Surgery, Gastroenterology and Oncology
Official journal of the International Association of Surgeons, Gastroenterologists and Oncologists
|
Biological Characteristics of Breast Cancer among Jordanian Women: A retrospective Single Center Cohort Study
ORIGINAL PAPER
ABSTRACT
Introduction: Breast cancer is a heterogeneous disease that encompasses a wide range of pathological entities and clinical behaviors. It is classified into several subtypes based on their histopathological characteristics, tumor grade and biological features. This classification is important for studying breast cancer etiology, predicting clinical course, and making decisions related to breast cancer treatment. This study aims to identify biological characteristics of breast cancer among a group of Jordanian women who were referred to a breast clinic in a tertiary hospital in Amman.
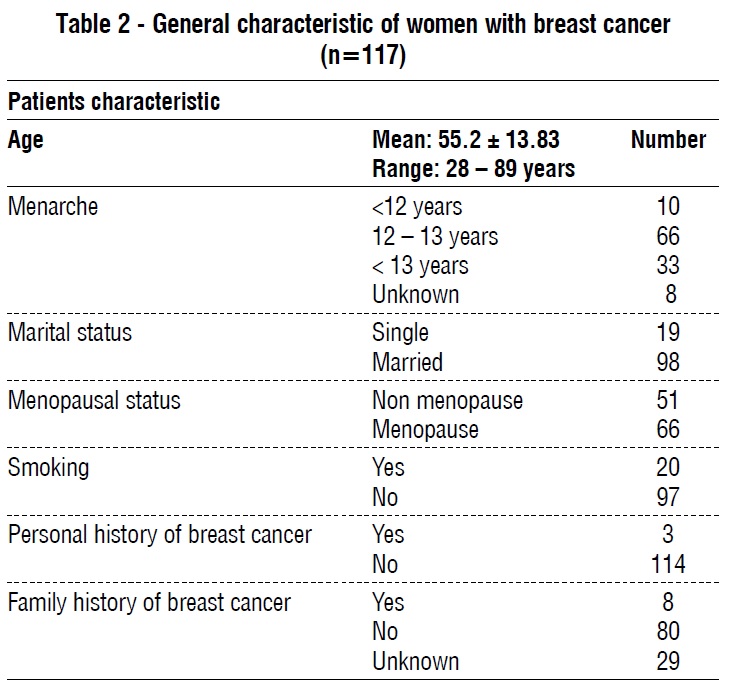
Methodology: This is a retrospective cohort study that included initially a total of 119 female patients with breast cancer during the period between January 2018 and January 2020 at tertiary hospital in Jordan. Patients’ data were retrieved from the electronic health record system including their pathologic reports. Pathological reports were reviewed by the pathologist were histopathological features and tumor biological characteristics were confirmed. Breast cancer cases were classified into different biological subtypes based on surrogate definitions of intrinsic subtypes of breast cancer that was adopted in the 2011 St Gallen Consensus.
Results: A total of 117 patients were available for analysis with a mean age of 55.2 years and age range between 28 and 89 years. Patients were divided into group A (age 50 years; n=50) and group B (age > 50; n=67). The most common tumor was invasive ductal carcinoma (84.6%) followed by invasive lobular carcinoma. Metastatic axillary lymph node involvement was proved in 66.4% and 19 patients had de-novo metastasis. Biologically, the expression of ER, PR and HER2 were 84.1%, 84.1% and 36.7% respectively. Patients younger than 50 years had statistically higher levels of Ki67. Luminal B breast cancer was the most prevalent subtype in our patients.
Conclusion: Luminal B breast cancer is the most common prevalent intrinsic subtype of breast cancer among our patients. The expression of HER2/neu gene is considered high as well as large percentage of patients have high Ki67. Although it is not informative as genebased assays, IHC-based assays can be utilized for classification of breast cancer based on its biological characteristics at reasonable costs especially in low-middle income countries.
INTRODUCTION
Globally, Breast cancer is the most prevalent invasive cancer and the second leading cause of cancer deaths in women. It is heterogeneous disease with a wide range of pathological entities and clinical behaviors, thus posing great challenges in understanding the precise molecular pathogenesis (1). In Jordan, according to the Jordanian cancer registry, it is considered the most common malignancy and the third in cancer related mortality following lung and colorectal cancer.
Breast cancer is classified into different subtypes, this classification is essential for studying its etiology, predicting its clinical course, and making treatment decisions (2). The Distinctions between breast cancer subtypes is based on both patient and tumor characteristics (i.e. histology, grade, biology, genetic profile and stage) (3).
Invasive breast carcinomas either ductal or lobular subtypes represent the main infiltrating carcinomas, with lobular subtype being less frequent (4). The rely on histopathological features alone to classify invasive mammary carcinomas might result in diagnostic difficulty because of overlapping histopathologic features, particularly with invasive lobular carcinoma (ILC) variants and pleomorphic ILC (4,5). Proper histopathologic categorization of breast carcinomas has prognostic implications (6).
Generally, the progression of breast cancer has been viewed as a linear multi-step process, from hyperplasia, atypical hyperplasia, carcinoma in situ, and to invasive and metastatic carcinoma, supported by evidence from clinical, pathological, and genetic studies (7,8). Breast carcinogenesis is believed to be a series of stochastic genetic events that lead to distinct and divergent pathways towards invasive carcinoma (9). One of the Key pathological features of breast cancer is the nuclear grade that is associated with clinical outcome, and distinct genetic changes (10,11).
Reliance on estrogen receptor (ER-α), progesterone receptor (PR), and human epidermal growth factor receptor 2 (HER2/neu) expression to guide the clinical management and to predict clinical outcome and disease prognosis is far from satisfactory. Additional molecular markers are also required to predict clinical outcome and devise optimal individualized therapy (12, 13).
The expression of these markers in pure DCIS and DCIS/IDC provides better understanding of the precise relationships between pure DCIS and co-existing DCIS/IDC and to identify potential molecular markers for prognostic, therapeutic, and preventive purposes has been shown by Shackney et al (14). Based on mammography screening, DCIS component is identified in 30%-60% of breast IDC (19,20), while those with pure IDC might develop de novo without identifying underlying in situ carcinoma. However, patients with combined IDC-DCIS are assumed to develops IDC from a pre-existing DCIS lesion (15,16). In comparison to patients with pure IDC, patients with IDC-DCIS are more likely to be younger, having smaller or non-palpable tumors with lower probability of lymph node involvement (16-18). In addition, it was tested that the progression of in situ ductal carcinoma of breast to invasive ductal carcinoma is associated with alterations of ER, PGR and HER-2/neu protein expression (2).
The Expression patterns of estrogen receptor are well-established and defined for invasive breast carcinoma, but are not as well-defined for breast carcinoma in situ (CIS) (21). Over years it has been postulated that age-adjusted incidence trends differed for CIS and Invasive breast carcinoma in the United States, this changes possibly due to screening mammography, and/or etiologic diversity. Age-specific incidence patterns suggested that carcinogenic events starting early in reproductive life had greater impact upon CIS and invasive breast carcinoma defined by ER-negative expression than invasive breast carcinoma defined by ER-positive expression (21).
Estrogen receptor (ER) status whether positive or negative has been established as a predictive factor for breast cancer treatment and to a lesser extent, the significance of progesterone receptor (PR) status. Estrogens are important for stimulating growth of significant percentage of breast cancers and progesterone has roles in breast development and tumorigenesis. ER positive tumors are more responsive to hormonal adjuvant therapies than ER negative tumors, and have better prognosis for survival in comparison to ER negative tumors (22,23).
The expression of the c-erbB2 gene (HER-2/neu) proto-oncogene that is expressed in 10-34% of breast cancers and is associated with an aggressive behavior and poor clinical outcome (24), this can be in presence or absence of estrogen or progesterone in cancerous cells. Triple-negative breast cancers are lacking the expression of ER, PR and HER2/neu. In majority of cases, they carry the "basal-like” molecular phenotype that is identified using cDNA microarrays. Although most triple-negative breast tumors do cluster within the basal-like subgroup, there is up to 30% discordance between the two groups, most of the BRCA-1 associated breast cancer are classified within this category.
