Surgery, Gastroenterology and Oncology

|
|
Background: Treatment of metastatic colorectal cancer (mCRC) with BRAFV600E mutation is challenging. Resistance to treatment is a significant cause of unfavourable clinical outcomes. Aim: This work investigated the effectiveness and safety of bevacizumab (BEV) mFOLFOXIRI as an initial line of treatment of mCRC with BRAF V600E mutation.
Materials & Methods: This prospective, phase II, open-label study included 30 participants with mCRC harboring RAS wild-type and BRAF V600E mutation in primary tumors or metastases. Patients received 5 mg/kg BEV plus mFOLFOXIRI biweekly for 12 cycles, until progression of the disease, intolerable toxicity, or resectability. The main end point was to evaluate response, additional end points were progression free survival, overall survival and toxicity.
Results: In the current study, 18 patients achieved objective response (6.67% complete responses, 53.33% partial responses). The disease was stable in 8 (26.67%) patients. Progression-Free Survival (PFS) was 76.67% at 6 months, 43.33% at 12 months, and 10% at 36 months. Overall Survival (OS) was 83.33% at 6 months, 73.33% at 12 months, and 26.67% at 36 months. The treatment protocol showed a high incidence of Diarrhea (73.33%). Hematological toxicities were dominated by neutropenia (40%). G-CSF prophylaxis was given to 76.66%, and the dose was reduced in 36.6% of patients.
Conclusions: BEV-mFOLFOXIRI combination demonstrates efficacy in treating BRAF mutant V600E mCRC, with a high response and disease control rate. However, the treatment is associated with considerable toxicities, requiring careful monitoring and management.
Introduction
Colorectal cancer (CRC) is a prevalent malignant tumor, ranked as the 3rd malignancy in men and the 2nd in women, based on data from the WHO GLOBOCAN database (1). Patients with metastatic colorectal cancer (mCRC) receiving multidisciplinary therapy have median survival period exceeding 30 months (2,3). Notably, the selection of initial treatment options remains a pivotal step in the treatment pathway (4).
The standard treatment approach has been a combination of two chemotherapy agents, typically fluoropyrimidine and either irinotecan or oxaliplatin, accompanied by targeted therapies (5).
BRAF is a considerable gene of the RAS-RAF-MAPK pathway. Mutations in the oncogenic BRAF gene cause the signaling system to be constitutively active, which in turn promotes tumor growth and inhibits cell death (6). Most BRAF mutations (about 90%) are the V600E variant, which causes the BRAF kinase activity to be hyperactivated in a way that is not dependent on Ras (7). Mutation of BRAF V600E is documented in up to 10% of all mCRC cases (8).
BRAFV600E mutation is steadily linked with poor outcome in mCRC. Patients diagnosed with mutated-BRAF V600E mCRC have an overall survival (OS) duration ranging from 9.8 to 18.2 months (9). Worsening resistance to treatment is a significant contributor to these unfavorable clinical outcomes.
The BRAF V600E mutation acts as a mechanism of resistance to EGFR inhibitors and randomized trials conducted in BRAF V600E mutant/RAS wild type mCRC patients suggested limited benefits (10).
The TRIBE trial stated that patients with BRAF V600E mutant mCRC achieved higher rates of objective response and considerable median OS (29.8 vs. 25.8 months) with FOLFOXIRI plus bevacizumab as first line of tretment (11).
Despite the established efficacy of FOLFOXIRI plus BEV, ongoing research continues to refine its benefits (12).
Material and Methods
This prospective phase II study included 30 patients, performed between February 2021 to February 2024 after the ethical approval of the Tanta University Hospital ethical committee in Egypt (approval code 36264PR418). Informed written consent was obtained from the patients.
Inclusion and Exclusion Criteria
All the participants in this trial aged above 18 years and had metastatic colorectal cancer with wild-type RAS gene (exons 2, 3, 4 of KRAS and NRAS) and BRAF V600E mutation in their primary or metastatic sites. Patients with Eastern Cooperative Oncology Group (ECOG) performance status of 0-1 and not received prior chemotherapy in a metastatic setting were included.
