Surgery, Gastroenterology and Oncology

|
|
Background: Gastrointestinal stromal tumours (GISTs) are uncommon sarcomas. Surgery and target therapy are the primary modalities in treating GIST. KIT expression is the cornerstone in diagnosis of GIST, yet its expression is absent in a small percent of GISTs. The precise behaviour and treatment effects, particularly target therapy, and the prognosis of these cases are unclear; hence this study was designed to investigate them.
Methods: In the current retrospective cohort research, all patients with GIST who presented and were treated at National Cancer Institute (NCI), Cairo University, between January 2011 to December 2020 were included. The cases were categorized into two groups (positive and negative) based on c-KIT status positivity. Both groups' patient characteristics, clinical and histopathological characteristics of tumours, and treatment modalities outcomes were compared.
Results: The stomach was the site of origin of 56 cases (48.3%) with c-KIT positive compared to only 5 cases (22.7%) without c-KIT expression (P-value= 0.009). The liver was the primary origin of GISTs in only one case (0.86%) in the c-KIT positive group, while it was the primary site in 6 cases (27.2%) in the c-KIT negative group (P-value= 0.002). The median overall survival in the c-KIT positive group was 60 months compared to the c-KIT negative group, which was only 30 months (P-value = 0.004). Regarding c-KIT negative cases, the five years OS of the 9 cases who received adjuvant target therapy was higher than the other 13 cases that did not (55.5 vs. 15.3%) (P-value = 0.05).
Conclusion: GISTs lacking c-KIT expression have different behaviour than those with the c-KIT expression. In terms of OS treatment results and prognosis, c-KIT negative GISTs demonstrated poor outcomes compared to c-KIT positive cases.
INTRODUCTION
Mesenchymal tumors rarely arise from GIT, with GIST accounting for 18% of all sarcomas (1). Despite this, GISTs arise from any site of GIT, but gastric GISTs represent at least 50% of all GISTs. The prognosis for GISTs has historically been considered to be dismal as the median survival for non-metastatic cases was only 60 months, while only 19 months in cases with metastasis, as reported by DeMatteo, et al. (2). Over the past 20 years, significant advancements in the treatment of GISTs have been made with the introduction of Target therapy resulting in increased survival rates. In 1998, there was a breakthrough in the detection and management of GISTs as Hirota, et al. described some important characteristics that would aid in the way we identify and treat patients with GIT mesenchymal tumors. Initially, fifty-eight mesenchymal tumors of the GIT were tested by immunohisto-chemistry to have KIT (CD 117) expression. In contrast to leiomyomas and schwannomas, which did not express KIT, 78% of GISTs were shown to be positive for KIT and CD34 (the known marker for GIST at that time) (3).
In addition, KIT is strongly expressed in approximately 95% of all GIST. In GIST, KIT expression is rarely absent. In these cases, confirming the diagnosis is done by other IHCs. To our knowledge, few studies have been conducted on GIST, which lacks c-KIT expression and is less helpful in assessing these tumors' behavior and prognosis. Therefore, this study was designed to compare cases of GIST with c-KIT expression versus those without, aiming to identify differences in clinical, prognosis, and treatment results.
PATIENT AND METHODS
Medical records and data
From January 2011 to December 2020, all GIST cases presented and treated at Cairo University's National Cancer Institute (NCI) were included in this retrospective cohort research. The patients' information was obtained from the medical records of the Biostatistics and Cancer Epidemiology Department of NCI. The cases were divided according to c-KIT status positivity into two groups (positive and negative). Both groups' patient characteristics, clinical and histopathological characteristics of tumours, treatment modalities, and outcomes were compared.
The definitive diagnosis of GIST obtained by histo-pathological examination of either surgically resected specimen or biopsy from suspected lesion. Starting with microscopic examination of hematoxylin and eosin (H & E) stained slides to evaluate morphological and microscopic characteristics concomitant with GIST, and after correlation with the clinical, radiological findings and the site of lesion, immune-phenotyping was done starting with CD117 (c-KIT) and CD34. In cases with c-KIT negative, Dog1 was done. Also exclusion of other spindle cell tumours like smooth muscle tumours (by negativity to Desmin and Caldesmon) was done.