METHODOLOGY
This is a retrospective cohort study that included initially a total of 119 female patients who were diagnosed with breast cancer during the period between January, 2018 and January, 2020 at the breast clinic in a tertiary hospital in Amman, Jordan. Patients’ data were retrieved from the electronic health record system including their pathologic reports, which were reviewed by the pathologist to confirm both histopathological and biological characteristics of breast tumors.
All the cases were routinely evaluated immunohistochemically for estrogen and progesterone hormone receptor status and for HER2/neu gene expression using standard immunoperoxidase method. Evaluation of IHC staining was routinely done by the local pathologists according to international guidelines.
With regard to the hormone receptors, the results were expressed as positive or negative, whereas for HER2 expression, the results were reported according to American Society of Clinical Oncology (ASCO)/College of American Pathologists (CAP) guideline, updated on 2013. Breast cancers expressing ER and PR in >1% of neoplastic cells were considered as positive. For Her2/neu, tumors were scored as negative if the membrane staining was negative or weak and incomplete in less than or equal to 10% of tumor cells (score 0 or 1+), whereas cases with strong complete membrane staining in >10% of cells were scored as positive (score 3+). All cases with weak to moderate complete membrane screening observed in >10% of tumor cells were considered equivocal (score 2+) and were considered as negative or positive according to the results of FISH analysis.
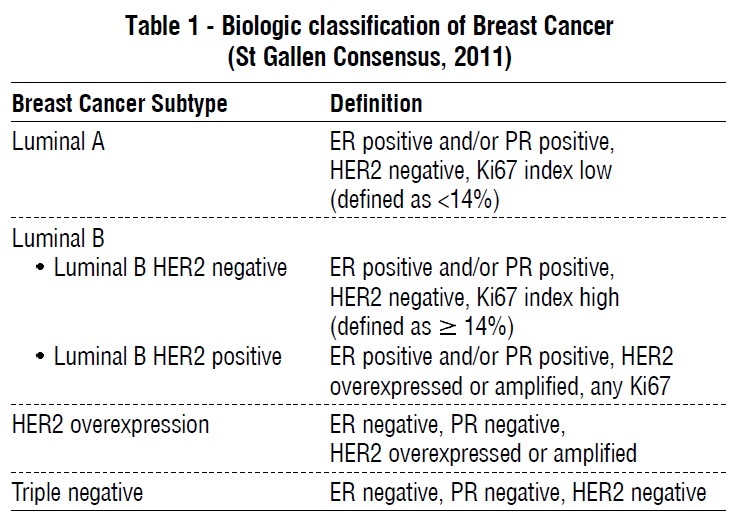 Patients were classified according on their age at diagnosis into 2 groups; group A (age 50 years or less) and group B (age more than 50 years). Analysis of patients' characteristics, tumor histology and tumor biology were performed. Breast cancer cases were classified into different biological subtypes based on surrogate definitions of intrinsic subtypes of breast cancer that was adopted in the 2011 St Gallen Consensus, table 1. Data were analyzed using the statistical package for the social sciences version 23 (SPSS Inc., Chicago, IL) statistical software. The analyses included descriptive statistics and chi-square test. A P < 0.05 was considered statistically significant.
Patients were classified according on their age at diagnosis into 2 groups; group A (age 50 years or less) and group B (age more than 50 years). Analysis of patients' characteristics, tumor histology and tumor biology were performed. Breast cancer cases were classified into different biological subtypes based on surrogate definitions of intrinsic subtypes of breast cancer that was adopted in the 2011 St Gallen Consensus, table 1. Data were analyzed using the statistical package for the social sciences version 23 (SPSS Inc., Chicago, IL) statistical software. The analyses included descriptive statistics and chi-square test. A P < 0.05 was considered statistically significant.
RESULTS
A total of 119 women were included in the study. Two patients were excluded because of incomplete data. Patient’s age ranged between 28 and 89 years, the mean age of patients was 55.2 years with a standard deviation of 13.83 years. Patients were grouped into two groups, the first group "Group A" included cases aged 50 years or less (n=50, mean age 42.34 years ± 6.17), and the second group "Group B" included cases older than 50 years of age (n=69, mean age 64.49 ± 9.7).
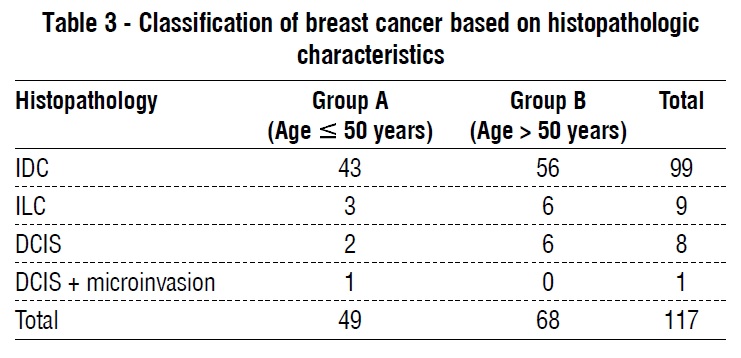
 A total of 67 patients (56.3%) had unilateral left sided breast cancer, 48 patients (40.3%) had unilateral right sided breast cancer, and only 4 patients (3.4%) were diagnosed with bilateral breast cancer table 2. Following the study of 117 cases, 99 cases (84.6%) were invasive ductal carcinoma (IDC), 9 cases (7.7%) invasive lobular carcinoma (ILC), 8 patients (6.8%) ductal carcinoma in situ (DCIS), and 1 case (0.9%) was ductal carcinoma in situ with micro-invasion. Tumor classification based on histopathologic characteristics is illustrated in table 3.
A total of 67 patients (56.3%) had unilateral left sided breast cancer, 48 patients (40.3%) had unilateral right sided breast cancer, and only 4 patients (3.4%) were diagnosed with bilateral breast cancer table 2. Following the study of 117 cases, 99 cases (84.6%) were invasive ductal carcinoma (IDC), 9 cases (7.7%) invasive lobular carcinoma (ILC), 8 patients (6.8%) ductal carcinoma in situ (DCIS), and 1 case (0.9%) was ductal carcinoma in situ with micro-invasion. Tumor classification based on histopathologic characteristics is illustrated in table 3.
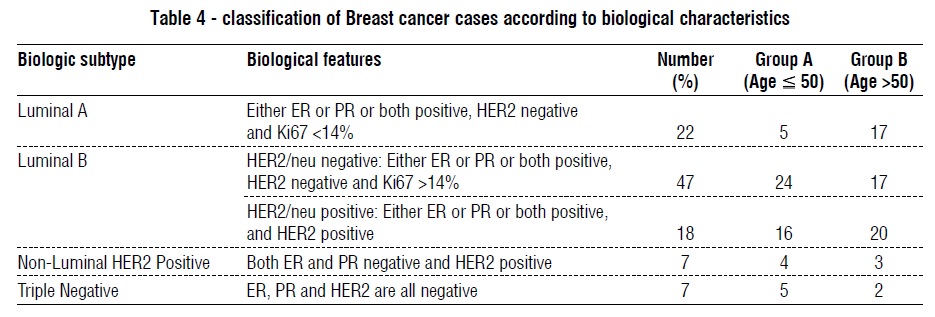
 Regarding clinical tumor characteristics, the minimal tumor size for invasive tumors was 1.0 cm, the maximal size 10.0 cm with a mean size of 3.73 cm ± 1.82. In group A, the mean size was 3.69 cm ± 1.69, while in group B the mean size was 3.758 cm ± 1.9 with no statistical difference between them. Among the 117 patients, metastatic axillary lymph nodes were positive in 66.4%, in group A, 70% were positive while in group B, malignant axillary nodes were positive in 63.8% of the cases. Metastatic work-up was available for 112 patients (group A: 45 patients; group B: 67 patients). Among them, 19 patients (17%) had distant metastasis at time of diagnosis, 9 patients in group A and 10 patients in group B, (P=0.6). The biological and molecular features of 107 cases of invasive carcinomas were studied (i.e., IDC, ILC, DCIS with micro-invasion), of which 47 cases were in the age group 50 years or younger (group A) and 60 cases in the age group above 50 years (group B). Estrogen receptors (ER) were positive in 84.1% of the cases (n= 90), among patients in group A; 36 cases (76.6%) were positive and 11 cases (23.4%) were negative. In comparison, 90% of patients in group B were ER positive (n = 54), while 6 cases were negative (10%), (P = 0.068). Similar to ER, progesterone receptors were positive in 84.1% of the cases, 80.9% of patients in group A and 86.7% in group B were positive for PR (P = 0.43). The expression of human epidermal growth factor receptor 2 (HER2) was positive in 43 cases (36.7%), 20 cases in group A (42.6%) and 23 cases in group B (38.3%), (P = 0.69). The expression of cell proliferation biomarker, Ki-67 was studied in 89 cases of which, 27 cases (30.3%) had Ki67 ? 14%, 46.1% (41 cases) had Ki67 between 15%-50%, and 23.6% (21 cases) had Ki67 > 50%. Patients younger than 50 years have statistically higher levels of Ki67 (P = 0.0079). Based on the previous results we were able to classify the invasive carcinomas into different biological subtypes based on surrogate definitions of intrinsic subtypes of breast cancer that was adopted in the 2011 St Gallen Consensus, table 1. Out of all the 107 cases studied; 22 cases were Luminal A (20.6%), 71 cases were Luminal B (66.4%) of which 24 cases were HER2/neu positive. Only 7 cases were classified as nonluminal HER2/neu positive and 7 cases as triple negative breast cancer. Overall, 31 patients (28.9%) with invasive breast cancer were positive for HER2/neu. Analysis of cases based on age group is further illustrated in table 4.