The study excluded cardiovascular disease that occurred within 12 months before enrollment, untreated CNS lesions, or a history of nonhealing wounds or bleeding.
Pretreatment Evaluation
All cases underwent a thorough history, physical examination, laboratory investigations including tumor markers CEA, CA19.9, and radiological tests including PET CT if available.
Genetic Testing for RAS and BRAF
The genetic panel was assessed using a real-time qualitative PCR assay on DNA extracted from Formalin-Fixed-Paraffin-embedded tissue.
Treatment Received
Patients received bevacizumab 5 mg/kg and mFOLFOXIRI [irinotecan (150 mg/m2), oxaliplatin (85 mg/m2), folinic acid (200 mg/m2), and 5-Fluorouracil (2400 mg/m2).
Treatment schedule
Treatment regimen was received every 2 weeks until progression of the disease, development of Unacceptable toxicities, determination of resectability, or a maximum of 12 cycles, whichever came first.
Maintenance treatment
Following 12 cycles, maintenance treatment with bevacizumab (5 mg/kg once every 2 weeks) along with capecitabine.
Follow-up period
Follow-up interval ranged from 6-18 months.
Dose reduction
A dose reduction was implemented for grade 3-4 acute toxicities.
End Points
The primary end point was response assessment by RECIST criteria 1.1 using a CT scan or MRI every 8-12 week.
Table 1 - Demographic data of the studied patients

Additional end points included progression-free survival (PFS) defined as the time from the date of diagnosis to disease progression or death, and overall survival (OS) defined as the time from the date of diagnosis to death due to any cause.
Toxicity
The grade of toxicity and adverse events were determined according to NCI-CTC criteria, version 5 (13). G-CSF was administered as a secondary Prophylaxis for patients who developed grade ?3 neutropenia and as a primary prophylaxis in cases of old age and low-performance status.
Statistical Analysis
Our data were analyzed using SPSS version 26 (IBM Inc., Chicago, Illinois, United States). The mean and standard deviation were used to represent quantitative data, while frequency and percentage were used to represent qualitative characteristics. The Kaplan-Meier curve was used to illustrate PFS and OS.
Table 2 - Response of the studied patients

Results
In the current study the Mean age at diagnosis was 57.5 years, more than one half of patients were ?60 years. Performance status was 0 in 7 (23.33%) patients and 1 in 23 (76.67%) patients. Seventeen patients (56.6%) had right side primary tumor. Ten patients (33.33%) had a single metastatic site, while 20 (66.67%) patients had multiple metastatic sites. Liver metastasis was observed in 16 (53.33%) patients, with the liver being the only affected site in 5 (16.67%) patients. In addition, 9 (30%) patients had lung metastases. Prior treatment consisted of primary resection in 15 (50%) patients, followed by adjuvant therapy in 13 (43.33%) patients (table 1).
Treatment Response
All patients were assessed for treatment response after full course of BEV, mFOLFOXIRI. Two patients (6.67%) achieved a complete response, while 16 (53.33%) patients showed a partial response. The disease was stable in 8 (26.67%) patients and progressive in 4 (13.33%) patients. The objective response rate was 60%. The disease control rate was 86.67%. Twenty patients (66.67%) had disease progression (table 2).
Survival
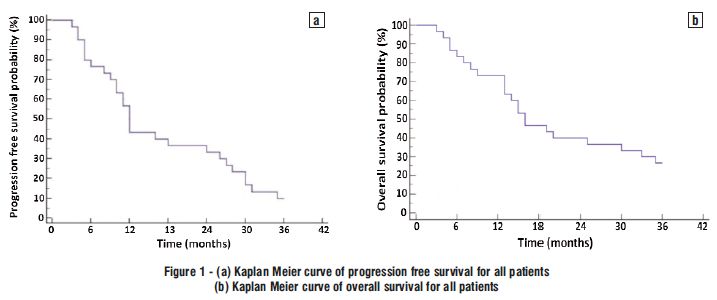
The PFS was 76.67% at 6 months, 43.33% at 12 months, 36.67% at 18 months, 33.33% at 24 months, and 10% at 36 months. The OS was 83.33 at 6 months, 73.33% at 12 months, 46.67% at 18 months, 40% at 24 months, and 26.67% at 36 months (fig. 1 a, b).