Statistical methods
Utilizing version 24 of the Statistical Package for Social Sciences (SPSS), data management and statistical analysis were carried out. Means and standard deviations, or medians and ranges were used to summarise numerical data. Percentages were used to summarise categorical data. The t-test was used to compare the two groups in terms of normally distributed numerical variables. The Mann-Whitney test was used to compare numeric variables that were not normally distributed. When appropriate, the chi-square and Fisher's exact test were used to analyse the differences for categorical variables. The Kaplan and Meier procedure was used to estimate the OS and progression-free rates, and the Log-rank test compared the different prognostic factors. The OS rates were computed from the diagnosis date to the date of death from any cause. Patients who were still living or who were lost to follow-up were censored on the last known alive date. Then significant variables were entered into the Cox Proportional Hazards Model. All p-values are two-sided. P-values ≤0.05 were considered significant.
RESULTS
138 patients with GISTs were diagnosed and treated at the NCI, Cairo University, between January 2011 and December 2020.The main presenting symptoms were abdominal distention (58 cases [42%]), Abdominal pain (35 cases [25.3%]).
One hundred and sixteen cases (84.1%) were positive for c- KIT expression, while only 22 cases (15.9%) were negative. Regarding age and gender, there were no significant differences between the two groups, as illustrated in Table 1. In contrast to cases with c-KIT positive, which mostly originated from the stomach (48%), c-KIT negative cases mostly originated from extragastrointestinal sites, mainly as pelvic or abdominal masses (31.8%) without direct continuity with the gastrointestinal tract or primarily originated from the liver (27.2%) without any apparent masses in GIT despite the careful investigation by endoscopy and imaging as depicted in table 1.
Table 1 - Patients' and tumors' characteristics
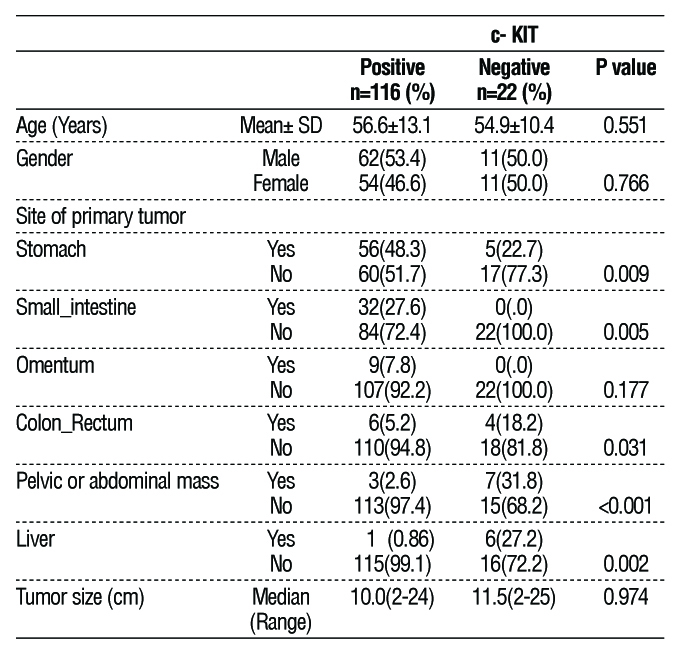
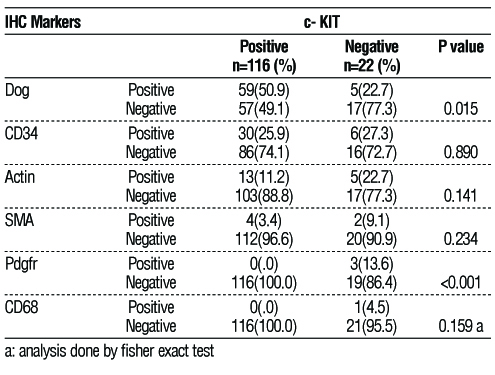
Other immuno-histochemistry’s tests were used for confirmation of diagnosis, as illustrated in
table 2. Dog1 was less expressed in the c-KIT negative (only 5 cases [22.7%]) compared to 59 (50.9%) cases in the c-KIT-positive group (P-value = 0.015).
Table 2 - Immunohistochemistry markers
Surgical resection was done for 96 (82.8%) cases in the c-KIT positive group, while only 15 (68.2%) in the c-KIT negative group (table 3).
