Surgery, Gastroenterology and Oncology

|
|
Background: MicroRNAs (miRNAs) are stable short non-coding RNAs that have an impact on oncogenic and tumour-suppressive signalling pathways, highlighting their utility as non-invasive biomarkers in bladder cancer (BC). Because of their possible diagnostic value, along with the absence of similar works in Iraqi samples, we evaluated here miR-129 (a well-documented tumour suppressor extensively shown as downregulated in urothelial carcinoma) and miR-210 (a hypoxia-induced miRNA implicated in hypervascularization and metabolic reprogramming).
Methods: This was a case-control hospital-based study of 30 male patients with BC (cases) and 30 healthy control persons who were age-matched, ranging from 50 to 80 years old, attending the Oncology Center at Al-Haboubi General Hospital, Al-Nassiriyah, Iraq. QRT-PCR measured the level of circulating miRNAs in the serum after normalizing to cel-miR-39.
Results: A similar reduction was also observed for median relative expression of miR-129 in the patients (0.79; IQR 0.52–1.67) compared to the controls (1.63; IQR 0.88–2.34), and this is mirrored in those concerning miR-210 as well (0.86 vs 1.47, IQRs: 0.58–1.73 and 0.72–2.15). There were no significant differences in the Mann–Whitney U test (p = 0.07 and 0.11, respectively); however, subgroup analysis results for smoking status and age indicated that these were significantly different (p < 0.05).
Conclusions: While the sample size was small, these findings need to be validated in larger stage-stratified cohorts as region-specific non-invasive biomarkers for early detection of BC in Iraq.
INTRODUCTION
Bladder cancer is one of the most frequent urological malignancies worldwide and has high recurrence rates, as well as significant morbidity, particularly when detected at an advanced stage (1-3). Easy access to early diagnostic centres for patients in Iraq is scarce and inadequate, with a low rate of bowel cancer screening; detection is often late, leading to worse outcomes. The existing clinical diagnosis approaches to BC, such as cystoscopy or urine cytology, are invasive, costly, and have low sensitivity, especially for low-grade tumours and pathologically or early-stage diseases (4-5). Even though the sensitivity of endoscopic cystoscopy exceeds that of conventional urine cytology, it is an imperfect surveillance method due to its invasiveness and poor yield in detecting flat or molecularly active lesions (6). These constraints highlight the urgent requirement for non-invasive and accurate biomarkers to achieve the objectives of early diagnosis, risk assessment, and individualised treatment, particularly in resource-deficient settings.
MicroRNAs (miRNAs) have emerged as promising candidates in filling this gap. These small, non-protein-coding RNA molecules (~22 nucleotides) regulate post-transcriptional gene expression and are very stable in biofluids like serum or urine, making them ideal candidates for the development of liquid biopsies (7). Increased evidence indicates that increased or reduced activity of individual miRNAs plays a role in the pathogenesis of cancer, and some miRNAs act as tumour suppressors and others as oncogenes, depending on the cell type or disease state.
Among these, miR-129 has been reported to be a classical tumour suppressor in BC. It is widely suppressed in BC tumour tissue and in circulating biofluids. At the same time, the overexpression of MEG3 exerts potent anti-tumour effects through targeting several key oncogenes (such as SOX4 and CDK6), followed by the induction of G1 cell cycle arrest and suppression of EMT by down-regulating Wnt/?-catenin signalling (8-9). Preclinical studies have demonstrated that restoration of miR-129 expression leads to the reduction of tumour aggressiveness and is therefore a potential therapeutic target as well as a diagnostic tool (10).
In contrast, miR-210 is a hypoxia-regulated microRNA (miRNA) upregulated by HIF-1? under low oxygen tension during the development of solid tumours. It confers benefits in terms of angiogenesis, metabolic repatterning, and cell survival through the regulation of EFNA3, ISCU1/2, and SDHD (11). Despite that, the expression of miR-210 is up-regulated in many late stages of cancers, and its expression decreased in early stages of BC; among Middle Eastern studies, as with our work, it was even differentially regulated depending on the tumour stage or aetiology for regional geo-environmental reasons.
There is no data about miR-129 and miR-210 expression status in the Iraqi community. The distinctive environmental features of AL-Nassiriya in southern Iraq, with a high smoking proportion and potential exposure to industrial pollutants, could be reflected in different molecular properties of BC. However, limited knowledge has largely led to the gap in applying internationally validated biomarker models in regional clinical practice.
Our evidence for this matter was the contrast in serum expression levels of miR-129 and miR-210 between thirty BC male patients who were diagnosed histopathologically, in addition to 30 healthy controls having a similar age group who had been recruited from the Oncology Center with a demographic linkage known. This study updates the knowledge about miRNA biology in BlCa and can also help to develop a regionally focused, non-invasive molecular diagnostic tool that reflects Iraq's epidemiology. We anticipate that the discovery of molecular signature-specific local risk factors will lead to improved strategies for early detection of cancer and management of patients, and also be an important resource for the development of other therapeutic approaches in this understudied group.
MicroRNA Biogenesis
The process of microRNA (miRNA) biogenesis constitutes yet another layer in the intricately controlled system of gene expression regulation. It initiates within the nucleus, where the RNA polymerase II transcription machinery creates the primary miRNA transcript (pri-miRNA). The endonuclease Drosha processes this primary transcript, which further modifies it into a precursor miRNA (pre-miRNA) featuring a distinctive hairpin shape. The pre-miRNAs are transported to the cytosol, where an enzyme called Dicer further shapes them into fully developed mature miRNAs. The mature miRNAs are bound to Argonaute proteins at this stage, forming RNA-induced silencing complexes (RISCs). Within RISCs, miRNAs command the post-transcriptional control of gene expression by binding to specific sequences within the mRNA, leading to their decay or halting the translation. This control mechanism functions in a wide range of fundamental processes, including, but not limited to, development, differentiation, and disease progression.
microRNA-129
MiR-129 is a crucial mediator in BC pathology as it regulates gene expression as a non-coding RNA, a small molecule. MiR-129 acts as a tumor suppressor and protects against excessive cell proliferation, apoptosis, metastasis, and other oncogenic pathways (12). Its downregulation has been very common in the tissues of patients with BC in advanced stages of the disease and poor prognosis(13). By targeting SOX4 and CDK6, miR-129 increases the G1 phase of the cell cycle and diminishes the invasiveness of cancer cells (14). Also, the aberrant Wnt/?-catenin signaling pathway in BC is inhibited by miR-129, thus reducing tumor growth and epithelial-mesenchymal transition (EMT) (15). Restoring the expression of miR-129 in BC cell lines decreased the aggressiveness of the tumors, which shows that therapeutic targeting of the oncogene provides a rationale for treatment (16). Additionally, by blocking the action of the anti-apoptotic BCL2, miR-129 demonstrates why apoptosis should not be considered absolute, speaking to the diverse roles of proteins in cancers (17). The data support the hypothesis that miR-129 can be considered a potential diagnostic marker and a therapeutic target for BC, which calls for more in-depth exploration.
