Surgery, Gastroenterology and Oncology
Official journal of the International Association of Surgeons, Gastroenterologists and Oncologists
|
|
The Commonest Medical, Surgical, and Oncological Causes of Acute Abdomen in Adults with COVID-19. A prospective Observational Study
Objectives: This study sought to determine the commonest causes of acute abdominal pain (medical, surgical, and oncological) during the COVID-19 pandemic. Methods: From May 1, 2020, to February 28, 2022, 910 adult patients > 18 years of age of both sexes were admitted to the emergency department or already admitted but complaining of an acute abdomen and diagnosed with COVID-19.
Results: The mean age of the studied group was 48.13+-11.97 years, with male predominance (66%). Medical causes of the acute abdomen were in 34% of cases, and 66% were surgical causes. Gastric causes of the acute abdomen were the most common medical causes. Referred pain from COVID-19 pneumonia was the cause of abdominal pain in 20.8% of cases. Acute appendicitis was the commonest surgical cause of acute abdomen (42.4%), mostly Grade V, followed by acute cholecystitis (11%) and biliary colic (9.8%). Acute pancreatitis was the cause in 12.7% of cases, and its causes were mainly idiopathic (48.7%).
Conclusions: Surgical causes of acute abdominal pain were more common than medical causes. The commonest medical causes of the acute abdomen during COVID-19 were gastric causes, while acute appendicitis, gallbladder diseases, and intestinal ischemia were the commonest surgical causes. Idiopathic acute pancreatitis was more common than gallstone pancreatitis, and most cases were mild. Surgery is the most common cause of death from the acute abdomen, followed by respiratory failure caused by COVID-19 and then medical causes of acute abdomen.
Trial registration: It was approved by the institutional review board (IRB) for all parts of this study by ZUMEC 152020-23.Clinical trial registration: NCT05295251.Retrospective registered.
INTRODUCTION
Coronaviruses are a large virus family that causes mild to severe respiratory infections. 2019-nCoV is a new coronavirus found in Wuhan, China. Its global spread has been rapid since its late 2019 discovery. Symptoms of exposure include dry cough, fever, and fatigue (1).
It is unclear how common gastrointestinal (GIT) symptoms are. However, as the pandemic progressed, COVID-19's clinical manifestations became more evident, and GIT symptoms more common (2). A recent meta-analysis of 47 studies with 10,890 COVID-19 patients found that 2.7% had abdominal pain, usually accompanied by diarrhoea, nausea, and vomiting (3). Since the beginning of the pandemic, COVID-19's acute abdomen has posed diagnostic and therapeutic challenges (4). Distinguishing between diseases that cause acute abdominal pain can be difficult. To avoid further mortality or morbidity, the examining physician should prioritise life-threatening conditions that may necessitate immediate surgical intervention. This study looked into the commonest causes of acute abdominal pain (medical, surgical, and oncological causes) during the COVID-19 pandemic. The study provides the first comprehensive coronavirus-era assessment of acute abdomen that a deep gap needed to be filled and helps understand this critical aspect during the COVID-19 pandemic.
MATERIALS AND METHODS
Participants and study design
This multi-centre study was conducted at our universities from May 1, 2020, to February 28, 2022, in three departments (surgery, internal medicine, and oncology). In this study, 20222 patients with acute abdominal pain with or without other GIT or respiratory symptoms were admitted to our emergency room, and 910 patients (4%) were COVID-19 positive. This study included adults over the age of 18 of both sexes who had been admitted to the emergency room or were already in the hospital but had an acute abdomen and were diagnosed with COVID-19 by a thoracic computed tomography (CT) scan (5) or reverse transcription-polymerase chain reaction (RT-PCR) of SARS-CoV-2 RNA (6). Patients under the age of 18 years who were COVID-19 negative were excluded. The patient's blood coagulation function, procalcitonin, D-dimer, and CRP (C-reactive protein) levels were measured in addition to routine laboratory tests such as complete blood count and urinalysis.