Progression occurred in 20 (66.67%) patients with a median PFS of 12 months. Death was reported in 22 (73.33%) patients with a median OS of 16 months (fig. 1 a, b).
Toxicity
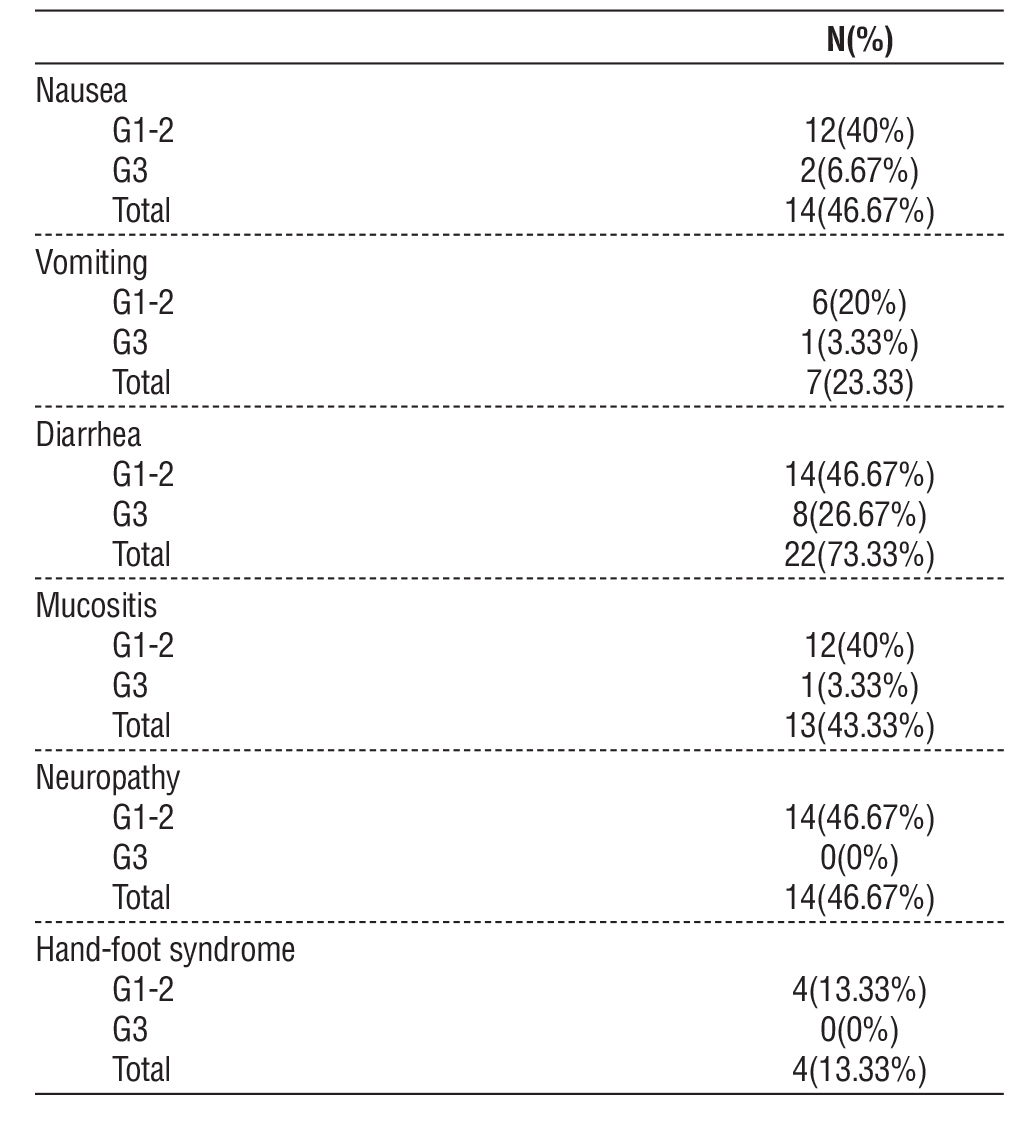
Diarrhea was the most prominent GIT toxicity in our study G1-2 in 14 (46.67%) patients and G3 in 8 (26.67%) patients. Grade 1-2 nausea and vomiting was reported in (40%,20%) of patients respectively, while G3 was reported in (6.67%,3.33%) of patients respectively. Mucositis was G1-2 in 12 (40%) patients and G3 in 1 (3.33%) patient. Neuropathy was G1-2 in 14 (46.67%) patients and hand-foot syndrome was G1-2 in 4 (13.33%) patients (table 3).
