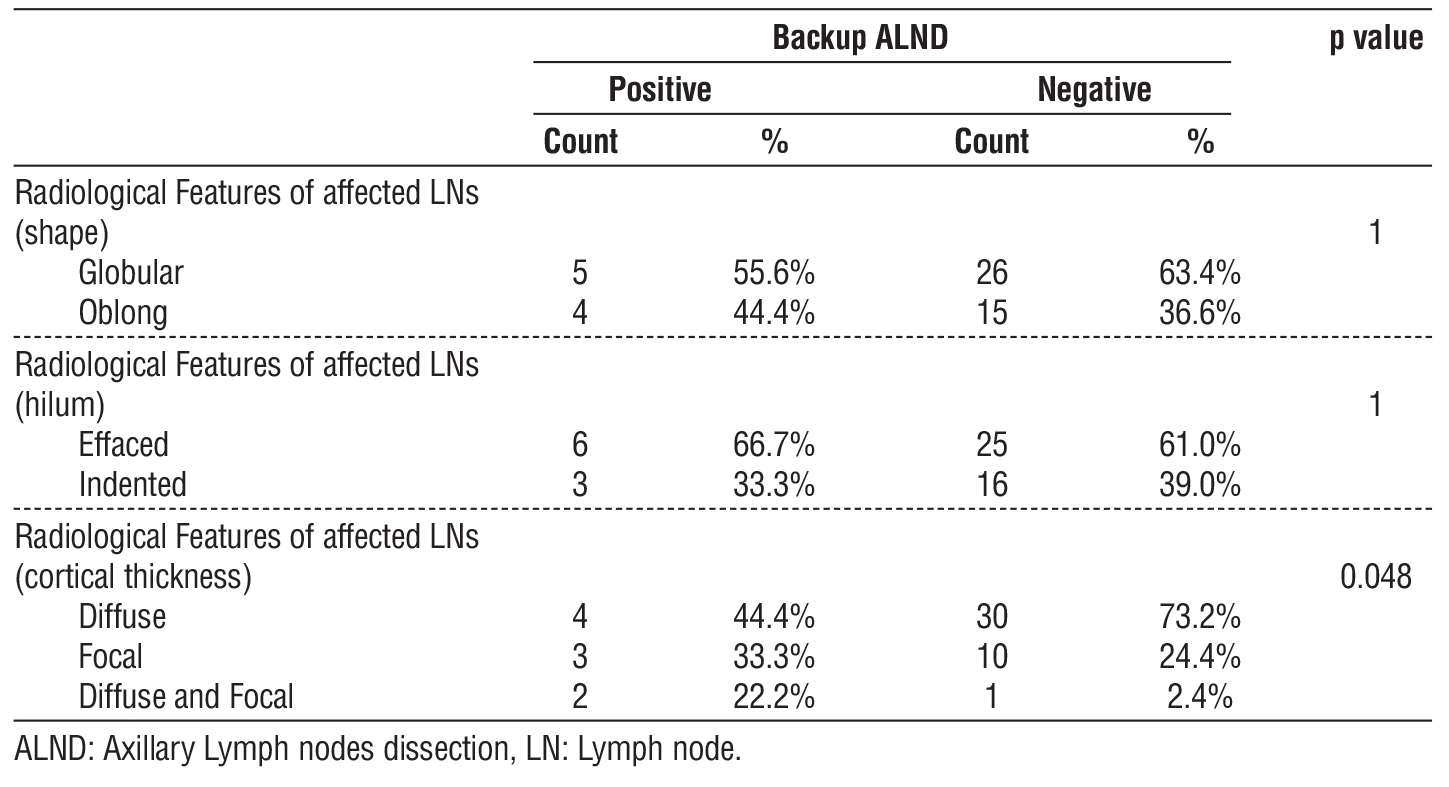
Regarding the LNs shape, thirty-two cases (64%) had globular-shaped LNs, and eighteen cases (36%) had oblong-shaped LNs. Among the patients who had positive backup ALND, 5 (55.6%) of them had globular-shaped LNs, while the remaining 4 (44.4%) had oblong-shaped LNs (table 4).