Mesurement of outcome
An acute abdomen is an acute onset of abdominal pain that requires immediate and accurate diagnosis and treatment to prevent morbidity and mortality (7). Acute pancreatitis (AP) was diagnosed according to the American College of Gastroenterology (ACG) guidelines, which requires at least two of the following three criteria: abdominal pain, elevated amylase or lipase levels (three times the upper limit of normal), and unusual imaging findings. The latter could be more important for COVID-19 patients with severe cases that require care in an intensive care unit with ventilatory support and where a clinical evaluation for abdominal symptoms might not be possible (8). Contrastenhanced computed tomography (CT) provides over 90% sensitivity and specificity for diagnosing acute pancreatitis (9). The severity of AP was classified into mild, moderately severe, and severe according to the revised Atlanta classification (RAC). Mild AP has neither organ failure nor local/systemic complications.
Moderate AP is characterised by transient organ failures or local complications, while severe AP is characterised by single or multiple persistent organ failures lasting more than 48 hours (10). The severity of acute appendicitis was based on the emergency surgery grading system of the American Association for Surgery of Trauma (11). The Tokyo Guidelines severity risk scale was used to assess acute cholecystitis (12). All cases underwent appendectomy, resected bowel, pancreatic tissues, aspirated fluid or pus, and excised lymph nodes were exposed to cytological or histopathological examination. The unusual morphological changes raise the possibility of COVID. They include apoptotic necrosis, a prominent histiocytic infiltrate, fibrinoid necrosis of blood vessels, microangiopathy, microthrombi, and increased platelet aggregation with no significant neutrophilic infiltration.
In this situation, PCR was requested on the removed specimens or fluids, including pus, for the possibility of being positive for SARS-Cov-2 RNA (nucleic acid), even if, during admission, three nose and throat swabs for COVID-19 were negative. The assay was performed according to the WHO guidelines (13).
Statistical analysis
For normally distributed continuous variables, the mean ± SD was used; for non-normally distributed continuous variables, the median was used. Percentages were used to represent categorical variables. The laboratory results were assessed to determine if they were within the normal range. SPSS version 20 was used for all statistical analyses (SPSS, Chicago, IL).
RESULTS
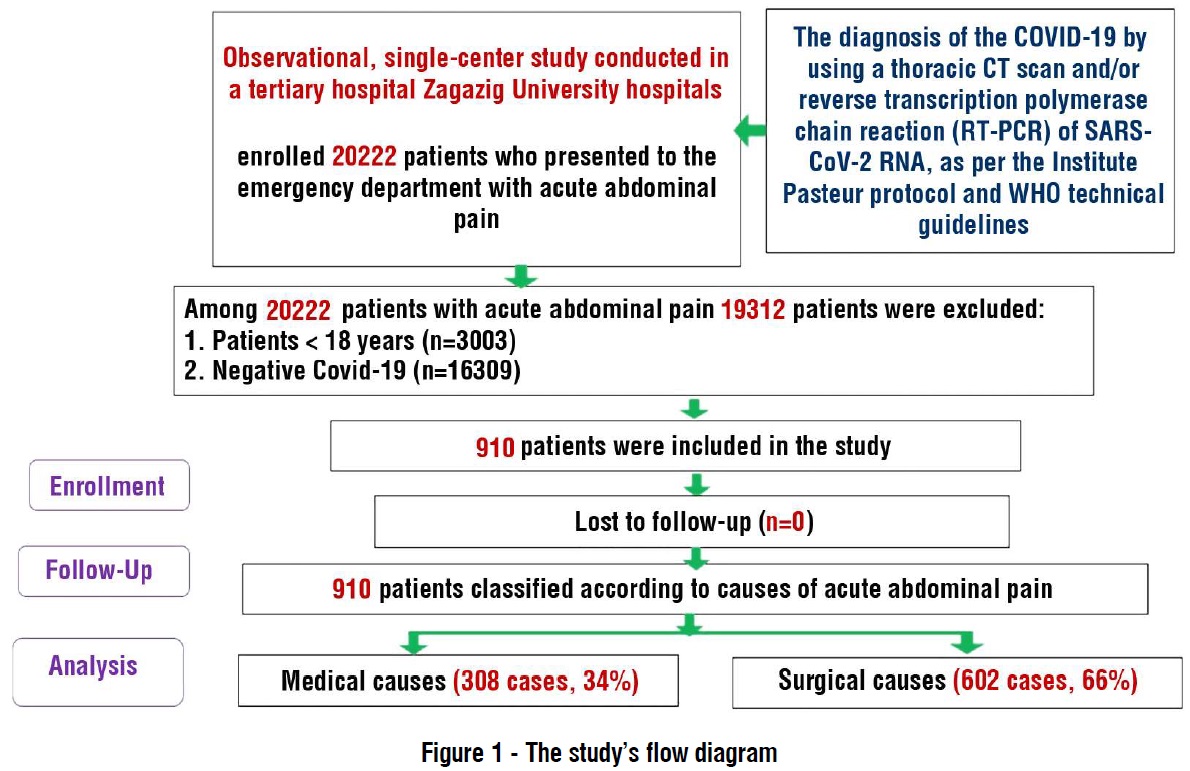
A flow diagram of inclusion and exclusion criteria is presented in fig. 1.