Regarding clinical tumor characteristics, the minimal tumor size for invasive tumors was 1.0 cm, the maximal size 10.0 cm with a mean size of 3.73 cm ± 1.82. In group A, the mean size was 3.69 cm ± 1.69, while in group B the mean size was 3.758 cm ± 1.9 with no statistical difference between them. Among the 117 patients, metastatic axillary lymph nodes were positive in 66.4%, in group A, 70% were positive while in group B, malignant axillary nodes were positive in 63.8% of the cases. Metastatic work-up was available for 112 patients (group A: 45 patients; group B: 67 patients). Among them, 19 patients (17%) had distant metastasis at time of diagnosis, 9 patients in group A and 10 patients in group B, (P=0.6). The biological and molecular features of 107 cases of invasive carcinomas were studied (i.e., IDC, ILC, DCIS with micro-invasion), of which 47 cases were in the age group 50 years or younger (group A) and 60 cases in the age group above 50 years (group B). Estrogen receptors (ER) were positive in 84.1% of the cases (n= 90), among patients in group A; 36 cases (76.6%) were positive and 11 cases (23.4%) were negative. In comparison, 90% of patients in group B were ER positive (n = 54), while 6 cases were negative (10%), (P = 0.068). Similar to ER, progesterone receptors were positive in 84.1% of the cases, 80.9% of patients in group A and 86.7% in group B were positive for PR (P = 0.43). The expression of human epidermal growth factor receptor 2 (HER2) was positive in 43 cases (36.7%), 20 cases in group A (42.6%) and 23 cases in group B (38.3%), (P = 0.69). The expression of cell proliferation biomarker, Ki-67 was studied in 89 cases of which, 27 cases (30.3%) had Ki67 ? 14%, 46.1% (41 cases) had Ki67 between 15%-50%, and 23.6% (21 cases) had Ki67 > 50%. Patients younger than 50 years have statistically higher levels of Ki67 (P = 0.0079). Based on the previous results we were able to classify the invasive carcinomas into different biological subtypes based on surrogate definitions of intrinsic subtypes of breast cancer that was adopted in the 2011 St Gallen Consensus, table 1. Out of all the 107 cases studied; 22 cases were Luminal A (20.6%), 71 cases were Luminal B (66.4%) of which 24 cases were HER2/neu positive. Only 7 cases were classified as nonluminal HER2/neu positive and 7 cases as triple negative breast cancer. Overall, 31 patients (28.9%) with invasive breast cancer were positive for HER2/neu. Analysis of cases based on age group is further illustrated in table 4.
DISCUSSION
In Jordan, according to the latest statistics from the Jordanian cancer registry, breast cancer is the most prevalent cancer and the 3rd leading cause of cancer related deaths following lung and colorectal cancer (25). In 2015, a total of 1138 cases of breast cancer have been diagnosed with a crude incidence of 34.1 per 100,000 and age standardized ratio of 45.7 per 100,000. These figures showed an increase by 69% during the past decade. Approximately, 44.7% of case were less than 50 years old at time of diagnosis (26). The median age at diagnosis among Jordanian women is 51 years, which is 10 years younger than women in western countries (27,28).
Breast cancer is classified into relevant subtypes, this classification is important for studying breast cancer etiology, predicting clinical course, and making decisions related to breast cancer treatment (2). The Distinctions between subtypes of breast cancer is made on the basis of patient characteristics or phenotypic or genotypic characteristics of the tumor itself, as tumor stage, grade, histology, and genetic profile (3). In Jordan, few reports have discussed biological characteristics of breast cancer and categorized them in a standardized manner to facilitate understanding trends of breast cancer biology in addition to its impact on treatment and prognosis.
In the perspective of the crucial importance of early detection and diagnosis of breast cancer for subsequent treatment and prognosis, we did this study aiming to identify the biological characteristics of breast cancer at the time of diagnosis in a group of Jordanian patients who were diagnosed from the breast clinic in a tertiary hospital in Jordan. We categorized breast cancer biology mainly into 4 main subtypes based on surrogate definitions of intrinsic subtypes of breast cancer that was adopted in the 2011 St Gallen Consensus (29). Although IHC-based assays do not provide as much biological insight into tumor biology as gene-based ones do, they allow classification of tumor at affordable costs and in the absence of fresh tissue specimens (30).
In Jordan, according to one study published on 2013 (31), breast cancer was classified into 4 main subtypes (luminal A, Luminal B, HER2 positive, and basal cell like).
In this study, Ki67 was not considered in distinguishing between luminal B and luminal A. In addition, luminal B was not categorized based on HER2 neu expression. According to it, the majority of all the cases were classified as luminal A (60%). This was higher than what was reported by Salhia et al in Egypt (44.3%) and by Ben Abdelkrim et al in Tunis (51.5%) (32, 33). Luminal A subtype was also predominant among Asian Americans (34). According to our study, only 20.56% of the cases were classified within this category.
In the previous study from Jordan, only 13% of all the cases were classified as luminal B subtype (31), which was similar to figures previously reported in North American and European women (6–19%), Tunisian women (16%) (33), but in contrast to what was found in Egyptian women (24.6%) (32). According to the figures in table 4, majority of our patients were classified as luminal B (66.35%), 22.4% of all cases were sub-classified into Luminal B/Her 2 positives, which is almost approximate to the results of Egyptian study and differ from the results obtained in Jordanian University seven years ago.
HER2/neu expression was positive in 36.7% of all cases, this was comparable to data published from the royal medical services on 2015 where overall expression rate was 36% (35). Only 7 cases (6.5%) were classified as non-luminal HER2/neu positive. The percentage of cases that were reported in Jordan previously classified as non-luminal HER2/neu+ was 12%, which was similar to that seen in Asian American (12%), Egyptian women (12%), and African American women (11.6%), but slightly lower than in Tunisian women (14.5%) (32, 33, 34, 36). Moreover, we figured out that HER2/neu+ subtype was more prominent in the younger age group (8.5%) in less than 50 years vs. 5% in cases more than 50 years, respectively. This might be indicative of the role of HER2+ mutation in the development of breast cancer at younger ages, which is similar to results that have concluded in other studies (31).
Triple negative breast cancer has higher incidence among young patients (10.6 %) in comparison to older women (3.3 %) with an overall rate of 6.5%. our results were lower than previously reported from Jordan were 15% of cases were also classified as BCL subtype similar to the incidence in Sudan (15.9%), which is slightly less than in Egyptian and Tunisian women (19% and 18%, respectively) (31,32,33,37). The reported incidence of BCL subtype in Moroccan breast cancer patients (12.6%) (38), 8.5% among American Chinese, 5.9% among American Filipino, 5.7% in American Japanese (34), and 21.2% in African American women (36).