microRNA-210
MiR-210 is an important hypoxia-responsive microRNA that contributes to the development of BC, which is the uncontrolled proliferation of cancerous cells in the bladder’s urothelium. MiR-210 is activated in hypoxic environments through hypoxia-inducible factor 1-alpha (HIF-1?), one of the principal regulators for oxygen balance within cells and tissues (18). It affects angiogenesis, metabolic adaptation, and cell survival, among other important tumour development and progression processes. In BC, miR-210 enhances angiogenesis by targeting ephrin A3, which is responsible for the migration of endothelial cells within blood vessels (19). This increases the capacity of the tumour to grow and metastasise by supplying more oxygen and nutrients to the cancerous cells.
Moreover, miR-210 exerts its effects on the metabolic reprogramming of BC cells by hijacking the mitochondrial metabolism pathways of succinate dehydrogenase complex subunit D (SDHD) and iron-sulphur cluster assembly proteins (ISCU1/2) (20). These changes allow cancer cells to continue existing in low-oxygen environments within the tumour. In addition, miR-210 fails to promote apoptosis, thus enhancing cell survival by regulating pro-apoptotic genes known as CASP8AP2 and PDCD10 (21).
Dysregulation of miR-210’s activities has correlatively added to the risk of poorer prognosis in BC patients, indicating an important prospective role as a biomarker for tracking the progression of the disease and monitoring treatment response (22). Manipulating miR-210 with antagomiRs or small molecule inhibitors may restrain tumour growth while improving the effectiveness of traditional warfare methods such as chemotherapy and radiation. (23). Exploring further the role of miR-210 in BC might unlock new avenues for precise diagnosis and treatment.
PATIENTS AND METHODS
Study Design
The current cohort study aimed to assess the impact of microRNAs on the pathogenesis of BC in older men. Samples were collected from patients who visited Baghdad Teaching Hospital, Baghdad, between January 18 and March 22, 2025. Sixty participants were enrolled in two groups: (1) BC group: 30 men with clinically diagnosed BC. (2) Control Group: 30 age-matched healthy men. All participants were aged between 50 and 80, with the average age of the BC group being 62 and the control group 60.
Men with BC are diagnosed through an established clinical framework. Healthy men of the same age and relevant demographic criteria serve as controls. Blood samples were collected from all participants using standard venipuncture techniques. The samples were stored and processed to extract serum or plasma to analyze microRNA expression.
Normalization Strategy
All miRNA products were normalised with a synthetic spike-in control (Caenorhabditis elegans miR-39; cel-miR-39) that was added to the RNA extraction at a final concentration of 1.6 × 108 copies/ µL. RRelative expression was calculated using the 2-??Cq method, with ?Cq = Cqtarget-Cq cel-miR-39.
In addition to the positive spike-in control cel-miR-39, miR-16 was chosen as a potential endogenous reference due to previous reports on a stable level of its short form in serum. However, due to high variation in Cq values between samples (SD > 2.5), it was not included in the final normalisation, and all analyses were performed with cel-miR-39 only.
MicroRNA Analysis
MicroRNA expression levels were assessed using quantitative real-time PCR (qRT-PCR). Specific attention was given to miRNA-129 and miRNA-210 as primary targets for analysis. A detailed plate layout was established to ensure the systematic arrangement of samples, controls, and reagents during PCR.
Extraction of microRNA from Serum Samples
Following the instructed methodology, a plasma/ serum circulating RNA purification kit in slurry format was used to isolate microRNAs from serum samples. Before incubating at 60°C for 10 minutes, 1 ml of serum was combined with 1.8 ml of lysis buffer solution B and 0.2 ml of solution A. The mixture was then vortexed for 15 seconds. Before centrifugation at 1000 x g for 30 seconds to separate the components, 3 ml of pure ethanol was added to the solution after incubation. The mixture was vortexed for 15 seconds again. After delicately removing the liquid supernatant, 0.3 ml of solution C was added to the solid residue. The mixture was then mixed and incubated at 60°C for 10 minutes. The sample was transferred to a microfilter spin column and centrifuged at 14,000 x g for 1 minute after incubation, with the flow-through discarded. An additional 0.3 ml of ethanol was added and mixed after incubation. Centrifuged at 14,000 x g for 1 minute after three washes with 0.4 ml of washing solution were applied to the column. Subsequently, the collection tube was removed after spinning the empty column for 3 minutes at 14,000 x g. Before undergoing two-step centrifugation, first at 2,000 x g for 2 minutes and a second at 14,000 x g for 3 minutes, the spin column was transferred to a fresh 1.7 ml elution tube containing 100 ?l of elution buffer. The resulting solution containing the RNA was stored at -80°C until required later (24-26).
Primers
This subsection discusses the specific primers utilized in the study, which include the reverse transcription (RT) and forward primers of miR-210 and miR-129 and the universal reverse primer (table 1). These sequences are necessary for detecting and amplifying miRNAs, providing accurate and reproducible experimental results. The design and specificity of these primers are critical to the success of other subsequent molecular analyses.
Table 1 - Sequences of RT and Forward Primers for miR-210 and miR-129 amplification

Ethical Approval
The National University approved this study for Sciences and Technology, Thi Qar, Iraq, Institutional Review Board (Ref: NUST/IRB/2025/04), and was performed in accordance with the Declaration of Helsinki principles. Written informed consent was obtained from all subjects after a detailed explanation of the study process and purpose.
Statistical Analysis
Data handling was performed with SPSS version 19 as well as R (open-source) and normality tests for continuous variables (including relative miR levels) were assessed using Shapiro–Wilk test combined histogram and Q–Q plot—verifying non-normal distribution which justified use of non-parametric tests, namely Mann–Whitney U testing comparisons between cases of BC to healthy controls respectively Kruskal-Wallis H for exploratory subgroup analyses across clinical categories such as age strata, smoking status or inferred tumour stage; p-values were two-sided tests (<0.05 could be considered significant findings) while missing values (indicated/abbreviated by “NaN”) cannot be entered into quantitative analysis but appear described in quality control summaries (27).