Thirty-one cases (62%) had suspicious LNs with effaced hilum, while the remaining nineteen (38%) had LNs with indented hilum. Among the patients who had positive backup ALND, six (66.7%) had effaced hilum, while the remaining three (33.3%) had indented hilum (table 4).
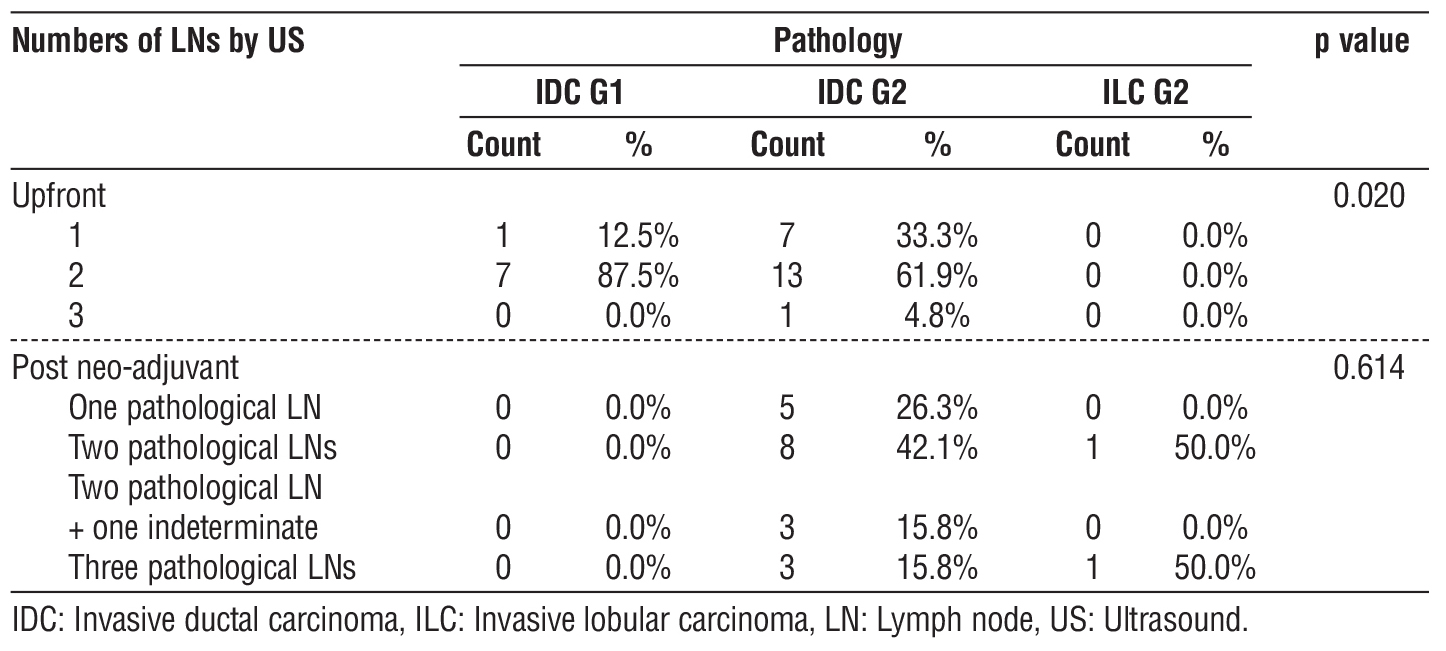
Correlation Between Tru-Cut Pathology and the Number of LNs Detected by US
In upfront cases, most patients with IDC G1(87.5%) had two suspicious LNs, while those with intra-ductal carcinoma (IDC) G2 (61.9%) had two suspicious LNs. This distribution variation shows a statistically significant difference (p=0.020) (table 5).