CONCLUSION
Luminal B breast cancer is the most common prevalent intrinsic subtype of breast cancer among our patients. The expression of HER2/neu gene is considered high as well as large percentage of patients have high Ki67. These biological features might be associated with unfavorable prognosis and response to treatment if not properly evaluated. Although it is not informative as gene-based assays, IHC-based assays can be utilized for classification of breast cancer based on its biological characteristics at reasonable coasts especially in low-middle income countries. This classification is essential for clinical practice as an indicator of tumor behavior, prognosis, survival and response to therapy.
The lack of a unified reporting system for breast cancer in Jordan mandates the urgent adoption of a well-recognized classification similar to the one we described. This will help in understanding current situation of breast cancer biology and correlate possible changes in future with underlying genetic mutations or other risk factors.
Limitation of the study
This is a single center retrospective study from a breast clinic in a tertiary hospital in Jordan, so these results reflect tumor characteristics for patients who are diagnosed in this center. The need for multicenter studies is essential before we can generalize our results for Jordanian women.
Conflict of interest
No conflict of interest among authors
Funding
This research is not funded.
Ethical approval
This study is approved by the institutional review board at the Hashemite University and Prince Hamza Hospital.
REFERENCES
1. Tang P, Hajdu SI, Lyman GH. Ductal carcinoma in situ: a review of recent advances. Curr Opin Obstet Gynecol. 2007;19(1):63-7.
2. Hussein MR, Abd-Elwahed SR, Abdulwahed AR. Alterations of estrogen receptors, progesterone receptors and c-erbB2 oncogene protein expression in ductal carcinomas of the breast. Cell Biol Int. 2008;32(6):698-707.
3. Phipps AI, Li CI. Breast Cancer Biology and Clinical Characteristics. C.I. Li (ed.) Breast Cancer Epidemiology. New York: Springer; 2010.
4. Pinder SE, Elston CW, Ellis IO, editors; Elston CW, Ellis IO, editors. Invasive carcinoma: usual histological types. The Breast. 3rd ed. Edinburgh, Scotland: Churchill Livingstone; 1998. p. 283–337.
5. Wahed A, Connelly J, Reese T. E-cadherin expression in pleomorphic lobular carcinoma: an aid to differentiation from ductal carcinoma. Ann Diagn Pathol. 2002;6(6):349-51.
6. Ellis IO, Galea M, Broughton N, Locker A, Blamey RW, Elston CW. Pathological prognostic factors in breast cancer. II. Histological type. Relationship with survival in a large study with long-term follow-up. Histopathology. 1992;20(6):479-89.
7. Dupont WD, Page DL. Risk factors for breast cancer in women with proliferative breast disease. N Engl J Med. 1985;312(3):146-51.
8. Singletary SE. A working model for the time sequence of genetic changes in breast tumorigenesis. J Am Coll Surg. 2002;194(2): 202-16.
9. Buerger H, Otterbach F, Simon R, Schäfer KL, Poremba C, Diallo R, et al. Different genetic pathways in the evolution of invasive breast cancer are associated with distinct morphological subtypes. J Pathol. 1999;189(4):521-6.
10. Buerger H, Otterbach F, Simon R, Poremba C, Diallo R, Decker T, et al. Comparative genomic hybridization of ductal carcinoma in situ of the breast-evidence of multiple genetic pathways. J Pathol. 1999; 187(4):396-402.
11. Steinman S, Wang J, Bourne P, Yang Q, Tang P. Expression of cytokeratin markers, ER-alpha, PR, HER-2/neu, and EGFR in pure ductal carcinoma in situ (DCIS) and DCIS with co-existing invasive ductal carcinoma (IDC) of the breast. Ann Clin Lab Sci. 2007;37(2): 127-34.
12. Nofech-Mozes S, Spayne J, Rakovitch E, Hanna W. Prognostic and predictive molecular markers in DCIS: a review. Adv Anat Pathol. 2005;12(5):256-64.
13. Gasparini G, Longo R, Torino F, Morabito A. Therapy of breast cancer with molecular targeting agents. Ann Oncol. 2005;16 Suppl 4:iv28-36.
14. Shackney SE, Silverman JF. Molecular evolutionary patterns in breast cancer. Adv Anat Pathol. 2003;10(5):278-90.
15. Pinder SE, Ellis IO. The diagnosis and management of pre-invasive breast disease: ductal carcinoma in situ (DCIS) and atypical ductal hyperplasia (ADH)--current definitions and classification. Breast Cancer Res. 2003;5(5):254-257.
16. Wong H, Lau S, Yau T, Cheung P, Epstein RJ. Presence of an in situ component is associated with reduced biological aggressiveness of size-matched invasive breast cancer. Br J Cancer. 2010;102(9): 1391-1396.
17. Dieterich M, Hartwig F, Stubert J, Klöcking S, Kundt G, Stengel B, Reimer T, Gerber B. Accompanying DCIS in breast cancer patients with invasive ductal carcinoma is predictive of improved local recurrence-free survival. Breast. 2014;23(4):346-51.
18. Chagpar AB, McMasters KM, Sahoo S, Edwards MJ. Does ductal carcinoma in situ accompanying invasive carcinoma affect prognosis? Surgery. 2009;146(4):561–567. discussion 567–568
19. Jo BH, Chun YK. Heterogeneity of invasive ductal carcinoma: proposal for a hypothetical classification. J Korean Med Sci. 2006; 21(3):460-468.
20. Holland R, Connolly JL, Gelman R, Mravunac M, Hendriks JH, Verbeek AL, et al. The presence of an extensive intraductal component following a limited excision correlates with prominent residual disease in the remainder of the breast. J Clin Oncol. 1990; 8(1):113-8.
21. Anderson WF, Chu KC, Devesa SS. Distinct incidence patterns among in situ and invasive breast carcinomas,with possible etiologic implications. Breast Cancer Res Treat. 2004;88(2):149-59.
22. Tamoxifen for early breast cancer: an overview of the randomised trials. Early Breast Cancer Trialists' Collaborative Group. Lancet. 1998;351(9114):1451-67.
23. Elledge RM, Fuqua SAW. Estrogen and progesterone receptors. in: Harris J.R. Lippman M.E. Morrow M. Osborne Diseases of the breast. 2nd ed. Philadelphia: Lippincott Williams & Wilkins; 2000: 471.
24. Anders C, Carey LA. Understanding and treating triple-negative breast cancer. Oncology (Williston Park). 2008;22(11):1233-1243.
25. Cancer incidence in Jordan. 2015. https://www.moh.gov.jo/ Pages/viewpage.aspx?pageID=240
26. Abdel-Razeq H, Mansour A, Jaddan D. Breast Cancer Care in Jordan. JCO Glob Oncol. 2020;6:260-268.
27. https://www.moh.gov.jo/Echobusv3.0/SystemAssets/d05af2a1-008d- 48ee-8f20-c9c62fe190b9.pdf Ministry of Health: Epidemiology and survival analysis of three cancers (breast, colorectal & lung).
28. Howlader N, Noone AM, Krapcho M, et al (eds): SEER Cancer Statistics Review, 1975-2016. https://seer.cancer.gov/csr/1975_2016/
29. Goldhirsch A, Winer EP, Coates AS, Gelber RD, Piccart-Gebhart M, Thürlimann B, et al. Personalizing the treatment of women with early breast cancer: highlights of the St Gallen International Expert Consensus on the Primary Therapy of Early Breast Cancer 2013. Ann Oncol. 2013;24(9):2206-23.
30. Jacquemier J, Ginestier C, Rougemont J, Bardou VJ, Jauffret EC, Geneix J, et al. Protein expression profiling identifies subclasses of breast cancer and predicts prognosis. Cancer Res. 2005;65(3): 767-79.
31. Shomaf M, Masad J, Najjar S, Faydi D. Distribution of breast cancer subtypes among Jordanian women and correlation with histopathological grade: molecular subclassification study. JRSM Short Rep. 2013;4(10):2042533313490516.
32. Salhia B, Tapia C, Ishak EA, Gaber S, Berghuis B, Hussain KH, et al. Molecular subtype analysis determines the association of advanced breast cancer in Egypt with favorable biology. BMC Womens Health. 2011;11:44.