Table 3 - Toxicity of the studied patients
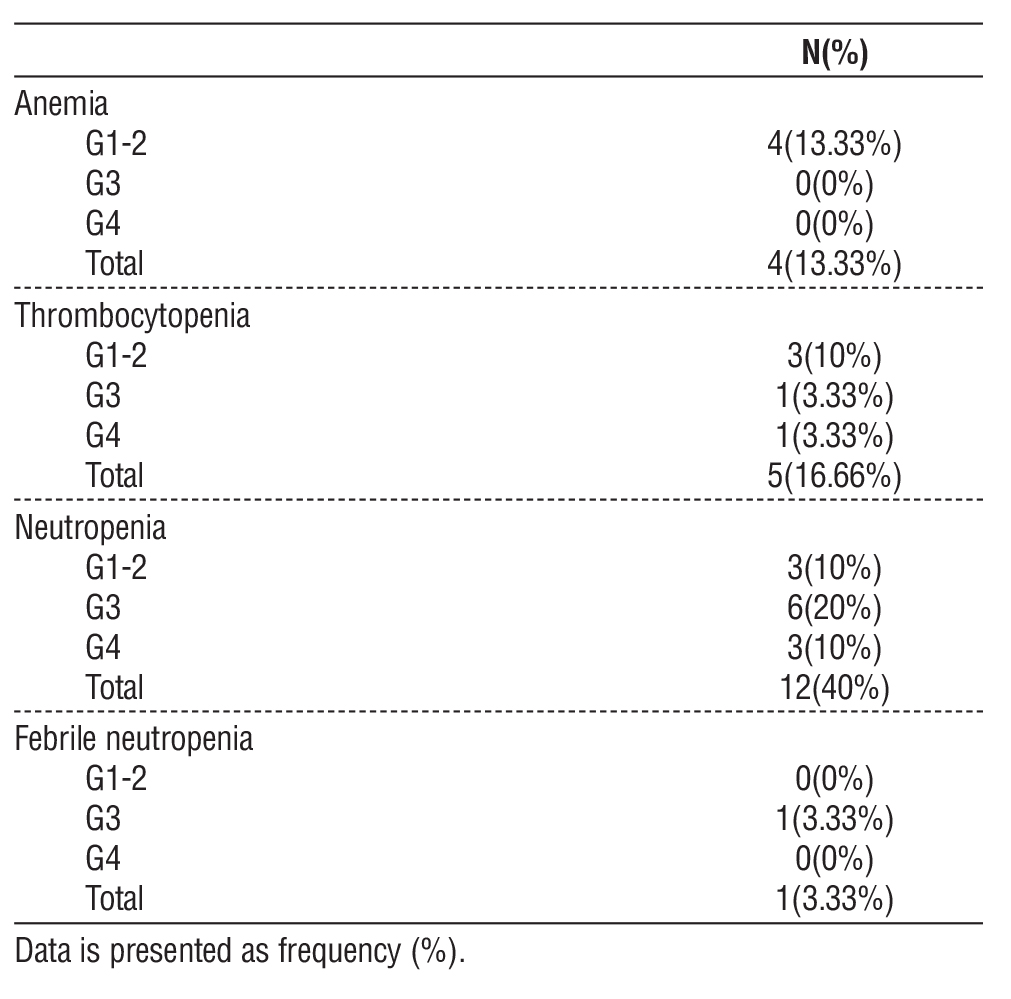
Regarding hematological disorders, anemia G1-2 was reported in 4 (13.33%) patients. Thrombo-
cytopenia was G1-2 in 3 (10%) patients, G3 in 1 (3.33%) patient, and G4 in 1 (3.33%) patient. Neutropenia was G1-2 in 3 (10%) patients, G3 in 6 (20%) patients and G4 in 3 (10%) patients. Febrile neutropenia was G3 in 1 (3.33%) patient (table 4).