In post-neo-adjuvant cases, most patients with IDC G2 (42.1%) had two pathological LNs, while those with intra-lobular carcinoma (ILC) G2 had an equal distribution between two and three pathological LNs (table 5).
Table 5 - Correlation between tru-cut pathology and numbers of LNs detected by US among study participants.
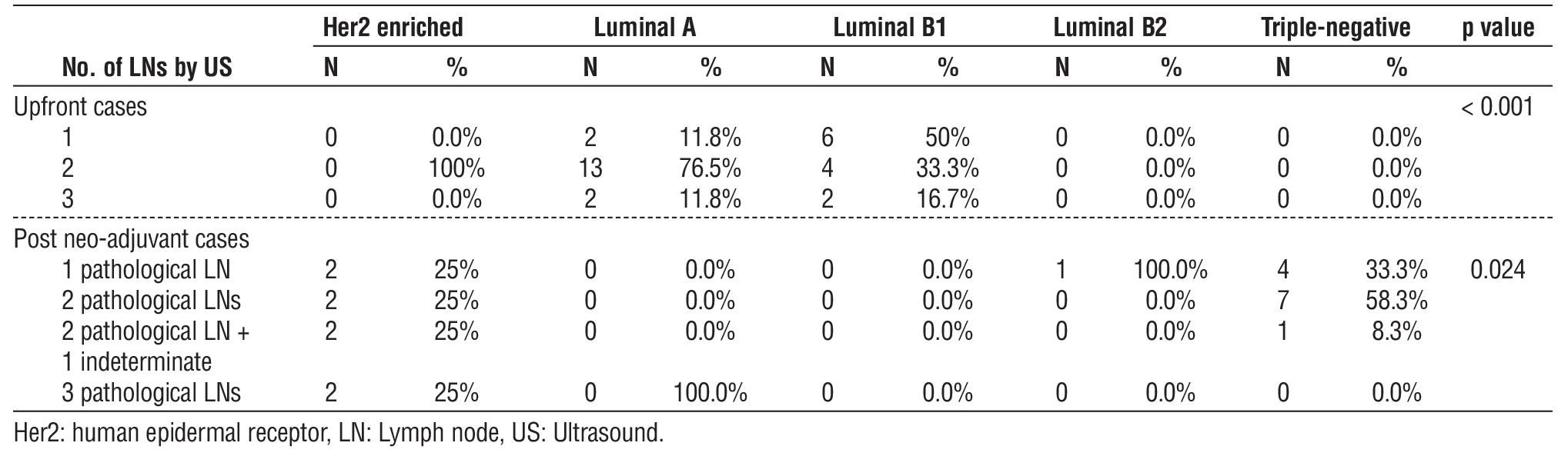
Correlation Between Molecular Biology and the Number of LNs detected by US
In upfront cases and post-neo-adjuvant cases, the difference between the distribution of cases along the molecular biology and the number of LNs defined as pathological or intermediate by US was significant (p-value < 0.05) (table 6).
Table 6 - Correlation between molecular biology and numbers of LNs detected by US among study participants.
DISCUSSION
TAS is not a new surgical procedure but rather an innovative approach that integrates various techniques to reduce tumor load in patients who typically receive Axillary Lymph Node Dissection (ALND) as the standard treatment. TAS aims to convert a clinically positive axilla into a clinically negative one by selectively removing visible nodal disease through palpation, effectively customizing the level of axillary surgery to match the degree of axillary disease (5).
Tattooing with carbon suspension has been extensively utilized for the identification of tumors, including metastatic axillary LNs. It is assumed that suspicious pathological LNs, which have been marked with BlackEye carbon particles and guided by ultrasound, serve as an effective method for decreasing tumor burden in patients experiencing clinically node-positive (cN1) BC (11).