33. Ben Abdelkrim S, Trabelsi A, Missaoui N, Beizig N, Bdioui A, Anjorin A, et al. Distribution of molecular breast cancer subtypes among Tunisian women and correlation with histopathological parameters: Chuang E, Paul C, Flam A, McCarville K, Forst M, Shin S, et al. Breast cancer subtypes in Asian-Americans differ according to Asian ethnic group. J Immigr Minor Health. 2012;14(5):754-8.
35. Telfah A, Obeidat M, Swailmeen A, AlJadayeh M, Bawaneh A, Fayyad L. Breast Cancers in Young Women: A Retrospective Study at King Hussein Medical Center. JRMS. 2015;22(1):62-68.
36. Ihemelandu CU, Leffall LD Jr, Dewitty RL, Naab TJ, Mezghebe HM, Makambi KH, et al. Molecular breast cancer subtypes in premenopausal and postmenopausal African-American women: age-specific prevalence and survival. J Surg Res. 2007;143(1): 109-18.
37. Awadelkarim KD, Arizzi C, Elamin EO, Hamad HMA, De Blasio P, Mekki SO, et al. Basal-like phenotype in a breast carcinoma case series from Sudan: prevalence and clinical/pathological correlations. Patholog Res Int. 2011;2011:806831.
38. Akasbi Y, Bennis S, Abbass F, Znati K, Joutei KA, Amarti A, et al. Clinicopathological, therapeutic and prognostic features of the triplenegative tumors in moroccan breast cancer patients (experience of Hassan II university hospital in Fez). BMC Res Notes. 2011;4:500.
Full Text Sources:
Abstract:
Views: 7618

ABSTRACT
Introduction: Breast cancer is a heterogeneous disease that encompasses a wide range of pathological entities and clinical behaviors. It is classified into several subtypes based on their histopathological characteristics, tumor grade and biological features. This classification is important for studying breast cancer etiology, predicting clinical course, and making decisions related to breast cancer treatment. This study aims to identify biological characteristics of breast cancer among a group of Jordanian women who were referred to a breast clinic in a tertiary hospital in Amman.
Methodology: This is a retrospective cohort study that included initially a total of 119 female patients with breast cancer during the period between January 2018 and January 2020 at tertiary hospital in Jordan. Patients’ data were retrieved from the electronic health record system including their pathologic reports. Pathological reports were reviewed by the pathologist were histopathological features and tumor biological characteristics were confirmed. Breast cancer cases were classified into different biological subtypes based on surrogate definitions of intrinsic subtypes of breast cancer that was adopted in the 2011 St Gallen Consensus.
Results: A total of 117 patients were available for analysis with a mean age of 55.2 years and age range between 28 and 89 years. Patients were divided into group A (age 50 years; n=50) and group B (age > 50; n=67). The most common tumor was invasive ductal carcinoma (84.6%) followed by invasive lobular carcinoma. Metastatic axillary lymph node involvement was proved in 66.4% and 19 patients had de-novo metastasis. Biologically, the expression of ER, PR and HER2 were 84.1%, 84.1% and 36.7% respectively. Patients younger than 50 years had statistically higher levels of Ki67. Luminal B breast cancer was the most prevalent subtype in our patients.
Conclusion: Luminal B breast cancer is the most common prevalent intrinsic subtype of breast cancer among our patients. The expression of HER2/neu gene is considered high as well as large percentage of patients have high Ki67. Although it is not informative as genebased assays, IHC-based assays can be utilized for classification of breast cancer based on its biological characteristics at reasonable costs especially in low-middle income countries.
INTRODUCTION
Globally, Breast cancer is the most prevalent invasive cancer and the second leading cause of cancer deaths in women. It is heterogeneous disease with a wide range of pathological entities and clinical behaviors, thus posing great challenges in understanding the precise molecular pathogenesis (1). In Jordan, according to the Jordanian cancer registry, it is considered the most common malignancy and the third in cancer related mortality following lung and colorectal cancer.
Breast cancer is classified into different subtypes, this classification is essential for studying its etiology, predicting its clinical course, and making treatment decisions (2). The Distinctions between breast cancer subtypes is based on both patient and tumor characteristics (i.e. histology, grade, biology, genetic profile and stage) (3).
Invasive breast carcinomas either ductal or lobular subtypes represent the main infiltrating carcinomas, with lobular subtype being less frequent (4). The rely on histopathological features alone to classify invasive mammary carcinomas might result in diagnostic difficulty because of overlapping histopathologic features, particularly with invasive lobular carcinoma (ILC) variants and pleomorphic ILC (4,5). Proper histopathologic categorization of breast carcinomas has prognostic implications (6).
Generally, the progression of breast cancer has been viewed as a linear multi-step process, from hyperplasia, atypical hyperplasia, carcinoma in situ, and to invasive and metastatic carcinoma, supported by evidence from clinical, pathological, and genetic studies (7,8). Breast carcinogenesis is believed to be a series of stochastic genetic events that lead to distinct and divergent pathways towards invasive carcinoma (9). One of the Key pathological features of breast cancer is the nuclear grade that is associated with clinical outcome, and distinct genetic changes (10,11).
Reliance on estrogen receptor (ER-α), progesterone receptor (PR), and human epidermal growth factor receptor 2 (HER2/neu) expression to guide the clinical management and to predict clinical outcome and disease prognosis is far from satisfactory. Additional molecular markers are also required to predict clinical outcome and devise optimal individualized therapy (12, 13).
The expression of these markers in pure DCIS and DCIS/IDC provides better understanding of the precise relationships between pure DCIS and co-existing DCIS/IDC and to identify potential molecular markers for prognostic, therapeutic, and preventive purposes has been shown by Shackney et al (14). Based on mammography screening, DCIS component is identified in 30%-60% of breast IDC (19,20), while those with pure IDC might develop de novo without identifying underlying in situ carcinoma. However, patients with combined IDC-DCIS are assumed to develops IDC from a pre-existing DCIS lesion (15,16). In comparison to patients with pure IDC, patients with IDC-DCIS are more likely to be younger, having smaller or non-palpable tumors with lower probability of lymph node involvement (16-18). In addition, it was tested that the progression of in situ ductal carcinoma of breast to invasive ductal carcinoma is associated with alterations of ER, PGR and HER-2/neu protein expression (2).
The Expression patterns of estrogen receptor are well-established and defined for invasive breast carcinoma, but are not as well-defined for breast carcinoma in situ (CIS) (21). Over years it has been postulated that age-adjusted incidence trends differed for CIS and Invasive breast carcinoma in the United States, this changes possibly due to screening mammography, and/or etiologic diversity. Age-specific incidence patterns suggested that carcinogenic events starting early in reproductive life had greater impact upon CIS and invasive breast carcinoma defined by ER-negative expression than invasive breast carcinoma defined by ER-positive expression (21).
Estrogen receptor (ER) status whether positive or negative has been established as a predictive factor for breast cancer treatment and to a lesser extent, the significance of progesterone receptor (PR) status. Estrogens are important for stimulating growth of significant percentage of breast cancers and progesterone has roles in breast development and tumorigenesis. ER positive tumors are more responsive to hormonal adjuvant therapies than ER negative tumors, and have better prognosis for survival in comparison to ER negative tumors (22,23).
The expression of the c-erbB2 gene (HER-2/neu) proto-oncogene that is expressed in 10-34% of breast cancers and is associated with an aggressive behavior and poor clinical outcome (24), this can be in presence or absence of estrogen or progesterone in cancerous cells. Triple-negative breast cancers are lacking the expression of ER, PR and HER2/neu. In majority of cases, they carry the "basal-like” molecular phenotype that is identified using cDNA microarrays. Although most triple-negative breast tumors do cluster within the basal-like subgroup, there is up to 30% discordance between the two groups, most of the BRCA-1 associated breast cancer are classified within this category.
METHODOLOGY
This is a retrospective cohort study that included initially a total of 119 female patients who were diagnosed with breast cancer during the period between January, 2018 and January, 2020 at the breast clinic in a tertiary hospital in Amman, Jordan. Patients’ data were retrieved from the electronic health record system including their pathologic reports, which were reviewed by the pathologist to confirm both histopathological and biological characteristics of breast tumors.
All the cases were routinely evaluated immunohistochemically for estrogen and progesterone hormone receptor status and for HER2/neu gene expression using standard immunoperoxidase method. Evaluation of IHC staining was routinely done by the local pathologists according to international guidelines.