The dose was reduced in 11 (36.6%) patients, including those with GIII diarrhea. Nine patients (30%) received G-CSF as secondary prophylaxis, while 14 patients (46.66%) started primary GCSF prophylaxis.
Table 4 - Hematological toxicity of the studied patients
Discussion
Treatment of metastatic colorectal cancer depends on various clinical factors, tumor location and molecular features as (RAS, RAF, MMR) status.
Persistent associations between BRAF V600E mutations and unfavorable outcomes in mCRC (14) highlight a critical need for improved treatment options. Combining BEV with FOLFOXIRI chemotherapy yields better results than FOLFOXIRI alone (15). We chose a BEV dose of 5 mg/ kg based on a previous study that used BEV ranging from 5 to 7.5 mg/kg (16).
Our research revealed favorable clinical results, with an objective response rate (ORR) of 60%, comprising 6.67% complete responses and 53.33% partial responses. These results were comparable to results from the TRIBE study, which showed an ORR of 65%. However, the results are slightly lower than the ORR of 72% reported by Loupakis et al. (17) in a phase II trial of FOLFOXIRI plus bevacizumab. However, our disease control rate (DCR) of 86.67% is comparable to their findings of 88%. The discrepancy in ORR might be attributed to differences in patient selection.
In addition, our results align with the outcomes of the phase II trial (FIRE-4.5; AIO KRK0016), which randomly assigned previously untreated RAS wild-type BRAF V600E-mutated mCRC patients to FOLFOXIRI plus either bevacizumab or cetuximab (18). FOLFOXIRI plus bevacizumab had an ORR of 67% versus 51% in the cetuximab arm.
The progression and mortality rates in our study were 66.67% and 73.33%, respectively, reflecting the aggressive nature of this mutation. This underscores the challenging nature of BRAF V600E-mutant mCRC, aligning with results from BRACELET study (19), that reported high progression rates of 53% despite the use of FOLFOXIRI plus anti-VEGF therapy group.
In the current study PFS decreased from 76.67% at 6 months to 10% at 36 months and a median OS of 16 months, declining from 83.33% at 6 months to 26.67% at 36 months.
The therapeutic impact observed in our study aligns with the Naz et al. (20) meta-analysis, which confirmed the benefit of combining bevacizumab with chemotherapy in improving survival outcomes (HR for OS: 0.689). Our findings also complement Song et al., (21) meta-analysis, highlighted the disease control advantages of bevacizumab-based regimens. However, unlike the BRACELET study (18), which found no significant survival benefit of FOLFOXIRI plus bevacizumab over doublet regimens, our study demonstrates a high RR, PFS, and OS. This contrast might stem from variations in patient selection.
Diarrhea as the most prominent GIT side effect, affecting 73.33% of patients, with 26.67% experiencing grade 3 severity. This finding is consistent with the AtezoTRIBE study by Antoniotti et al. (22), who reported Diarrhea as a significant adverse effect of FOLFOXIRI. Moreover, Aranda et al. (16) also reported significant gastrointestinal toxicities. Other GIT toxicities included nausea (46.67%), mucositis (43.33%), and polyneuropathy (46.67%), with severity profiles resembling those observed in the TRIBE study (22).
Hematological toxicities were dominated by neutropenia (40%), a rate that mirrors findings from the BRACELET study. Shimozaki et al., (19), where neutropenia was the most common grade ?3 adverse events in triplet regimens. Also, our findings are like the Hurwitz et al. (15) study, which observed grade ?3 treatment-emergent adverse events in over 85% of patients receiving FOLFOXIRI plus bevacizumab. In our research, dose reduction was implemented in 39.6% of patients. Thirty percent of the patients received G-CSF in subsequent doses due to the observed high incidence of neutropenia, and 46.66% received GCSF as a primary prophylaxis.