Herein, 50 patients had cN1 disease based on clinical and radiological evaluation. Our main objective was to assess the accuracy of ultrasound in assessing axillary LN status in upfront and post-neo-adjuvant cases.
We successfully identified the BlackEye ink in 94.3% (50 of 53) of the cases; the mean delay between ink injection and surgery in days was 3.5 days (range, 1 to 7 days). We found that axillary LNs tattooing is a simple technique, easily identified during surgery, and a less expensive method than the use of a radioactive seed, which is more sophisticated and faces legal issues. Moreover, tattoo ink persists at the site for an extended duration, and additional radiological imaging or nuclear medicine interventions for localization are unnecessary. The tattooing of LNs offers several benefits over other methods for labeling and excising first suspicious LNs, encompassing the application of clips, radioactive or magnetic seeds, or radiofrequency identification devices. It is offered at a little cost, eliminating the need for preoperative localization operations, hence reducing patient pain and avoiding extra radiation exposure (11).
There is a lack of information concerning the carbon tattooing technique, despite the recent publication of results from the prospective pre-ATNEC trial that indicated a detection rate of 64% for the tattooed LNs in 22 patients (12). Furthermore, various studies have shown a detection rate of 100%, with no false-negative results reported (13,14).
Additional comprehensive retrospective studies have validated the heightened detection rate for carbon-labeled LNs. Kim et al. documented a detection rate of 98% in 45 patients, even though the carbon injection was administered after main systemic treatment in chronically suspicious LNs patients (15). Natsiopoulos et al. documented a detection rate of 100% across 75 instances. The tattooing was performed before the primary systemic treatment, and all suspicious LNs were labeled (1-5) and later excised (16).
When planning this study, we were interested to see how the pragmatic concept of TAS would be translated into clinical practice. Therefore, we documented key aspects of TAS including details on preoperative workup (e.g., type of ink and imaging) and the intervention (e.g., number of lymph nodes removed and localization technique).
In our study, the tattooed LN removal rate was 100%, which was confirmed and evaluated by an experienced pathologist to ascertain the presence of tattoo pigment within the cortical surface of the node in the TAS cohort. Surgeons estimated the excision and labeling of a median of 2 (IQR 1-3) ink-tattooed nodes, corresponding to a median of 4 (IQR 1-7) nodes diagnosed by pathologists, of which two were positive (IQR 1-3).
Since the present study focused on use of IGL and its impact on TAS, data collection on the 10 excluded patients was limited, and hence, no false-negative rate of TAS could be evaluated.
Our study's detection rate compares favorably to clip marking, which has been documented at rates below 80% (17-19). Caudle et al. reported a false-negative rate of 2.0% and a congruence rate of 77% (20), while Patel et al. reported 100% congruence, likely due to a smaller number of node-positive patients after systemic therapy (14). Both Caudle and Patel included all palpable lymph nodes as SLNs, averaging 2.7 and 3.1 SLNs removed, whereas excluding suspicious nodes reduced the average to 1.6 (18).
In our study, we found that backup ALND results showed metastatic deposits in 18% (9 out of 50). Of 9 cases that had metastatic deposits, two were upfront cases (6.9%), and seven were post-neo-adjuvant therapy cases (33.3%) (P=0.025). The mean number of suspicious LNs detected by ultrasound in upfront cases was 1.76 (range, 1 to 3), while in post-neo-adjuvant cases, the mean number of LNs detected by ultrasound was 2.10 (range, 1 to 3).
Consequently, all patients excised an average of 15.84 extra LN during backup ALND, with 18% of these being positive, in contrast to another research with 100 patients who had completion ALND, which resulted in a mean removal of 14 additional LNs, with 70% being positive (5).