Survival analysis
According to c-KIT expression, the median OS for c-KIT positive was 60 months, whereas it was only 30 months for c-KIT negative cases (P-value =0.004) (fig. 1). With respect to c-KIT negative cases, the five years of OS of the 9 cases who received adjuvant target therapy was higher than the 13 cases who did not receive it (55.5 vs. 15.3%) (P-value 0.05). There was no significant difference in DFS between the two groups (fig. 2).
Figure 1 - The Cumulative (Cum) survival for cases with c-KIT positive and c-KIT negative GIST (P-value =0.004)
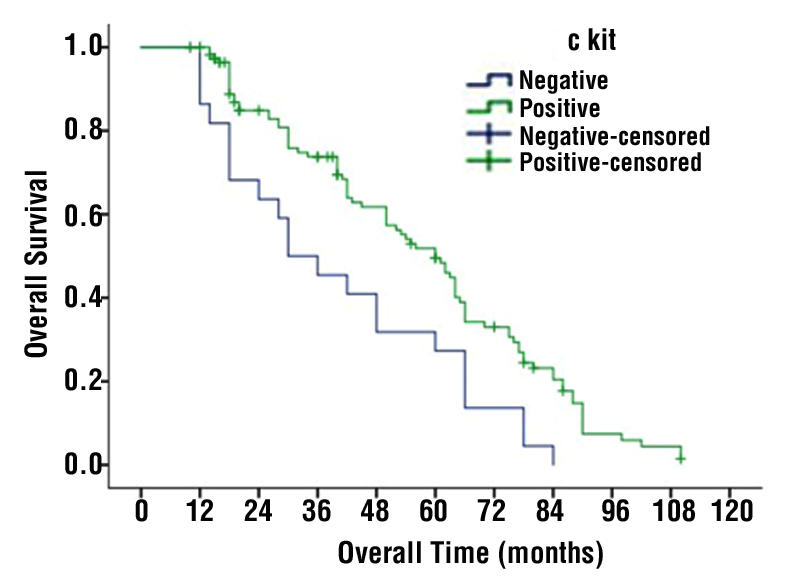
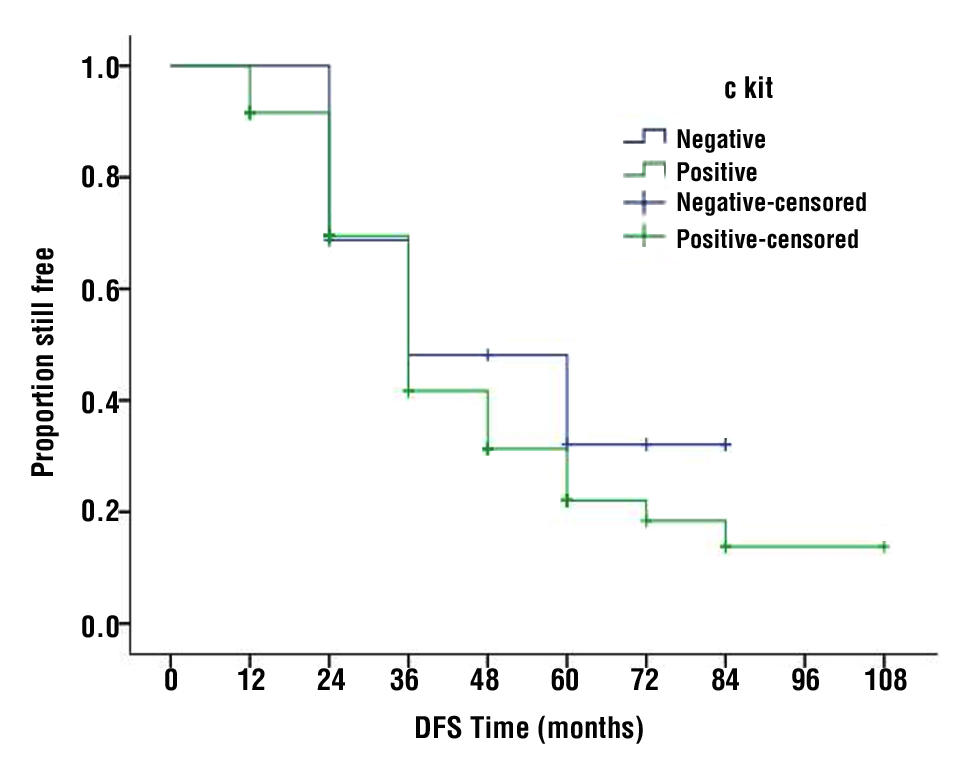
Figure 2 - Disease free survival (DFS) for cases with c-KIT positive and c-KIT negative GIST
DISCUSSION
Gastrointestinal sarcomas are rare diseases, and the discovery of the c-KIT gene mutation markedly improved their diagnosis and classification, as it is not expressed in the diffuse matter in sarcomas other than GISTs. Approximately 95% of GISTs express c-KIT; however, to our knowledge, few studies have investigated the behavior, treatment effects, and prognosis of GISTs devoid of KIT expression. The analysis identified only 22 cases of c-KIT negative GIST out of 138 cases enrolled in our study. Regarding the age of patients in the conducting study, no significant difference was found between c-KIT positive and c-KIT negative GIST, with a mean age of 56.6 and 54.9 years, respectively. These results are comparable to Kontogianni-Katsarou, et al study on KIT negative GISTs as the mean of age was 62 ± 14.5 years (4). No pediatric cases were present in NCI registries which represents a limitation in this study as Pappo, et al. reported that 85% of paediatric GIST were negative for c-KIT and PDGFRA expression (5). In the current study, no gender predilection was found in the c-KIT negative group, while the incidence of c-KIT positive GIST was relatively higher in males (53.4 %: 46.6 %). These findings are consistent with those in the previous studies, which discussed the incidence of GIST in both c-KIT positive & negative (4,6).
In the current study, c-KIT negative GISTs originated primarily from extragastrointestinal sites (pelvic, abdominal mass of unspecified origin (31.8%) and in the liver (27.2%), whereas c-KIT positive GISTs originated mainly from the stomach (48.3%). The same results were reported by Yamamoto, et al. on 10 cases of c-KIT negative GIST. Five patients had tumors in the omentum, two in the mesentery, one in the retro-peritoneum, one in the pelvic cavity, and one in the abdominal cavity (7).