In the current study, the Bevacizumab-FOLFOXIRI regimen demonstrated significant therapeutic efficacy, achieving a favorable objective response rate (ORR) of 60%, which comprised 6.67% complete responses and 53.33% partial responses. Although Grade 3 diarrhea and neutropenia were reported in 26.6% and 20% of the cohort, respectively, the majority of adverse events were limited to Grades 1–2. These results suggest that while high-grade toxicities occur, they remain clinically manageable, ultimately supporting the regimen's robust clinical benefit.
However, the relatively lower incidence of thrombo-cytopenia (16.66%) and anemia (13.33%) further underscores the tolerability of the regimen in our cohort. Additionally, our toxicity findings resonate with those reported in the TRIBE study Cremolini et al., (23), confirming the regimen’s feasibility in real-world settings.
BRAFV600E mCRC patients have poor survival, and there are unmet needs for more effective treatment for this group of patients, BRAF mutation induces resistance and affects the response to EGFR inhibitors (24-26). Therefore, Resistance to EGFR inhibitors can be mitigated by adding a BRAF inhibitor to the treatment regimen. Data from the BEACON trial supporting the use of the chemotherapy-free protocol, BRAF inhibitor (encorafenib) plus cetuximab, for patients with RAS wild-type- BRAF V600E mutant mCRC that has progressed after initial chemotherapy (27).
Limitations of the Study
Limitations of this study should be taken into consideration when interpreting the findings. While the lack of a control arm in this study may limit a definitive assessment of the role of Bevacizumab-FOLFOXIRI in treating BRAF V600E mutant mCRC, our conclusions are bolstered by established data from previous large randomized controlled trials. A larger sample size with subgroup analysis is highly recommended, in the current study MMR/MSI status was assessed for financially supported patients only and should be assessed for all patients. This is significant because the BRAF mutation and dMMR/MSI-H status frequently overlap (28).
Conclusions
Bevacizumab, in combination with FOLFOXIRI, demonstrates efficacy in treating BRAF V600E mutant mCRC patients with good performance status, achieving high response rates and disease control. However, the treatment is associated with considerable toxicities, requiring careful monitoring and management.
Author’s Contribution
Dr. Asmaa Elkady and Dr. Sally Mashaal contributed to the trial conception, design and data analysis. Dr. Fatma Gharib contributed to the writing, critical revision, and the final approval of the version to be published.
Conflict of Interest
None.
References
1. Siegel RL, Kratzer TB, Giaquinto AN, Sung H, Jemal A. Cancer statistics, 2025. CA Cancer J Clin 2025; 75:10.
2. Golshani G, Zhang Y. Advances in immunotherapy for colorectal cancer: a review. Ther Adv Gastroenterol. 2020;13:17562848 20917527.
3. Ilic M, Ilic I. Cancer of colon, rectum and anus: the rising burden of disease worldwide from 1990 to 2019. J Public Health (Oxf). 2024;46(1):20-29.
4. Zheng E, W?odarczyk M, W?giel A, Osielczak A, Mo?d?an M, Biskup, L, et al. Navigating through novelties concerning mCRC treatment-the role of immunotherapy, chemotherapy, and targeted therapy in mCRC. Front Surg. 2024;11:1398289.
5. Modest DP, Pant S, Sartore-Bianchi A. Treatment sequencing in metastatic colorectal cancer. Eur J Cancer. 2019;109:70-83.
6. Hanrahan AJ, Chen Z, Rosen N, Solit DB. BRAF - A tumour-agnostic drug target with lineage-specific dependencies. Nat Rev Clin Oncol. 2024;21(3):224-247.
7. Tsai HL, Huang CW, Chen YC, Su WC, Chang TK, Chen PJ, et al. Real-world outcomes of first-Line FOLFIRI plus bevacizumab with irinotecan dose escalation versus FOLFOXIRI plus bevacizumab in BRAFV600E-mutant metastatic colorectal cancer: The preliminary data from a single-center observational study. Medicina (Kaunas). 2023;59(12):2108.
8. Morris VK, Bekaii-Saab T. Improvements in clinical outcomes for BRAF(V600E) -mutant metastatic colorectal cancer. Clin Cancer Res. 2020;26(17):4435-4441.