The histopathological assessment of the TLN following primary systemic treatment is crucial for two
reasons. A non-invasive approach that reliably predicts post-neoadjuvant pathologic nodes (ypN) status is presently unavailable. Axillary ultrasound imaging, a typical diagnostic method, does not consistently rule out residual LNs metastases after main systemic treatment, with a sensitivity of 37 to 100% (11), nor does ultrasound-guided needle biopsy (sensitivity 79.6%) (21). Secondly, further treatment decisions are contingent upon LN status after first systemic therapy (20). When the TAS is positive, additional lymph node metastases can be found in up to 51% of patients (20); hence, completion ALND is strongly advised.
In our study, there was a significant correlation between positive backup ALND and cT4 stage, whereas 4 out of 4 patients (100%) with cT4b stage, 1 out of 1 patient (100%) with cT4a and 1 out of 1 patient (100%) with cT4d BC included in this study had positive backup ALND ( In our study, there was one such case, and after neoadjuvant therapy, the axillary lymph node staging became N1. Therefore, we decided to include this patient, especially since, in our protocol, we perform backup axillary evacuation), while two out 15 (13.3%) with cT2, 1 out of 26 (3.7%) with cT3 and 0 out of 2 (0.0%) with cT1 BC included in this study had positive backup ALND.
Regarding molecular biology, there was a significant correlation between positive backup ALND and Luminal B1, whereas 6 out of 12 (50%) cases with Luminal B1, one out of 8 (12.5%) cases with Her2-enriched, 2 out of 17 (11.8%) cases with Luminal A, and 0 out of (1 & 12) patients with Luminal B and Triple negative BC receptively had positive backup ALND.
In comparison to another study, researchers identified a pN1 rate of 44.6% in a cohort of 91.5% ER-positive patients (22). Furthermore, a further cohort of HR+/Her2-negative patients with palpable nodal disease undergoing backup ALND demonstrated that 43% of individuals had two or fewer affected LN (23).
In our study, we found that the mean number of positive LNs in ALND was 3.14 (IQR,1 to 12). One case had 12 positive LNs (post-neo-adjuvant, initially was cT4dN2, Luminal B1), two cases had two positive LNs, and six cases had one positive LN in the backup ALND.
The relevance of finding positive nodes in patients after TAS undergoing ALND, currently remains unclear. Hence, we recommend waiting for oncologic outcome analysis of TAXIS before adopting the much less radical approach of TAS in clinical practice.
Limitations
One limitation of the present study is the small sample size. Additionally, we were not able to assess differences in the number of nodes detected by using single versus dual localizing techniques.
CONCLUSION
Charcoal tattooing of ultrasound-suspicious ALN is technically viable for clinical use, especially in resource-constrained healthcare settings. Consequently, it should be effective in these environments.
TAS may replace ALND for clinically node-positive breast cancer, as it selectively removes positive lymph nodes with less invasiveness. However, its outcomes need further analysis before adoption. ALND is advised for cT4 cases, while TAS is recommended for upfront N1 disease and selected post-neoadjuvant patients.
Recommendations
Regarding the strong correlation between positive backup ALND and cT4 stage, classic ALND is highly
recommended in these cases. In general we recommend application of TAS technique in upfront cases with N1 disease and selected post neo-adjuvant cases.
Author’s Contributions
Ismail Shafeek generated the idea and revised the manuscript. Omar sherif generated the idea, interpreted the data, and revised the manuscript. Sherihan Fakhry wrote and revised the manuscript. Bishoy Mikhail collected and analyzed the data and wrote the manuscript. All authors read and approved the final manuscript.
Conflicts of Interest
No conflict of interest.
Source of Funding: None.
Ethical Consideration
Our protocol received approval from the Institutional Ethical Research Committee (Code: MD-256-2022). The techniques and objectives of the study were explicitly communicated to the participants. Prior to participation in the study, written informed consent was secured from the participants, accompanied by an elucidation of the procedure's advantages and disadvantages. The participant had the liberty to withdraw from the study at any time; participation was wholly voluntary. In compliance with the Declaration of Helsinki, all phases of data collection, entry, and analysis were executed with utmost confidentiality and privacy.