According to Yamamoto H, et al., Extragastrointestinal stromal tumors (EGIST) are GIST that develop outside of the gastrointestinal wall, such as in the omentum, retroperitoneum, pelvic cavity, or mesentery (8). Even though the exact mechanism behind the development of these tumors is unknown, some GISTs may extend outward, eventually they completely lose their primary link to the GIT tract and attach to nearby soft tissue. Additionally, they may represent metastatic deposits from GIST, but the primary tumor is not discovered (8). GIST is thought to develop from interstitial cells of Cajal (ICCs), many organs other than GIT harboring interstitial cells named interstitial Cajal-like cells (ICLCs) like upper and lower urinary tract, pancreas, and other sites. In addition, ICLCs have been found in liver fibrosis, as reported in Fu et al. study (9); this may explain the primary hepatic GIST. In this study, identified 6 cases of primary c-KIT-negative GIST originating from the liver that did not exhibit imaging or endoscopic evidence of metastasis from any site of GIT.
Immunohistochemistry is the cornerstone of the GIST diagnosis. In the current study, the cases of c-KIT negative GIST were positive for CD 34: 27.3 %, Actin: 22.7 %, Dog1: 22.7 %, PDGFR: 13.6%. According to West RB, et al., DOG1 was positive in 98% of c-KIT negative cases (10), whereas Kang et al. demonstrated that the expression of DOG1 was positive in (92%) of c-KIT negative GIST (11), respectively.
One of the obstacles in treating c-KIT negative GISTs is their response to target therapy, as it is necessary as a neoadjuvant to manage some of these cases to downsize the tumor and make them amenable for resection as well as an adjuvant in tumors with advanced stage. Imatinib mesylate works as an inhibitor of tyrosine kinases, such as KIT and PDGFR (12-19). The effect of Imatinib on c-KIT negative GIST is not well known. In our study, all cases of c-KIT positive received target therapy (Imatinib) varied between neoadjuvant & adjuvant and definitive treatment, while only 9 cases with c-KIT negative GIST received Imatinib as an adjuvant treatment. IHC revealed that 3 of 9 cases treated with Imatinib were PDGFR-positive.