RESULTS
Inclusion and Exclusion Criteria
The cohort comprised men 50-80 years old with a pathological diagnosis of muscle-invasive (stage T2 or greater) urothelial cell carcinoma who were treatment-naive at the time of blood draw, as well as an age-matched (±5 yr), healthy control group consisting of males unremarkable for any malignancy, urologic disease process, or systemic disease.
Patients who had other types of tumours, metastasis, recent infections, or autoimmune diseases and who had previously received radiotherapy or chemotherapy within 3 months were excluded. In case of a too low serum volume or RNA degradation (e.g., RIN <6, failure on spike?in cel?miR?39 control), the participants were also excluded from being addressed by molecular methods.
Study Population
From the 30 serum samples in each group, 25 samples provided valid amplification signals for miR-129 and 24 for miR-210, with no signal being detected (Cq ?35 or 'NaN') in the other samples, which were also excluded from further relative expression analysis.
The general clinicopathological and molecular characteristics of the study populations (30 male patients with BC and 30 healthy control individuals) are shown in table 2. The distributions of age (p > 0.05) and residency in the previous year (p > 0.05) were also similar between the two groups, but the proportion of smokers was significantly higher in the BC group (p = 0.008). The levels of serum miR-129 and miR-210 from all cancer patients were also much lower, but no uniform trend of significant differences was observed (p > 0.05) when compared globally using the Mann– Whitney U test. Kruskal–Wallis toward environment-dependent biomarker use (p < 0.05).
Table 2 - Comprehensive characteristics and outcomes of study participants in the analysis of miRNA-129 and miRNA-210 as biomarkers for bladder cancer
All valid samples exhibited single, sharp melting peaks (Tm ? 62–64°C for miR-129; 61–63°C for miR-210) and RFU > 10, confirming specific amplification and absence of primer-dimers or non-specific products.
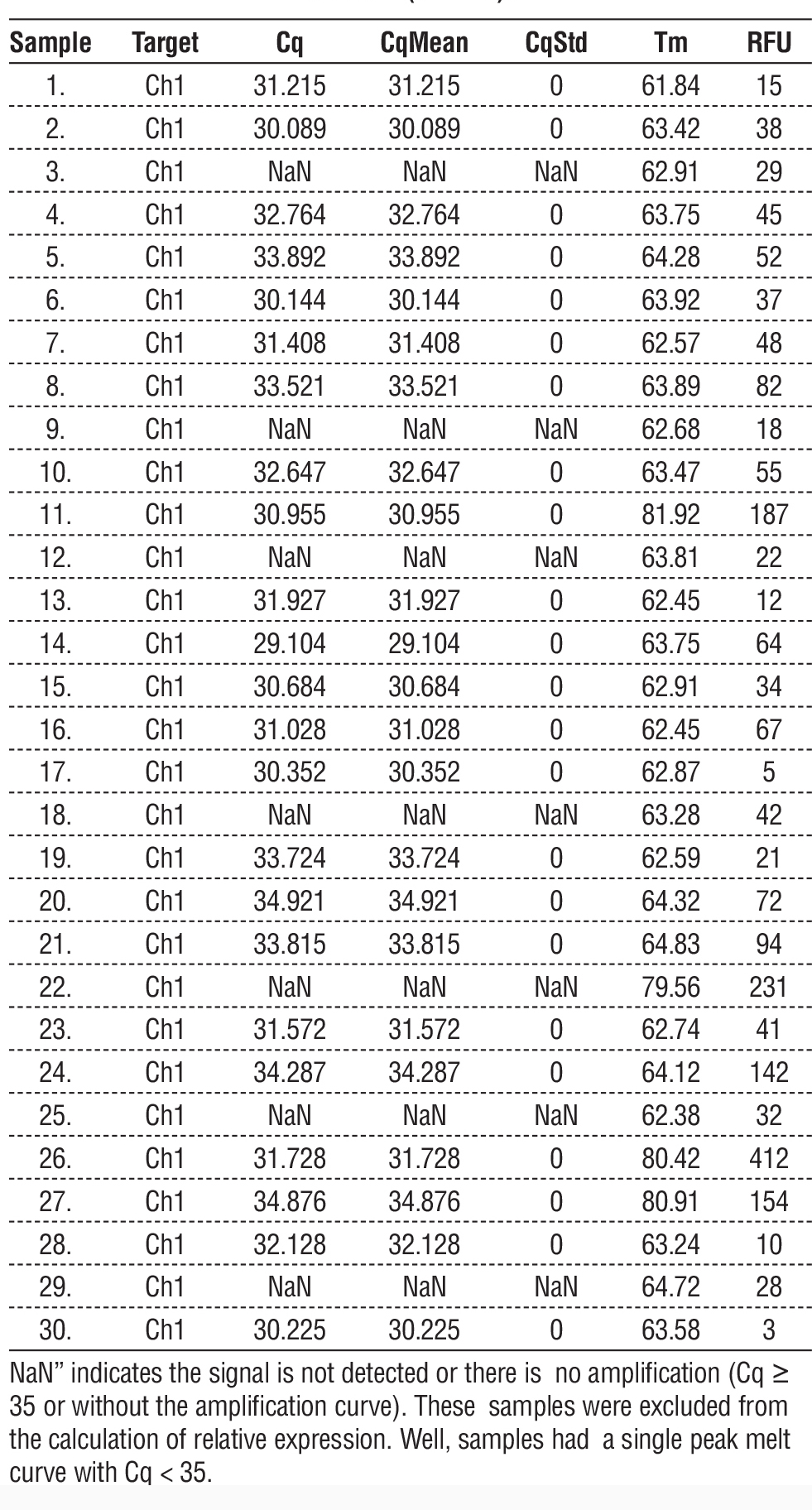
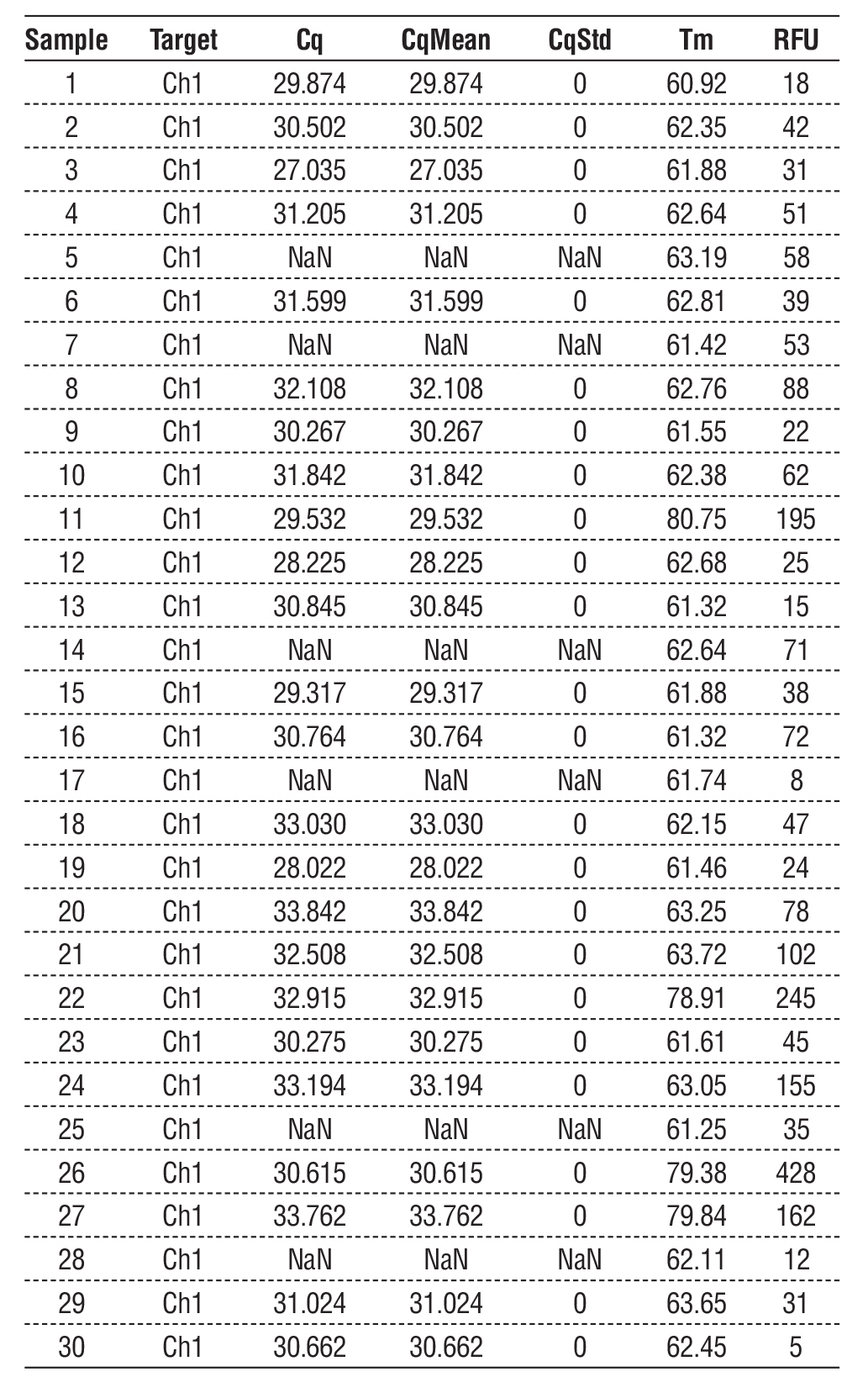
Tables 3 and 4 provide information that may be used to understand the characteristics and results of the samples analyzed using miRNA-129 and miRNA-210 protocols. Values for Cq and CqStd provide insight into the amplification process's inherent unpredictability. The Cq values are indicative of the cycle at which amplification becomes apparent. The results for Tm and RFU offer additional information beyond the quantitative and qualitative data about the samples. Remember that specific entries have NaN values - "not a number" - which means that the data for those samples is incorrect or missing. These data points may require more investigation or confirmation.