9. Kayhanian H, Goode E, Sclafani F, Ang JE, Gerlinger M, de Castro DG, et al. Treatment and survival outcome of BRAF-mutated metastatic colorectal cancer: a retrospective matched case-control study. Clin Colorectal Cancer. 2018;17(1):e69-e76.
10. Pietrantonio F, Morano F, Corallo S, Miceli R, Lonardi S, Raimondi A, et al. Maintenance therapy with panitumumab alone vs panitumumab plus fluorouracil-leucovorin in patients with RAS wild-type metastatic colorectal cancer: a phase 2 Randomized Clinical Trial. JAMA Oncol. 2019;5(9):1268-1275.
11. Moretto R, Elliott A, Rossini D, Intini R, Conca V, Pietrantonio F, et al. Benefit from upfront FOLFOXIRI and bevacizumab in BRAFV600E-mutated metastatic colorectal cancer patients: does primary tumour location matter? Br J Cancer. 2022;127(5):957-967.
12. Gmeiner WH. Recent advances in therapeutic strategies to improve colorectal cancer treatment. Cancers (Basel). 2024; 16(5):1029.
13. Shivaji U, Jeffery L, Gui X, Smith S, Ahmad O, Akbar A, et al. Immune checkpoint inhibitor-associated gastrointestinal and hepatic adverse events and their management. Ther Adv Gastroenterol. 2019;12: 1756284819884196.
14. Yuan ZX, Wang XY, Qin QY, Chen DF, Zhong QH, Wang L, et al. The prognostic role of BRAF mutation in metastatic colorectal cancer receiving anti-EGFR monoclonal antibodies: a meta-analysis. PLoS One. 2013;8(6):e65995.
15. Hurwitz HI, Tan BR, Reeves JA, Xiong H, Somer B, Lenz HJ, et al. Phase II Randomized Trial of Sequential or Concurrent FOLFOXIRI-Bevacizumab Versus FOLFOX-Bevacizumab for Metastatic Colorectal Cancer (STEAM). Oncologist. 2019;24(7): 921-932.
16. Aranda E, Viéitez JM, Gómez-España A, Gil Calle S, Salud-Salvia A, Graña B, et al. OLFOXIRI plus bevacizumab versus FOLFOX plus bevacizumab for patients with metastatic colorectal cancer and ?3 circulating tumour cells: the randomised phase III VISNÚ-1 trial. ESMO Open. 2020;5(6):e000944.
17. Loupakis F, Cremolini C, Salvatore L, Masi G, Sensi E, Schirripa M, et al. FOLFOXIRI plus bevacizumab as first-line treatment in BRAF mutant metastatic colorectal cancer. Eur J Cancer. 2014; 50(1):57-63.
18. Stintzing S, Heinrich K, Tougeron D, Modest DP, Schwaner I, Eucker J, et al. FOLFOXIRI plus Cetuximab or Bevacizumab as first-line treatment of BRAF V600E-mutant metastatic colorectal cancer: The randomized phase II FIRE-4.5 (AIO KRK0116) study. J Clin Oncol. 2023;41(25):4143-4153.
19. Shimozaki K, Hirata K, Sato T, Nakamura M, Kato K, Hirano H, et al. WJOG13219G: The efficacy and safety of FOLFOXIRI or doublet plus anti-VEGF therapy in previously untreated BRAFV600E mutant metastatic colorectal cancer: A multi-institutional registry-based study (BRACELET study). Clin Colorectal Cancer. 2022;21(4):339-346.
20. Naz T, Rehman Au, Shahzad A, Rasool MF, Saleem Z, Hussain R. Impact of bevacizumab on clinical outcomes and its comparison with standard chemotherapy in metastatic colorectal cancer patients: a systematic review and meta-analysis. J Pharm Policy Pract. 2024;17(1):2354300.
21. Song Y, Mao Q, Zhou M, Liu CJ, Kong, L. & Hu, T. 2024. Effectiveness of bevacizumab in the treatment of metastatic colo-rectal cancer: a systematic review and meta-analysis. BMC Gastroenterol. 2024;24(1):58.