In the current study, the effect of Imatinib as an adjuvant treatment in KIT negative can be distinguished from the higher OS of the cases who received it as an adjuvant compared to the others who did not receive it. These findings are consistent with Medeiros et al. study (20), and since some KIT-negative GISTs have imatinib-sensitive PDGFRA mutations, Imatinib therapy shouldn't be denied from KIT-negative GIST patients. In the current study, patients with c-KIT positive GIST showed higher OS with a median of 60 months compared with the c-KIT negative group, which had a median of 30 months. This finding may indicate a difference in tumor behavior or due to omitting or resistance of
c-KIT negative cases to target therapy, emphasizing the need for additional research to determine the effects of target therapy on c-KIT negative GISTs. The conducted study emphasized the characteristics of c-KIT negative GIST, but it had limitations due to its retrospective methodology and the limited number of c-KIT negative GIST cases. However, the study's findings were valuable because they demonstrated that c-KIT negative GISTs behave differently from c-KIT positive GISTs from the site of origin and having lower overall survival rates. Also that some cases of c-KIT negative GISTs may respond well to target therapy.
Furthermore, the authors recommend conducting studies on c-KIT negative GISTs as a different category from c-KIT positive, which may need closer follow-up and more aggressive surgical resection as this is the only effective treatment that still aims to improve survival.
CONCLUSION
GISTs lacking c-KIT expression have different behavior than those with the c-KIT expression. A more significant percentage of cases originated from extra-intestinal sites, with a higher percentage originating primarily in the liver. In terms of OS treatment results and prognosis, c-KIT negative GISTs demonstrated poor outcomes compared to c-KIT positive cases.
Conflict of interest
All authors declare no conflict of interest.
Ethical approval
This study is a retrospective analysis of the medical records of 138 GIST patients. All measures were taken to protect the security and confidentiality of the individual patient's information. The approval of the NCI ethical committee was secured through the expedited pathway. IRB approval No: 2106-41004-004.
REFERENCES
1. Ducimetiere F, Lurkin A, Ranchere-Vince D, Decouvelaere AV, Peoc'h M, Istier L, et al. Incidence of sarcoma histotypes and molecular subtypes in a prospective epidemiological study with central pathology review and molecular testing. PLoS One. 2011; 6(8):e20294.
2. DeMatteo RP, Lewis JJ, Leung D, Mudan SS, Woodruff JM, Brennan MF. Two hundred gastrointestinal stromal tumors: recurrence patterns and prognostic factors for survival. Ann Surg. 2000; 231(1):51–8.
3. Hirota S, Isozaki K, Moriyama Y, Hashimoto K, Nishida T, Ishiguro S, et al. Gain-of-function mutations of c-kit in human gastrointestinal stromal tumors. Science. 1998; 279(5350):577–80.
4. Kontogianni-Katsarou K, Lariou C, Tsompanaki E, Vourlakou C, Kairi-Vassilatou E, Mastoris C, et al. KIT-negative gastrointestinal stromal tumors with a long term follow-up: a new subgroup does exist. World J Gastroenterol. 2007; 13(7):1098-102.
5. Pappo AS, Janeway KA. Pediatric gastrointestinal stromal tumors. Hematol Oncol Clin North Am. 2009; 23:15–34, vii.
6. Nilsson B, Bumming P, Meis-Kindblom JM, Odén A, Dortok A, Gustavsson B, et al. Gastrointestinal stromal tumors: the incidence, prevalence, clinical course, and prognostication in the preimatinib mesylate era–a population- based study in western Sweden. Cancer. 2005; 103(4):821–9.
7. Yamamoto H, Kojima A, Nagata S, Tomita Y, Takahashi S, Oda Y. KIT-negative gastrointestinal stromal tumor of the abdominal soft tissue: a clinicopathologic and genetic study of 10 cases. Am J Surg Pathol. 2011; 35(9):1287-95
8. Yamamoto H, Oda Y, Kawaguchi K, Nakamura N, Takahira T, Tamiya S, et al. c-kit and PDGFRA mutations in extragastrointestinal stromal tumor (gastrointestinal stromal tumor of the soft tissue). Am J Surg Pathol. 2004; 28:479–88.