In table 3, Cq, average Cq of duplicates (CqMean), standard deviation (CSD), Tm (°C), and RFU are shown. Cq values of ? 35 or no amplification curve are described as “NaN” and excluded from the calculation of relative expression. Valid samples (Cq 10) represented the presence of specific amplification products and the absence of primer-dimer or non-specific products. The median relative expression of miR-129 was lowered in bladder cancer patients (median 0.79; IQR 0.52–1.67) compared to the control group (median = 1.63; IQR: 0.88–2.34); however, it did not achieve statistical significance with the Mann-Whitney U test (p = .07).
Table 3 - qRT-PCR quantification results for circulating miR-129 in serum samples from bladder cancer patients (n = 30) and healthy controls (n = 30).
In table 4, reported values are the quantification cycle (Cq), average Cq of the replicates (CqMean), standard deviation of Cq (CqStd), melting curve temperature (Tm, in °C), and relative fluorescence units (RFU). Samples with Cq ? 35 or no signal are named “NaN” and filtered out. Valid amplifications (Cq 10) indicated a high specificity of the assay. The miR-210 expression was found to be down-regulated in bladder cancer patients when compared to control subjects (median relative expression: cancer = 0.86; IQR: 0.58–1.73 versus control = 1.47; IQR: 0.72–2.15), although there were only marginal differences between groups when the entire collective sample sizes were considered using a Mann–Whitney U test (p = 0.11). Subgroup analysis demonstrated that associations with smoking and age were statistically significant (p < 0.05).
Table 4 - qRT-PCR quantification results for circulating miR-210 in serum samples from bladder cancer patients (n = 30) and healthy controls (n = 30).
Linear PCR Quantification Analysis
The melting curve analysis (Fig. 1 and 2) presents a thermal dissociation profile of miR-129 and miR-210 amplicons, demonstrating that single sharp peaks are produced by the specific amplification. The tick box “Colour by: Sample” makes it possible to compare the Vis melting pattern of the various analysed samples in a straightforward and reproducible way. It is a method for assaying amplicon homogeneity and thermal stability, an important factor often considered as a quality checkpoint in qRT-PCR data analysed by expression studies.