REFERENCES
1. Giuliano AE, Hunt KK, Ballman KV, Beitsch PD, Whitworth PW, Blumencranz PW, et al. Axillary dissection vs no axillary dissection in women with invasive breast cancer and sentinel node metastasis: a randomized clinical trial. JAMA. 2011;305(6):569-75.
2. Humphrey KL, Saksena MA, Freer PE, Smith BL, Rafferty EA. To do or not to do: axillary nodal evaluation after ACOSOG Z0011 trial. Radiographics. 2014;34(7):1807-16.
3. Caudle AS, Cupp JA, Kuerer HM. Management of axillary disease. Surgical Oncology Clinics. 2014;23(3):473-86.
4. Maggi N, Nussbaumer R, Holzer L, Weber WP. Axillary surgery in node-positive breast cancer. The Breast. 2022;62:S50-3.
5. Weber WP, Matrai Z, Hayoz S, Tausch C, Henke G, Zwahlen DR, et al. Tailored axillary surgery in patients with clinically node-positive breast cancer: pre-planned feasibility substudy of TAXIS (OPBC-03, SAKK 23/16, IBCSG 57-18, ABCSG-53, GBG 101). Breast. 2021; 60:98-110.
6. Hartmann S, Kühn T, De Boniface J, Stachs A, Winckelmann A, Frisell J, et al. Carbon tattooing for targeted lymph node biopsy after primary systemic therapy in breast cancer: prospective multicentre TATTOO trial. Br J Surg. 2021;108(3):302-307.
7. Brierley JD, Gospodarowicz MK, Wittekind C, editors. TNM classification of malignant tumours. John Wiley & Sons; 2017.
8. Chan YH. Biostatistics 102: quantitative data-parametric & non-parametric tests. blood Press. 2003;140(24.08):79.
9. Chan YH. Biostatistics 103: qualitative data-tests of independence. Singapore Med J. 2003;44(10):498-503.
10. Chan YH. Biostatistics 104: correlational analysis. Singapore Med J. 2003;44(12):614-9.
11. Banys-Paluchowski M, Gruber IV, Hartkopf A, Paluchowski P, Krawczyk N, Marx M, et al. Axillary ultrasound for prediction of response to neoadjuvant therapy in the context of surgical strategies to axillary dissection in primary breast cancer: a systematic review of the current literature. Arch Gynecol Obstet. 2020;301(2):341-353.
12. Goyal A, Puri S, Marshall A, Valassiadou K, Hoosein MM, Carmichael AR, et al. A multicentre prospective feasibility study of carbon dye tattooing of biopsied axillary node and surgical localisation in breast cancer patients. Breast Cancer Res Treat. 2021;185(2):433-440.
13. Park S, Koo JS, Kim GM, Sohn J, Kim SI, Cho YU, et al. Feasibility of charcoal tattooing of cytology-proven metastatic axillary lymph node at diagnosis and sentinel lymph node biopsy after neoadjuvant chemotherapy in breast cancer patients. Cancer Res Treat. 2018; 50(3):801-812.
14. Patel R, MacKerricher W, Tsai J, Choy N, Lipson J, Ikeda D, et al. Pretreatment tattoo marking of suspicious axillary lymph nodes: reliability and correlation with sentinel lymph node. Ann Surg Oncol. 2019;26(8):2452-2458.
15. Kim WH, Kim HJ, Jung JH, Park HY, Lee J, Kim WW, et al. Ultrasound-guided restaging and localization of axillary lymph nodes after neoadjuvant chemotherapy for guidance of axillary surgery in breast cancer patients: experience with activated charcoal. Ann Surg Oncol. 2018;25(2):494-500.
16. Natsiopoulos I, Intzes S, Liappis T, Zarampoukas K, Zarampoukas T, Zacharopoulou V, et al. Axillary lymph node tattooing and targeted axillary dissection in breast cancer patients who presented as cN+ before neoadjuvant chemotherapy and became cN0 after treatment. Clin Breast Cancer. 2019;19(3):208-215.