9. Fu S, Wang F, Cao Y, Huang Q, Xiao J, Yang C, et al. Telocytes in human liver fibrosis. J Cell Mol Med. 2015; 19:676–683.
10. West RB, Espinosa I, Lee CH, Kim MK, Rouse BT, Subramanian S, et al. A novel monoclonal antibody against DOG1 is a sensitive and specific marker for gastrointestinal stromal tumors. Am J Surg Pathol. 2008; 32(2):210-8.
11. Kang GH, Srivastava A, Kim YE, Park HJ, Park CK, Sohn TS, et al. DOG1 and PKC-q are useful in the diagnosis of KIT-negative gastrointestinal stromal tumors. Mod Pathol. 2011; 24(6):866-75.
12. Joensuu H, Roberts PJ, Sarlomo-Rikala M, Andersson L C, Tervahartiala P, Tuveson D, et al. Effect of the tyrosine kinase inhibitor STI571 in a patient with a metastatic gastrointestinal stromal tumor. N Engl J Med. 2001; 344:1052–6.
13. Kelly CM, Gutierrez Sainz L, Chi P. The management of metastatic GIST: current standard and investigational therapeutics. J Hematol Oncol. 2021;14(1):1-2.
14. Mehren MV, Joensuu H. Gastrointestinal Stromal Tumors. J Clin Oncol. 2018;36(2):136-143.
15. Waller CF. Imatinib Mesylate. Recent Results Cancer Res. 2018; 212:1-27.
16. Reichardt P. The Story of Imatinib in GIST-a Journey through the Development of a Targeted Therapy. Oncol Res Treat. 2018;41(7-8):472-7.
17. Klug LR, Khosroyani HM, Kent JD, Heinrich MC. New treatment strategies for advanced-stage gastrointestinal stromal tumours. Nat Rev Clin Oncol. 2022;19(5):328-41.
18. Al-Share B, Alloghbi A, Al Hallak MN, Uddin H, Azmi A, Mohammad RM, et al. Gastrointestinal stromal tumor: a review of current and emerging therapies. Cancer Metastasis Rev. 2021;40(2):625-41.
19. Bauer S, George S, Von Mehren M, Heinrich MC. Early and next-generation KIT/PDGFRA kinase inhibitors and the future of treatment for advanced gastrointestinal stromal tumor. Front Oncol. 2021; 11:672500.
20. Medeiros F, Corless CL, Duensing A, Hornick JL, Oliveira AM, Heinrich MC, et al. KIT-negative gastrointestinal stromal tumors: proof of concept and therapeutic implications. Am J Surg Pathol. 2004;28(7):889-94.
Full Text Sources:
Abstract:
Views: 2039

For Authors
Journal Subscriptions

Dec 2025
Supplements
Instructions for authors
Online submission
Contact
e-ISSN: 2601 - 1700 (online)
ISSN-L: 2559 - 723X
Journal Abbreviation: Surg. Gastroenterol. Oncol.
Surgery, Gastroenterology and Oncology (SGO) is indexed in:
- SCOPUS
- EBSCO
- DOI/Crossref
- Google Scholar
- SCImago
- Harvard Library
- Open Academic Journals Index (OAJI)
Surgery, Gastroenterology and Oncology (SGO) is an open-access, peer-reviewed online journal published by Celsius Publishing House. The journal allows readers to read, download, copy, distribute, print, search, or link to the full text of its articles.
Time to first editorial decision: 25 days
Rejection rate: 61%
CiteScore: 0.2

Meetings and Courses in 2025
Meetings and Courses in 2024
Meetings and Courses in 2023
Meetings and Courses in 2022
Meetings and Courses in 2021
Meetings and Courses in 2020
Meetings and Courses in 2019
Verona expert meeting 2019

Surgery, Gastroenterology and Oncology applies the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits readers to copy and redistribute the material in any medium or format, remix, adapt, build upon the published works non-commercially, and license the derivative works on different terms, provided the original material is properly cited and the use is non-commercial. Please see: https://creativecommons.org/licenses/by-nc/4.0/
Publisher’s Note:
The opinions, statements, and data contained in article are solely those of the authors and not of Surgery, Gastroenterology and Oncology journal or the editors. Publisher and the editors disclaim responsibility for any damage resulting from any ideas, instructions, methods, or products referred to in the content.
 IASGO Society News
IASGO Society News