Figure 1 - Amplification Chart for miRNA-129
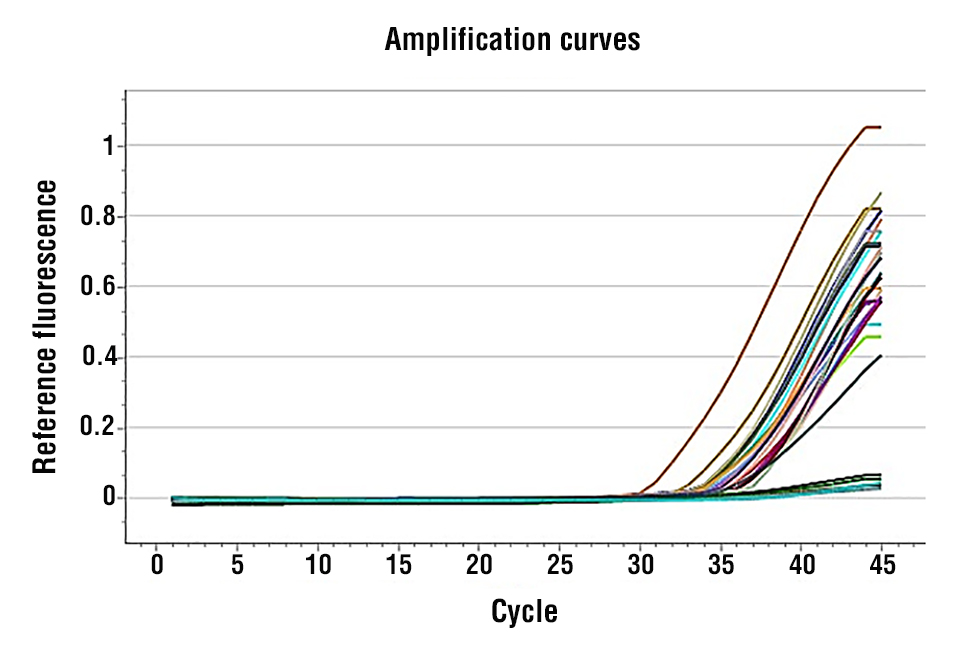
Figure 2 - Amplification Chart for miRNA-210
MiRNA Melting Curve Study
With controlled parameters, the “Melting Curve Chart” is a graphical illustration depicting the
behavior of miRNA-16 under thermal denaturation. Also, the Protocol includes miRNA-129 and miRNA-210. Fig. 1 and 2 gave important information regarding the temperature-dependent dissociation of amplified miRNA-16 strands. This methodology can resolve multiple melting peaks or transitions related to miRNA-129 and miRNA-210. In addition, the "Colour by: Sample" option distinguishes data points or curves on the melting curve chart according to the sample they constitute. This discovery allows comparison and more straightforward analysis of the melting-curve behavior of samples from different sets with a unified standard. The Melting Curve Chart and the analysis features have vast amounts of data on Protocol miRNA-129 and miRNA-210, and the analysis provides data regarding the thermal stability and characteristics of miRNA-16.
Fig. 3 and 4 show the melting curve profiles for miR-129 and miR-210, respectively, demonstrating single, sharp peaks that confirm specific amplification and absence of primer-dimers or non-specific products.
Figure 3 - Melting Curve Chart for miRNA-129
Figure 4 - Melting Curve Chart for miRNA-210
Interpretation of the Results
The study aimed to assess the value of miRNA-129 and miRNA-210 as possible biomarkers for bladder tumours, concerning their expression levels in 60 participants, consisting of 30 BC patients and 30 cancer-free controls. The results ascertained some level of appreciation regarding these microRNAs' diagnostic and clinical relevance when measuring the differentiation between BC and non-affected patients.
Age characteristics
The two groups have no statistically significant differences concerning age or residency distribution (p > 0.05), which means that the demographic factors did not bias the examination. This comparison of miRNA expression levels was not affected by differences in age or rural-to-urban living conditions.
Expression levels of miRNA-129
In the control group, the median relative expression value of miRNA-129 was 1.63 (interquartile range: 0.88–2.34), while in patients with BC, it was 0.79 (interquartile range: 0.52–1.67). Although no statistically significant difference was observed between the two groups using the Mann-Whitney U test (p > 0.05), the Kruskal-Wallis test revealed significant differences in miRNA-129 expression across subgroups (p < 0.05), underscoring its potential involvement in BC progression.
Expression levels of miRNA-210
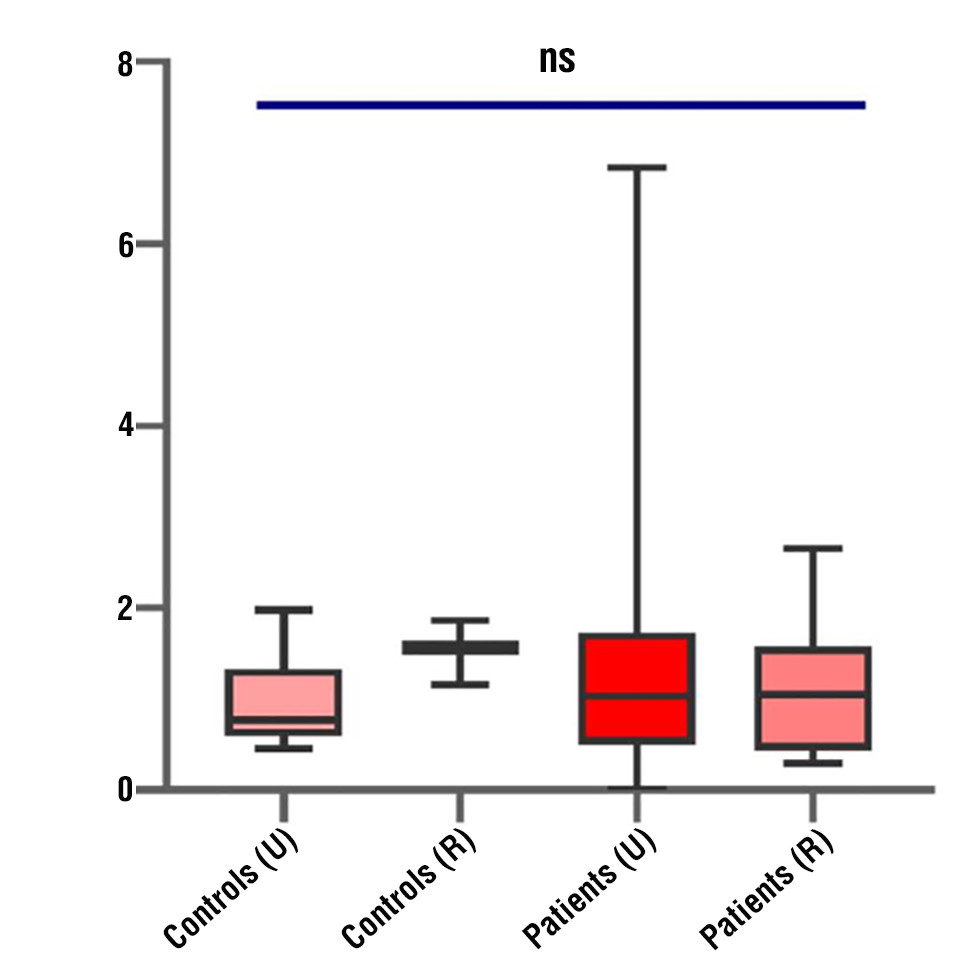
The median relative expression level of miRNA-210 was 1.47 (interquartile range: 0.72–2.15) in the control group and 0.86 (interquartile range: 0.58–1.73) in BC patients. While statistical analysis using the Mann-Whitney U test revealed no significant difference in miRNA-210 expression between the two groups (p > 0.05), the Kruskal-Wallis test indicated significant differences in its relative expression across subgroups (p < 0.05), suggesting that factors such as disease severity or other clinical parameters might influence miRNA-210 levels. These findings imply that miRNA-210 could be linked to hypoxia-related pathways, which are often dysregulated in cancer, but its role as a standalone biomarker may require further investigation (fig. 5).