17. Nguyen TT, Hieken TJ, Glazebrook KN, Boughey JC. Localizing the clipped node in patients with node-positive breast cancer treated with neoadjuvant chemotherapy: early learning experience and challenges. Ann Surg Oncol. 2017;24(10):3011-3016.
18. Hartmann S, Reimer T, Gerber B, Stubert J, Stengel B, Stachs A. Wire localization of clip-marked axillary lymph nodes in breast cancer patients treated with primary systemic therapy. Eur J Surg Oncol. 2018;44(9):1307-1311.
19. Kuemmel S, Heil J, Rueland A, Seiberling C, Harrach H, Schindowski D, et al. A prospective, multicenter registry study to evaluate the clinical feasibility of targeted axillary dissection (TAD) in node-positive breast cancer patients. Ann Surg. 2022;276(5):e553-e562.
20. Caudle AS, Yang WT, Krishnamurthy S, Mittendorf EA, Black DM, Gilcrease MZ, et al. Improved axillary evaluation following neo-adjuvant therapy for patients with node-positive breast cancer using selective evaluation of clipped nodes: implementation of targeted axillary dissection. J Clin Oncol. 2016;34(10):1072-8.
21. Houssami N, Ciatto S, Turner RM, Cody III HS, Macaskill P. Preoperative ultrasound-guided needle biopsy of axillary nodes in invasive breast cancer: meta-analysis of its accuracy and utility in staging the axilla. Ann Surg. 2011;254(2):243-51.
22. Angarita S, Ye L, Rünger D, Hadaya J, Baker JL, Dawson N, et al. Assessing the burden of nodal disease for breast cancer patients with clinically positive nodes: hope for more limited axillary surgery. Ann Surg Oncol. 2021;28(5):2609-2618.
23. Crown A, Sevilimedu V, Morrow M. Palpable adenopathy does not indicate high-volume axillary nodal disease in hormone receptor-positive breast cancer. Ann Surg Oncol. 2021;28(11):6060-6068.
Full Text Sources:
Abstract:
Views: 901

For Authors
Journal Subscriptions

Dec 2025
Supplements
Instructions for authors
Online submission
Contact
e-ISSN: 2601 - 1700 (online)
ISSN-L: 2559 - 723X
Journal Abbreviation: Surg. Gastroenterol. Oncol.
Surgery, Gastroenterology and Oncology (SGO) is indexed in:
- SCOPUS
- EBSCO
- DOI/Crossref
- Google Scholar
- SCImago
- Harvard Library
- Open Academic Journals Index (OAJI)
Surgery, Gastroenterology and Oncology (SGO) is an open-access, peer-reviewed online journal published by Celsius Publishing House. The journal allows readers to read, download, copy, distribute, print, search, or link to the full text of its articles.
Time to first editorial decision: 25 days
Rejection rate: 61%
CiteScore: 0.2

Meetings and Courses in 2025
Meetings and Courses in 2024
Meetings and Courses in 2023
Meetings and Courses in 2022
Meetings and Courses in 2021
Meetings and Courses in 2020
Meetings and Courses in 2019
Verona expert meeting 2019

Surgery, Gastroenterology and Oncology applies the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits readers to copy and redistribute the material in any medium or format, remix, adapt, build upon the published works non-commercially, and license the derivative works on different terms, provided the original material is properly cited and the use is non-commercial. Please see: https://creativecommons.org/licenses/by-nc/4.0/
Publisher’s Note:
The opinions, statements, and data contained in article are solely those of the authors and not of Surgery, Gastroenterology and Oncology journal or the editors. Publisher and the editors disclaim responsibility for any damage resulting from any ideas, instructions, methods, or products referred to in the content.
 IASGO Society News
IASGO Society News