With regard to the hormone receptors, the results were expressed as positive or negative, whereas for HER2 expression, the results were reported according to American Society of Clinical Oncology (ASCO)/College of American Pathologists (CAP) guideline, updated on 2013. Breast cancers expressing ER and PR in >1% of neoplastic cells were considered as positive. For Her2/neu, tumors were scored as negative if the membrane staining was negative or weak and incomplete in less than or equal to 10% of tumor cells (score 0 or 1+), whereas cases with strong complete membrane staining in >10% of cells were scored as positive (score 3+). All cases with weak to moderate complete membrane screening observed in >10% of tumor cells were considered equivocal (score 2+) and were considered as negative or positive according to the results of FISH analysis.
 Patients were classified according on their age at diagnosis into 2 groups; group A (age 50 years or less) and group B (age more than 50 years). Analysis of patients' characteristics, tumor histology and tumor biology were performed. Breast cancer cases were classified into different biological subtypes based on surrogate definitions of intrinsic subtypes of breast cancer that was adopted in the 2011 St Gallen Consensus, table 1. Data were analyzed using the statistical package for the social sciences version 23 (SPSS Inc., Chicago, IL) statistical software. The analyses included descriptive statistics and chi-square test. A P < 0.05 was considered statistically significant.
Patients were classified according on their age at diagnosis into 2 groups; group A (age 50 years or less) and group B (age more than 50 years). Analysis of patients' characteristics, tumor histology and tumor biology were performed. Breast cancer cases were classified into different biological subtypes based on surrogate definitions of intrinsic subtypes of breast cancer that was adopted in the 2011 St Gallen Consensus, table 1. Data were analyzed using the statistical package for the social sciences version 23 (SPSS Inc., Chicago, IL) statistical software. The analyses included descriptive statistics and chi-square test. A P < 0.05 was considered statistically significant. RESULTS
A total of 119 women were included in the study. Two patients were excluded because of incomplete data. Patient’s age ranged between 28 and 89 years, the mean age of patients was 55.2 years with a standard deviation of 13.83 years. Patients were grouped into two groups, the first group "Group A" included cases aged 50 years or less (n=50, mean age 42.34 years ± 6.17), and the second group "Group B" included cases older than 50 years of age (n=69, mean age 64.49 ± 9.7).
 A total of 67 patients (56.3%) had unilateral left sided breast cancer, 48 patients (40.3%) had unilateral right sided breast cancer, and only 4 patients (3.4%) were diagnosed with bilateral breast cancer table 2. Following the study of 117 cases, 99 cases (84.6%) were invasive ductal carcinoma (IDC), 9 cases (7.7%) invasive lobular carcinoma (ILC), 8 patients (6.8%) ductal carcinoma in situ (DCIS), and 1 case (0.9%) was ductal carcinoma in situ with micro-invasion. Tumor classification based on histopathologic characteristics is illustrated in table 3.
A total of 67 patients (56.3%) had unilateral left sided breast cancer, 48 patients (40.3%) had unilateral right sided breast cancer, and only 4 patients (3.4%) were diagnosed with bilateral breast cancer table 2. Following the study of 117 cases, 99 cases (84.6%) were invasive ductal carcinoma (IDC), 9 cases (7.7%) invasive lobular carcinoma (ILC), 8 patients (6.8%) ductal carcinoma in situ (DCIS), and 1 case (0.9%) was ductal carcinoma in situ with micro-invasion. Tumor classification based on histopathologic characteristics is illustrated in table 3.  Regarding clinical tumor characteristics, the minimal tumor size for invasive tumors was 1.0 cm, the maximal size 10.0 cm with a mean size of 3.73 cm ± 1.82. In group A, the mean size was 3.69 cm ± 1.69, while in group B the mean size was 3.758 cm ± 1.9 with no statistical difference between them. Among the 117 patients, metastatic axillary lymph nodes were positive in 66.4%, in group A, 70% were positive while in group B, malignant axillary nodes were positive in 63.8% of the cases. Metastatic work-up was available for 112 patients (group A: 45 patients; group B: 67 patients). Among them, 19 patients (17%) had distant metastasis at time of diagnosis, 9 patients in group A and 10 patients in group B, (P=0.6). The biological and molecular features of 107 cases of invasive carcinomas were studied (i.e., IDC, ILC, DCIS with micro-invasion), of which 47 cases were in the age group 50 years or younger (group A) and 60 cases in the age group above 50 years (group B). Estrogen receptors (ER) were positive in 84.1% of the cases (n= 90), among patients in group A; 36 cases (76.6%) were positive and 11 cases (23.4%) were negative. In comparison, 90% of patients in group B were ER positive (n = 54), while 6 cases were negative (10%), (P = 0.068). Similar to ER, progesterone receptors were positive in 84.1% of the cases, 80.9% of patients in group A and 86.7% in group B were positive for PR (P = 0.43). The expression of human epidermal growth factor receptor 2 (HER2) was positive in 43 cases (36.7%), 20 cases in group A (42.6%) and 23 cases in group B (38.3%), (P = 0.69). The expression of cell proliferation biomarker, Ki-67 was studied in 89 cases of which, 27 cases (30.3%) had Ki67 ? 14%, 46.1% (41 cases) had Ki67 between 15%-50%, and 23.6% (21 cases) had Ki67 > 50%. Patients younger than 50 years have statistically higher levels of Ki67 (P = 0.0079). Based on the previous results we were able to classify the invasive carcinomas into different biological subtypes based on surrogate definitions of intrinsic subtypes of breast cancer that was adopted in the 2011 St Gallen Consensus, table 1. Out of all the 107 cases studied; 22 cases were Luminal A (20.6%), 71 cases were Luminal B (66.4%) of which 24 cases were HER2/neu positive. Only 7 cases were classified as nonluminal HER2/neu positive and 7 cases as triple negative breast cancer. Overall, 31 patients (28.9%) with invasive breast cancer were positive for HER2/neu. Analysis of cases based on age group is further illustrated in table 4.
Regarding clinical tumor characteristics, the minimal tumor size for invasive tumors was 1.0 cm, the maximal size 10.0 cm with a mean size of 3.73 cm ± 1.82. In group A, the mean size was 3.69 cm ± 1.69, while in group B the mean size was 3.758 cm ± 1.9 with no statistical difference between them. Among the 117 patients, metastatic axillary lymph nodes were positive in 66.4%, in group A, 70% were positive while in group B, malignant axillary nodes were positive in 63.8% of the cases. Metastatic work-up was available for 112 patients (group A: 45 patients; group B: 67 patients). Among them, 19 patients (17%) had distant metastasis at time of diagnosis, 9 patients in group A and 10 patients in group B, (P=0.6). The biological and molecular features of 107 cases of invasive carcinomas were studied (i.e., IDC, ILC, DCIS with micro-invasion), of which 47 cases were in the age group 50 years or younger (group A) and 60 cases in the age group above 50 years (group B). Estrogen receptors (ER) were positive in 84.1% of the cases (n= 90), among patients in group A; 36 cases (76.6%) were positive and 11 cases (23.4%) were negative. In comparison, 90% of patients in group B were ER positive (n = 54), while 6 cases were negative (10%), (P = 0.068). Similar to ER, progesterone receptors were positive in 84.1% of the cases, 80.9% of patients in group A and 86.7% in group B were positive for PR (P = 0.43). The expression of human epidermal growth factor receptor 2 (HER2) was positive in 43 cases (36.7%), 20 cases in group A (42.6%) and 23 cases in group B (38.3%), (P = 0.69). The expression of cell proliferation biomarker, Ki-67 was studied in 89 cases of which, 27 cases (30.3%) had Ki67 ? 14%, 46.1% (41 cases) had Ki67 between 15%-50%, and 23.6% (21 cases) had Ki67 > 50%. Patients younger than 50 years have statistically higher levels of Ki67 (P = 0.0079). Based on the previous results we were able to classify the invasive carcinomas into different biological subtypes based on surrogate definitions of intrinsic subtypes of breast cancer that was adopted in the 2011 St Gallen Consensus, table 1. Out of all the 107 cases studied; 22 cases were Luminal A (20.6%), 71 cases were Luminal B (66.4%) of which 24 cases were HER2/neu positive. Only 7 cases were classified as nonluminal HER2/neu positive and 7 cases as triple negative breast cancer. Overall, 31 patients (28.9%) with invasive breast cancer were positive for HER2/neu. Analysis of cases based on age group is further illustrated in table 4. 