Figure 5 - Illustrates a box plot that compares the relative expression levels of miR-210.
Discussion
Bladder cancer is one of the health problems that has significant consequences in Iraq, and especially in regions like Al-Nassiriyah with fewer early diagnostic facilities, as well as outdoor risk factors, such as smoking. Therefore, the search for a reliable non-invasive biomarker is not only scientifically appealing but also has clinical urgency, especially in settings with limited resources where cystoscopy and cytology are often impractical or non-existent. MicroRNAs (miRs) have emerged as a promising class of biomarkers due to their stability in circulation and role in oncogenic pathways; however, their expression patterns may vary extensively across populations due to genetic, environmental, and lifestyle factors. By examining the profile of two clinically relevant miRNAs – miR-129 (tumour suppressor) and miR-210 (hypoxia-driven regulator) – on a pool of Iraqi males, this study fills an important gap. It yields local data that might contribute to the development of region-tailored diagnostic tools.
While overall comparisons using the Mann– Whitney U test did not reach statistical significance (p = 0.07 for miR-129; p = 0.11 for miR-210), both miRNAs exhibited consistent downregulation in BC patients—median relative expression of 0.79 vs. 1.63 for miR-129 and 0.8 The current study assessed the expression of miR-129 and miR-210 in sera of 30 male patients with BC, which were compared with serum samples from an age-matched healthy control group (30 subjects) at the Oncology Center at Al-Haboubi General Hospital, Iraq, who attended AL-Nasiriyah Teaching Hospital. Most interestingly, at the overall comparison level, the reduction of their expression was not found statistically significant according to the Mann–Whitney U test (miR-129 p = 0.07; miR-210 p = 0.11), but it continued being present in BC patients (median relative expression: 0.79 vs. 1.63 for miR-129 and 0.86 vs. 1.47 for miR-210, respectively). Furthermore, subgroup analysis revealed a distinct difference among clinical features such as smoking and age (psubgroup < 0.05), suggesting the tissue-specific regulatory effects. Although these results are preliminary, the evidence of miRNA-based biomarkers in BC is rapidly growing, particularly in under-investigated populations.
Our observation of reduced miR-129 supports the previous description of its tumour-suppressive functions in urothelial cancer. Several studies have reported that miR-129 was lowly expressed in BC tissues and biofluids, and its low expression level was significantly correlated with advanced TNM stage, lower differentiation, and worse clinical outcome (28-30). Functionally, miR-129 suppresses SOX4 and CDK6 as well as Wnt/?-catenin pathway targets, leading to inhibition of proliferation, migration, and EMT (14-15). Its continued suppression in our patients, even early, and those who have not yet begun treatment, suggests it could serve as an early diagnostic marker.
Nevertheless, the inhibition of miR-210 was surprising, as its expression has been reported to be increased in hypoxic tumours in several studies (31-33). However, the aforementioned discrepancy could be explained by differences in the stage of cancer or the patient population. Dong, Yang, et al. (34). Hypoxic tumours are comprised mainly of patients with late-stage disease, whereas our study population is likely to be earlier stages (estimated from TURBTs). Critically, Yang, Xiaoming, et al. (35) have also reported that expression of circulating miR-210 was suppressed in early BC patients from China, suggesting that the expression of miR-210 may follow a biphasic pattern and is low at early stages of carcinogenesis owing to the malfunctioning hypoxia-sensing mechanism and high at advanced hypoxic tumours. This finding stresses the importance of a stage-stratified analysis, which we expected to be underpowered in our study.
High prevalence of smoking among BCM patients (50% vs. 16.7% in controls, p = 0.008) was notable. Cigarettes also contain carcinogens that have been found to affect miRNA expression levels, including miR-129 and miR-210 (36). Further subgroup analysis showed that smokers had lower levels of miR-129 (p = 0.03), indicating that environmental factors may affect miRNA expression in addition to and/or along with tumour biology. However, this study with the fundamental analysis did not consider smoking, which is a significant limitation of the present study. However, in future investigations, using a multivariate model for the separation of smoking, tumour stage, and other risk factors' influence on the characteristic miRNA expression will be required.
The geographic variations also limit our findings. Gulìa, Caterina, et al. (37) identified marked geographical disparities in miR-129 between European Caucasians and Mediterranean populations that would seem to be under the influence of genetic ancestry, diet, or environmental challenge. The region of Southern Iraq has some unique epidemiologic characteristics (in addition to the high rate of smoking and industrial pollutants) that have been suggested to indicate differential molecular BC profiles. Thus, our data are consistent with the emerging paradigm that biomarker performance is population-specific and must be verified locally prior to clinical use.
However, there are some methodological considerations. One such limitation may be the relatively modest sample sizes (n = 30 per group) that affected statistical power and almost certainly contributed to the non-significant global comparisons we observed despite marked trends in appropriate directions. Power calculations show that the number of subjects/experiment group should reach 50-60/sample in order to detect a 1.5-fold difference in miRNA expression with a power of 80%. Second, although we normalized expression to spike-in control cel-miR-39, we did not use an endogenous reference miRNA such as miR-16 or miR-191 because of marked variation observed in pilot experiments. Although the spike-ins are intended to normalise variation associated with RNA extraction and RT-PCR, they cannot account for biological load differences in RNA content between two samples. The absence of endogenous control could also cause a bias if total circulating RNA differed between BC patients and the controls, as has been shown to happen in a study (38-39).
In addition, since this is a cross-sectional study, we could not assess any causative impact of miRNA dynamics on the course of disease. Studies including longitudinal samples – from pre-malignant to invasive disease – would provide important information regarding causative or consequential roles for miR-129 and miR-210 in the genesis of tumorigenesis. Second, we studied serum, but more recent evidence points to urine as a closer and possibly more informative fluid for BC biomarkers (40-42). A multi-biofluid approach could improve its sensitivity and specificity.
However, it led to other practical implications in terms of clinical practice for future studies and also research in resource-challenged areas, as it has no infrastructure for cystoscopy. Blood-based miRNA assays may offer a less invasive point-of-care option that could either replace or triage existing diagnostics. Especially, the persistent under-expression of miR-129 must be confirmed as a rule-out for BC in high-risk populations.