22. Antoniotti C, Borelli B, Rossini D, Pietrantonio F, Morano F, Salvatore L, et al. AtezoTRIBE: a randomised phase II study of FOLFOXIRI plus bevacizumab alone or in combination with atezolizumab as initial therapy for patients with unresectable metastatic colorectal cancer. BMC Cancer. 2020;20(1):683.
23. Cremolini C, Loupakis F, Antoniotti C, Lupi C, Sensi E, Lonardi S, et al. FOLFOXIRI plus bevacizumab versus FOLFIRI plus bevacizumab as first-line treatment of patients with metastatic colorectal cancer: updated overall survival and molecular subgroup analyses of the open-label, phase 3 TRIBE study. Lancet Oncol. 2015;16(13):1306-15.
24. Pietrantonio F, Petrelli F, Coinu A, Di Bartolomeo M, Borgonovo K, Maggi C, et al. 2015. Predictive role of BRAF mutations in patients with advanced colorectal cancer receiving cetuximab and panitumumab: a meta-analysis. Eur J Cancer. 2015;51(5): 587-94.
25. Rowland A, Dias MM, Wiese MD, Kichenadasse G, McKinnon RA, Karapetis CS, et al. Meta-analysis of BRAF mutation as a predictive biomarker of benefit from anti-EGFR monoclonal antibody therapy for RAS wild-type metastatic colorectal cancer. Br J Cancer. 2015; 112(12):1888-94.
26. Cohen R, Liu H, Fiskum J, Adams R, Chibaudel B, Maughan TS, et al. BRAF V600E mutation in first-line metastatic colorectal cancer: An analysis of individual patient data from the ARCAD database. J Natl Cancer Inst. 2021;113(10):1386-1395.
27. Van Cutsem E, Huijberts S, Grothey A, Yaeger R, Cuyle PJ, Elez E, et al. Binimetinib, Encorafenib, and Cetuximab triplet therapy for patients with BRAF V600E-mutant metastatic colorectal cancer: safety lead-in results from the phase III BEACON colorectal cancer study. J Clin Oncol. 2019;37(17):1460-1469.
28. Fouchardière C, Cohen R, Malka D, Guimbaud R, Bourien H, Lièvre A, et al. Characteristics of BRAF V600E mutant, deficient mismatch repair/proficient mismatch repair, metastatic colorectal cancer: a multicenter series of 287 patients. Oncologist. 2019;24(12):e1331-e1340.
Full Text Sources:
Abstract:
Views: 250

For Authors
Journal Subscriptions

Dec 2025
Supplements
Instructions for authors
Online submission
Contact
e-ISSN: 2601 - 1700 (online)
ISSN-L: 2559 - 723X
Journal Abbreviation: Surg. Gastroenterol. Oncol.
Surgery, Gastroenterology and Oncology (SGO) is indexed in:
- SCOPUS
- EBSCO
- DOI/Crossref
- Google Scholar
- SCImago
- Harvard Library
- Open Academic Journals Index (OAJI)
Surgery, Gastroenterology and Oncology (SGO) is an open-access, peer-reviewed online journal published by Celsius Publishing House. The journal allows readers to read, download, copy, distribute, print, search, or link to the full text of its articles.
Time to first editorial decision: 25 days
Rejection rate: 61%
CiteScore: 0.2

Meetings and Courses in 2025
Meetings and Courses in 2024
Meetings and Courses in 2023
Meetings and Courses in 2022
Meetings and Courses in 2021
Meetings and Courses in 2020
Meetings and Courses in 2019
Verona expert meeting 2019

Surgery, Gastroenterology and Oncology applies the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits readers to copy and redistribute the material in any medium or format, remix, adapt, build upon the published works non-commercially, and license the derivative works on different terms, provided the original material is properly cited and the use is non-commercial. Please see: https://creativecommons.org/licenses/by-nc/4.0/
Publisher’s Note:
The opinions, statements, and data contained in article are solely those of the authors and not of Surgery, Gastroenterology and Oncology journal or the editors. Publisher and the editors disclaim responsibility for any damage resulting from any ideas, instructions, methods, or products referred to in the content.
 IASGO Society News
IASGO Society News