DISCUSSION
In Jordan, according to the latest statistics from the Jordanian cancer registry, breast cancer is the most prevalent cancer and the 3rd leading cause of cancer related deaths following lung and colorectal cancer (25). In 2015, a total of 1138 cases of breast cancer have been diagnosed with a crude incidence of 34.1 per 100,000 and age standardized ratio of 45.7 per 100,000. These figures showed an increase by 69% during the past decade. Approximately, 44.7% of case were less than 50 years old at time of diagnosis (26). The median age at diagnosis among Jordanian women is 51 years, which is 10 years younger than women in western countries (27,28).
Breast cancer is classified into relevant subtypes, this classification is important for studying breast cancer etiology, predicting clinical course, and making decisions related to breast cancer treatment (2). The Distinctions between subtypes of breast cancer is made on the basis of patient characteristics or phenotypic or genotypic characteristics of the tumor itself, as tumor stage, grade, histology, and genetic profile (3). In Jordan, few reports have discussed biological characteristics of breast cancer and categorized them in a standardized manner to facilitate understanding trends of breast cancer biology in addition to its impact on treatment and prognosis.
In the perspective of the crucial importance of early detection and diagnosis of breast cancer for subsequent treatment and prognosis, we did this study aiming to identify the biological characteristics of breast cancer at the time of diagnosis in a group of Jordanian patients who were diagnosed from the breast clinic in a tertiary hospital in Jordan. We categorized breast cancer biology mainly into 4 main subtypes based on surrogate definitions of intrinsic subtypes of breast cancer that was adopted in the 2011 St Gallen Consensus (29). Although IHC-based assays do not provide as much biological insight into tumor biology as gene-based ones do, they allow classification of tumor at affordable costs and in the absence of fresh tissue specimens (30).
In Jordan, according to one study published on 2013 (31), breast cancer was classified into 4 main subtypes (luminal A, Luminal B, HER2 positive, and basal cell like).
In this study, Ki67 was not considered in distinguishing between luminal B and luminal A. In addition, luminal B was not categorized based on HER2 neu expression. According to it, the majority of all the cases were classified as luminal A (60%). This was higher than what was reported by Salhia et al in Egypt (44.3%) and by Ben Abdelkrim et al in Tunis (51.5%) (32, 33). Luminal A subtype was also predominant among Asian Americans (34). According to our study, only 20.56% of the cases were classified within this category.
In the previous study from Jordan, only 13% of all the cases were classified as luminal B subtype (31), which was similar to figures previously reported in North American and European women (6–19%), Tunisian women (16%) (33), but in contrast to what was found in Egyptian women (24.6%) (32). According to the figures in table 4, majority of our patients were classified as luminal B (66.35%), 22.4% of all cases were sub-classified into Luminal B/Her 2 positives, which is almost approximate to the results of Egyptian study and differ from the results obtained in Jordanian University seven years ago.
HER2/neu expression was positive in 36.7% of all cases, this was comparable to data published from the royal medical services on 2015 where overall expression rate was 36% (35). Only 7 cases (6.5%) were classified as non-luminal HER2/neu positive. The percentage of cases that were reported in Jordan previously classified as non-luminal HER2/neu+ was 12%, which was similar to that seen in Asian American (12%), Egyptian women (12%), and African American women (11.6%), but slightly lower than in Tunisian women (14.5%) (32, 33, 34, 36). Moreover, we figured out that HER2/neu+ subtype was more prominent in the younger age group (8.5%) in less than 50 years vs. 5% in cases more than 50 years, respectively. This might be indicative of the role of HER2+ mutation in the development of breast cancer at younger ages, which is similar to results that have concluded in other studies (31).
Triple negative breast cancer has higher incidence among young patients (10.6 %) in comparison to older women (3.3 %) with an overall rate of 6.5%. our results were lower than previously reported from Jordan were 15% of cases were also classified as BCL subtype similar to the incidence in Sudan (15.9%), which is slightly less than in Egyptian and Tunisian women (19% and 18%, respectively) (31,32,33,37). The reported incidence of BCL subtype in Moroccan breast cancer patients (12.6%) (38), 8.5% among American Chinese, 5.9% among American Filipino, 5.7% in American Japanese (34), and 21.2% in African American women (36).
CONCLUSION
Luminal B breast cancer is the most common prevalent intrinsic subtype of breast cancer among our patients. The expression of HER2/neu gene is considered high as well as large percentage of patients have high Ki67. These biological features might be associated with unfavorable prognosis and response to treatment if not properly evaluated. Although it is not informative as gene-based assays, IHC-based assays can be utilized for classification of breast cancer based on its biological characteristics at reasonable coasts especially in low-middle income countries. This classification is essential for clinical practice as an indicator of tumor behavior, prognosis, survival and response to therapy.
The lack of a unified reporting system for breast cancer in Jordan mandates the urgent adoption of a well-recognized classification similar to the one we described. This will help in understanding current situation of breast cancer biology and correlate possible changes in future with underlying genetic mutations or other risk factors.
Limitation of the study
This is a single center retrospective study from a breast clinic in a tertiary hospital in Jordan, so these results reflect tumor characteristics for patients who are diagnosed in this center. The need for multicenter studies is essential before we can generalize our results for Jordanian women.
Conflict of interest
No conflict of interest among authors
Funding
This research is not funded.
Ethical approval
This study is approved by the institutional review board at the Hashemite University and Prince Hamza Hospital.
REFERENCES
1. Tang P, Hajdu SI, Lyman GH. Ductal carcinoma in situ: a review of recent advances. Curr Opin Obstet Gynecol. 2007;19(1):63-7.
2. Hussein MR, Abd-Elwahed SR, Abdulwahed AR. Alterations of estrogen receptors, progesterone receptors and c-erbB2 oncogene protein expression in ductal carcinomas of the breast. Cell Biol Int. 2008;32(6):698-707.
3. Phipps AI, Li CI. Breast Cancer Biology and Clinical Characteristics. C.I. Li (ed.) Breast Cancer Epidemiology. New York: Springer; 2010.
4. Pinder SE, Elston CW, Ellis IO, editors; Elston CW, Ellis IO, editors. Invasive carcinoma: usual histological types. The Breast. 3rd ed. Edinburgh, Scotland: Churchill Livingstone; 1998. p. 283–337.
5. Wahed A, Connelly J, Reese T. E-cadherin expression in pleomorphic lobular carcinoma: an aid to differentiation from ductal carcinoma. Ann Diagn Pathol. 2002;6(6):349-51.
6. Ellis IO, Galea M, Broughton N, Locker A, Blamey RW, Elston CW. Pathological prognostic factors in breast cancer. II. Histological type. Relationship with survival in a large study with long-term follow-up. Histopathology. 1992;20(6):479-89.
7. Dupont WD, Page DL. Risk factors for breast cancer in women with proliferative breast disease. N Engl J Med. 1985;312(3):146-51.
8. Singletary SE. A working model for the time sequence of genetic changes in breast tumorigenesis. J Am Coll Surg. 2002;194(2): 202-16.
9. Buerger H, Otterbach F, Simon R, Schäfer KL, Poremba C, Diallo R, et al. Different genetic pathways in the evolution of invasive breast cancer are associated with distinct morphological subtypes. J Pathol. 1999;189(4):521-6.
10. Buerger H, Otterbach F, Simon R, Poremba C, Diallo R, Decker T, et al. Comparative genomic hybridization of ductal carcinoma in situ of the breast-evidence of multiple genetic pathways. J Pathol. 1999; 187(4):396-402.
11. Steinman S, Wang J, Bourne P, Yang Q, Tang P. Expression of cytokeratin markers, ER-alpha, PR, HER-2/neu, and EGFR in pure ductal carcinoma in situ (DCIS) and DCIS with co-existing invasive ductal carcinoma (IDC) of the breast. Ann Clin Lab Sci. 2007;37(2): 127-34.
12. Nofech-Mozes S, Spayne J, Rakovitch E, Hanna W. Prognostic and predictive molecular markers in DCIS: a review. Adv Anat Pathol. 2005;12(5):256-64.