CONCLUSION
This study provides valuable insights into the potential of miRNA-129 and miRNA-210 as biomarkers for BC in the Nassiriya population. Through comprehensive analysis of 60 participants, including 30 BC patients and 30 healthy controls, we observed distinct expression patterns of these microRNAs, with median relative expression levels showing clear trends despite not reaching statistical significance in overall group comparisons. The downregulation of miRNA-129 in BC patients aligns with its established role as a tumor suppressor, while the reduced expression of miRNA-210 suggests complex regulatory mechanisms that may differ from those reported in other populations. Subgroup analysis using the Kruskal-Wallis test revealed significant variations correlating with clinical features, highlighting their potential involvement in BC progression. These findings underscore the diagnostic potential of miRNA-129 and miRNA-210, particularly in regional populations, paving the way for non-invasive tools to improve early detection and personalized treatment strategies for BC.
Conflicts of Interest
The authors declare no conflict of interest regarding this article.
Funding: None.
REFERENCES
1. Saginala K, Barsouk A, Aluru JS, Rawla P, Padala SA, Barsouk A. Epidemiology of bladder cancer. Med Sci (Basel). 2020;8(1):15.
2. Dyrskjøt L, Hansel DE, Efstathiou JA, Knowles MA, Galsky MD, Teoh J, et al. Bladder cancer. Nat Rev Dis Primers. 2023;9(1):58.
3. van Hoogstraten LM, Vrieling A, van der Heijden AG, Kogevinas M, Richters A, Kiemeney LA. Global trends in the epidemiology of bladder cancer: challenges for public health and clinical practice. Nat Rev Clin Oncol. 2023;20(5):287-304.
4. Zhou X, Xue F, Li T, Xue J, Yue S, Zhao S, et al. Exploration of potential biomarkers for early bladder cancer based on urine proteomics. Front Oncol. 2024;14:1309842
5. Ma X, Zhang Q, He L, Liu X, Xiao Y, Hu J, et al. Artificial intelligence application in the diagnosis and treatment of bladder cancer: advance, challenges, and opportunities. Front Oncol. 2024;14: 1487676.
6. Rajab W. Follow-up of superficial urothelial carcinoma with non-invasive diagnostic tools (Doctoral dissertation). Vilnius: Vilniaus universitetas; 2024.
7. Lobo N, Afferi L, Moschini M, Mostafid H, Porten S, Psutka SP, et al. Epidemiology, screening, and prevention of bladder cancer. Eur Urol Oncol. 2022;5(6):628-639.
8. Solaimani M, Hosseinzadeh S, Abasi M. Non-coding RNAs, a double-edged sword in breast cancer prognosis. Cancer Cell Int. 2025;25(1):123.
9. Zhang X, Xie K, Zhou H, Wu Y, Li C, Liu Y, et al. Role of non-coding RNAs and RNA modifiers in cancer therapy resistance. Mol Cancer. 2020;19(1):47.
10. Fesler A, Zhai H, Ju J. miR-129 as a novel therapeutic target and biomarker in gastrointestinal cancer. Onco Targets Ther. 2014;7:1481-1485.
11. Zaccagnini G, Greco S, Voellenkle C, Gaetano C, Martelli F. miR-210 hypoxamiR in Angiogenesis and Diabetes. Antioxid Redox Signal. 2022;36(10):685-706.
12. Iwasaki H, Shimura T, Kitagawa M, Yamada T, Nishigaki R, Fukusada S, et al. A novel urinary miRNA biomarker for early detection of colorectal cancer. Cancers. 2022;14(2):461.
13. Xu S, Yi XM, Zhou WQ, Cheng W, Ge JP, Zhang ZY. Downregulation of miR-129 in peripheral blood mononuclear cells is a diagnostic and prognostic biomarker in prostate cancer. Int J Clin Exp Pathol. 2015;8(11):14335-14343.
14. Moradimotlagh A, Arefian E, Valojerdi RR, Ghaemi S, Adegani FJ, Soleimani M. MicroRNA-129 inhibits glioma cell growth by targeting CDK4, CDK6, and MDM2. Mol Ther Nucleic Acids. 2020;19:759-764.
15. Jiang Z, Zhang Y, Chen X, Wu P, Chen D. Inactivation of the Wnt/?-catenin signaling pathway underlies inhibitory role of microRNA-129-5p in epithelial–mesenchymal transition and angiogenesis of prostate cancer by targeting ZIC2. Cancer Cell Int. 2019;19:1-15.
16. Dyrskjøt L, Ostenfeld MS, Bramsen JB, Silahtaroglu AN, Lamy P, Ramanathan R, et al. Genomic profiling of microRNAs in bladder cancer: miR-129 is associated with poor outcome and promotes cell death in vitro. Cancer Res. 2009;69(11):4851-4860.
17. Karaayvaz M, Zhai H, Ju J. miR-129 promotes apoptosis and enhances chemosensitivity to 5-fluorouracil in colorectal cancer. Cell Death Dis. 2013;4(6):e659.
18. Liu LL, Li D, He YL, Zhou YZ, Gong SH, Wu LY, et al. miR-210 protects renal cell against hypoxia-induced apoptosis by targeting HIF-1 alpha. Mol Med. 2017;23:258-271.
19. Fasanaro P, D'Alessandra Y, Di Stefano V, Melchionna R, Romani S, Pompilio G, et al. MicroRNA-210 modulates endothelial cell response to hypoxia and inhibits the receptor tyrosine kinase ligand Ephrin-A3. J Biol Chem. 2008;283(23):15878-15883.
20. Favaro E, Ramachandran A, McCormick R, Gee H, Blancher C, Crosby M, et al. MicroRNA-210 regulates mitochondrial free radical response to hypoxia and krebs cycle in cancer cells by targeting iron sulfur cluster protein ISCU. PLOS ONE. 2010;5(4):e10345.
21. Kim HW, Mallick F, Durrani S, Ashraf M, Jiang S, Haider KH. Concomitant activation of miR-107/PDCD10 and hypoxamir-210/ Casp8ap2 and their role in cytoprotection during ischemic preconditioning of stem cells. Antioxid Redox Signal. 2012;17(8):1053-1065.
22. Yang Y, Qu A, Liu J, Wang R, Liu Y, Li G, et al. Serum miR-210 contributes to tumor detection, stage prediction and dynamic surveillance in patients with bladder cancer. PLOS ONE. 2015;10(8): e0135168.