13. Gasparini G, Longo R, Torino F, Morabito A. Therapy of breast cancer with molecular targeting agents. Ann Oncol. 2005;16 Suppl 4:iv28-36.
14. Shackney SE, Silverman JF. Molecular evolutionary patterns in breast cancer. Adv Anat Pathol. 2003;10(5):278-90.
15. Pinder SE, Ellis IO. The diagnosis and management of pre-invasive breast disease: ductal carcinoma in situ (DCIS) and atypical ductal hyperplasia (ADH)--current definitions and classification. Breast Cancer Res. 2003;5(5):254-257.
16. Wong H, Lau S, Yau T, Cheung P, Epstein RJ. Presence of an in situ component is associated with reduced biological aggressiveness of size-matched invasive breast cancer. Br J Cancer. 2010;102(9): 1391-1396.
17. Dieterich M, Hartwig F, Stubert J, Klöcking S, Kundt G, Stengel B, Reimer T, Gerber B. Accompanying DCIS in breast cancer patients with invasive ductal carcinoma is predictive of improved local recurrence-free survival. Breast. 2014;23(4):346-51.
18. Chagpar AB, McMasters KM, Sahoo S, Edwards MJ. Does ductal carcinoma in situ accompanying invasive carcinoma affect prognosis? Surgery. 2009;146(4):561–567. discussion 567–568
19. Jo BH, Chun YK. Heterogeneity of invasive ductal carcinoma: proposal for a hypothetical classification. J Korean Med Sci. 2006; 21(3):460-468.
20. Holland R, Connolly JL, Gelman R, Mravunac M, Hendriks JH, Verbeek AL, et al. The presence of an extensive intraductal component following a limited excision correlates with prominent residual disease in the remainder of the breast. J Clin Oncol. 1990; 8(1):113-8.
21. Anderson WF, Chu KC, Devesa SS. Distinct incidence patterns among in situ and invasive breast carcinomas,with possible etiologic implications. Breast Cancer Res Treat. 2004;88(2):149-59.
22. Tamoxifen for early breast cancer: an overview of the randomised trials. Early Breast Cancer Trialists' Collaborative Group. Lancet. 1998;351(9114):1451-67.
23. Elledge RM, Fuqua SAW. Estrogen and progesterone receptors. in: Harris J.R. Lippman M.E. Morrow M. Osborne Diseases of the breast. 2nd ed. Philadelphia: Lippincott Williams & Wilkins; 2000: 471.
24. Anders C, Carey LA. Understanding and treating triple-negative breast cancer. Oncology (Williston Park). 2008;22(11):1233-1243.
25. Cancer incidence in Jordan. 2015. https://www.moh.gov.jo/ Pages/viewpage.aspx?pageID=240
26. Abdel-Razeq H, Mansour A, Jaddan D. Breast Cancer Care in Jordan. JCO Glob Oncol. 2020;6:260-268.
27. https://www.moh.gov.jo/Echobusv3.0/SystemAssets/d05af2a1-008d- 48ee-8f20-c9c62fe190b9.pdf Ministry of Health: Epidemiology and survival analysis of three cancers (breast, colorectal & lung).
28. Howlader N, Noone AM, Krapcho M, et al (eds): SEER Cancer Statistics Review, 1975-2016. https://seer.cancer.gov/csr/1975_2016/
29. Goldhirsch A, Winer EP, Coates AS, Gelber RD, Piccart-Gebhart M, Thürlimann B, et al. Personalizing the treatment of women with early breast cancer: highlights of the St Gallen International Expert Consensus on the Primary Therapy of Early Breast Cancer 2013. Ann Oncol. 2013;24(9):2206-23.
30. Jacquemier J, Ginestier C, Rougemont J, Bardou VJ, Jauffret EC, Geneix J, et al. Protein expression profiling identifies subclasses of breast cancer and predicts prognosis. Cancer Res. 2005;65(3): 767-79.
31. Shomaf M, Masad J, Najjar S, Faydi D. Distribution of breast cancer subtypes among Jordanian women and correlation with histopathological grade: molecular subclassification study. JRSM Short Rep. 2013;4(10):2042533313490516.
32. Salhia B, Tapia C, Ishak EA, Gaber S, Berghuis B, Hussain KH, et al. Molecular subtype analysis determines the association of advanced breast cancer in Egypt with favorable biology. BMC Womens Health. 2011;11:44.
33. Ben Abdelkrim S, Trabelsi A, Missaoui N, Beizig N, Bdioui A, Anjorin A, et al. Distribution of molecular breast cancer subtypes among Tunisian women and correlation with histopathological parameters: Chuang E, Paul C, Flam A, McCarville K, Forst M, Shin S, et al. Breast cancer subtypes in Asian-Americans differ according to Asian ethnic group. J Immigr Minor Health. 2012;14(5):754-8.
35. Telfah A, Obeidat M, Swailmeen A, AlJadayeh M, Bawaneh A, Fayyad L. Breast Cancers in Young Women: A Retrospective Study at King Hussein Medical Center. JRMS. 2015;22(1):62-68.
36. Ihemelandu CU, Leffall LD Jr, Dewitty RL, Naab TJ, Mezghebe HM, Makambi KH, et al. Molecular breast cancer subtypes in premenopausal and postmenopausal African-American women: age-specific prevalence and survival. J Surg Res. 2007;143(1): 109-18.
37. Awadelkarim KD, Arizzi C, Elamin EO, Hamad HMA, De Blasio P, Mekki SO, et al. Basal-like phenotype in a breast carcinoma case series from Sudan: prevalence and clinical/pathological correlations. Patholog Res Int. 2011;2011:806831.
38. Akasbi Y, Bennis S, Abbass F, Znati K, Joutei KA, Amarti A, et al. Clinicopathological, therapeutic and prognostic features of the triplenegative tumors in moroccan breast cancer patients (experience of Hassan II university hospital in Fez). BMC Res Notes. 2011;4:500.
Full Text Sources:
Abstract:
Views: 7618

Watch Video Articles
For Authors
Journal Subscriptions

Current Issue
Dec 2025
Supplements
Instructions for authors
Online submission
Contact
ISSN: 2559 - 723X (print)
e-ISSN: 2601 - 1700 (online)
ISSN-L: 2559 - 723X
Journal Abbreviation: Surg. Gastroenterol. Oncol.
Surgery, Gastroenterology and Oncology (SGO) is indexed in:
e-ISSN: 2601 - 1700 (online)
ISSN-L: 2559 - 723X
Journal Abbreviation: Surg. Gastroenterol. Oncol.
Surgery, Gastroenterology and Oncology (SGO) is indexed in:
- SCOPUS
- EBSCO
- DOI/Crossref
- Google Scholar
- SCImago
- Harvard Library
- Open Academic Journals Index (OAJI)
Open Access Statement
Surgery, Gastroenterology and Oncology (SGO) is an open-access, peer-reviewed online journal published by Celsius Publishing House. The journal allows readers to read, download, copy, distribute, print, search, or link to the full text of its articles.
Surgery, Gastroenterology and Oncology (SGO) is an open-access, peer-reviewed online journal published by Celsius Publishing House. The journal allows readers to read, download, copy, distribute, print, search, or link to the full text of its articles.
Journal Metrics
Time to first editorial decision: 25 days
Rejection rate: 61%
CiteScore: 0.2
Time to first editorial decision: 25 days
Rejection rate: 61%
CiteScore: 0.2

Meetings and Courses in 2025
Meetings and Courses in 2024
Meetings and Courses in 2023
Meetings and Courses in 2022
Meetings and Courses in 2021
Meetings and Courses in 2020
Meetings and Courses in 2019
Verona expert meeting 2019

Surgery, Gastroenterology and Oncology applies the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits readers to copy and redistribute the material in any medium or format, remix, adapt, build upon the published works non-commercially, and license the derivative works on different terms, provided the original material is properly cited and the use is non-commercial. Please see: https://creativecommons.org/licenses/by-nc/4.0/
Publisher’s Note:
The opinions, statements, and data contained in article are solely those of the authors and not of Surgery, Gastroenterology and Oncology journal or the editors. Publisher and the editors disclaim responsibility for any damage resulting from any ideas, instructions, methods, or products referred to in the content.
 IASGO Society News
IASGO Society News