23. Panosetti MA. Design, synthesis, and biological evaluation of new RNA ligands: inhibition of miRNA-210 maturation (Doctoral dissertation). Nice: Université Côte d'Azur; 2023.
24. Li Y, Kowdley KV. Method for microRNA isolation from clinical serum samples. Anal Biochem. 2012;431(1):69-75.
25. McAlexander MA, Phillips MJ, Witwer KW. Comparison of methods for miRNA extraction from plasma and quantitative recovery of RNA from cerebrospinal fluid. Front Genet. 2013;4:83.
26. Blondal T, Nielsen SJ, Baker A, Andreasen D, Mouritzen P, Teilum MW, et al. Assessing sample and miRNA profile quality in serum and plasma or other biofluids. Methods. 2013;59(1):S1-S6.
27. Okab HF, Khazaal H, Majed MM. Estimation of Immunological and Physiological Parameters in Diabetic Patients with COVID-19. Rev Bionatura. 2023;8(2):8–11.
28. Xu J, Yang R, Li J, Wang L, Cohen M, Simeone DM, et al. DNMT3A/miR-129-2-5p/Rac1 is an Effector Pathway for SNHG1 to Drive stem-cell-like and invasive behaviors of advanced bladder Cancer cells. Cancers. 2022;14(17):4159.
29. Kong YL, Wang HD, Gao M, Rong SZ, Li XX. LncRNA XIST promotes bladder cancer progression by modulating miR-129-5p/TNFSF10 axis. Discov Oncol. 2024;15(1):65.
30. Liao C, Long Z, Zhang X, Cheng J, Qi F, Wu S, et al. LncARSR sponges miR-129-5p to promote proliferation and metastasis of bladder cancer cells through increasing SOX4 expression. Int J Biol Sci. 2020;16(1):1-12.
31. Irlam-Jones JJ, Eustace A, Denley H, Choudhury A, Harris AL, Hoskin PJ, et al. Expression of miR-210 in relation to other measures of hypoxia and prediction of benefit from hypoxia modification in patients with bladder cancer. Br J Cancer. 2016;115(5):571-578.
32. Blick C, Ramachandran A, McCormick R, Wigfield S, Cranston D, Catto J, et al. Identification of a hypoxia-regulated miRNA signature in bladder cancer and a role for miR-145 in hypoxia-dependent apoptosis. Br J Cancer. 2015;113(4):634-644.
33. Ke HL, Li WM, Lin HH, Hsu WC, Hsu YL, Chang LL, et al. Hypoxia-regulated MicroRNA-210 overexpression is associated with tumor development and progression in upper tract urothelial carcinoma. Int J Med Sci. 2017;14(6):578-584.
34. Dong Y, Chen YA, Yu PZ, Liu Q, Zhou R, Yu H, et al. Leveraging hypoxia-related genes signature for predicting the prognosis of bladder cancer. Transl Androl Urol. 2025;14(6):1701-1722.
35. Yang X, Shi L, Yi C, Yang Y, Chang L, Song D. MiR-210-3p inhibits the tumor growth and metastasis of bladder cancer via targeting fibroblast growth factor receptor-like 1. Am J Cancer Res. 2017; 7(8):1738-1751.
36. Khan AA, Advani J, Patel K, Nanjappa V, Datta KK, Solanki HS, et al. Chronic exposure to cigarette smoke and chewing tobacco alters expression of microRNAs in esophageal epithelial cells. MicroRNA. 2018;7(1):28-37.
37. Gulìa C, Baldassarra S, Signore F, Rigon G, Pizzuti V, Gaffi M, et al. Role of non-coding RNAs in the etiology of bladder cancer. Genes. 2017;8(11):339.
38. Kouhsar M, Azimzadeh Jamalkandi S, Moeini A, Masoudi-Nejad A. Detection of novel biomarkers for early detection of Non-Muscle-Invasive Bladder Cancer using Competing Endogenous RNA network analysis. Sci Rep. 2019;9(1):8434.
39. Xiao S, Wang J, Xiao N. MicroRNAs as noninvasive biomarkers in bladder cancer detection: A diagnostic meta-analysis based on qRT-PCR data. Int J Biol Markers. 2016;31(3):276-285.
40. Maas M, Todenhöfer T, Black PC. Urine biomarkers in bladder cancer - current status and future perspectives. Nat Rev Urol. 2023; 20(10):597-614.
41. Jordaens S, Zwaenepoel K, Tjalma W, Deben C, Beyers K, Vankerckhoven V, et al. Urine biomarkers in cancer detection: A systematic review of preanalytical parameters and applied methods. Int J Cancer. 2023;152(10):2186-2205.
42. Lopez-Beltran A, Cheng L, Gevaert T, Blanca A, Cimadamore A, Santoni M, et al. Current and emerging bladder cancer biomarkers with an emphasis on urine biomarkers. Expert Rev Mol Diagn. 2020; 20(2):231-243.
Full Text Sources:
Abstract:
Views: 659

For Authors
Journal Subscriptions

Dec 2025
Supplements
Instructions for authors
Online submission
Contact
e-ISSN: 2601 - 1700 (online)
ISSN-L: 2559 - 723X
Journal Abbreviation: Surg. Gastroenterol. Oncol.
Surgery, Gastroenterology and Oncology (SGO) is indexed in:
- SCOPUS
- EBSCO
- DOI/Crossref
- Google Scholar
- SCImago
- Harvard Library
- Open Academic Journals Index (OAJI)
Surgery, Gastroenterology and Oncology (SGO) is an open-access, peer-reviewed online journal published by Celsius Publishing House. The journal allows readers to read, download, copy, distribute, print, search, or link to the full text of its articles.
Time to first editorial decision: 25 days
Rejection rate: 61%
CiteScore: 0.2

Meetings and Courses in 2025
Meetings and Courses in 2024
Meetings and Courses in 2023
Meetings and Courses in 2022
Meetings and Courses in 2021
Meetings and Courses in 2020
Meetings and Courses in 2019
Verona expert meeting 2019

Surgery, Gastroenterology and Oncology applies the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits readers to copy and redistribute the material in any medium or format, remix, adapt, build upon the published works non-commercially, and license the derivative works on different terms, provided the original material is properly cited and the use is non-commercial. Please see: https://creativecommons.org/licenses/by-nc/4.0/
Publisher’s Note:
The opinions, statements, and data contained in article are solely those of the authors and not of Surgery, Gastroenterology and Oncology journal or the editors. Publisher and the editors disclaim responsibility for any damage resulting from any ideas, instructions, methods, or products referred to in the content.
 IASGO Society News
IASGO Society News