Demographic and patient characteristics showed that the mean age of the studied group was 48.13 +- 11.97 years, with male predominance (66%).
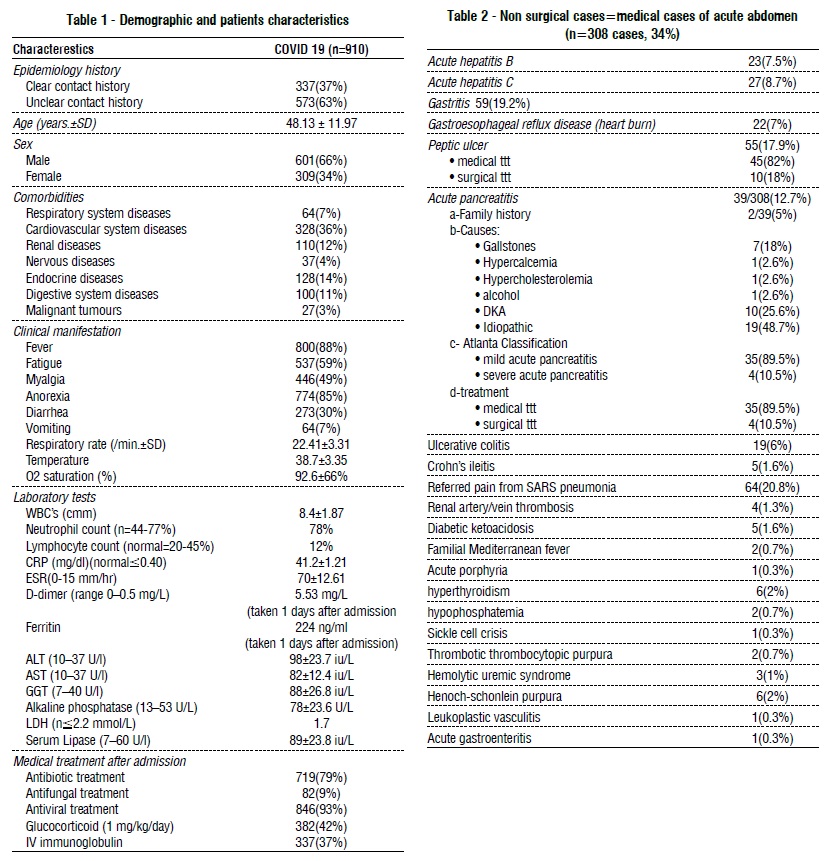
The commonest symptoms were fever (88%) and lack of appetite (85%). The associated respiratory symptoms with acute abdomen were present in 36% of patients, with a median respiratory rate of 22.41 ± 3.31/min and O2 saturation of 92.6 ± 66%. Laboratory findings include: WBCs (8.4±1.87/mm), neutrophil count (78%), CRP (41.2±1.21mg/dl), D-Dimer (5.53mg/l), ALT (98±23.7 IU/L) and lipase (89±23.8 IU/L). 37% of people who had COVID-19 had clear contact with someone who had COVID-19 before, with cardiovascular disease the most common co-morbidity (36%). The majority of cases (93%) received antiviral treatment, followed by antibiotics (79%) and glucocorticoids (42%), as shown in table 1.
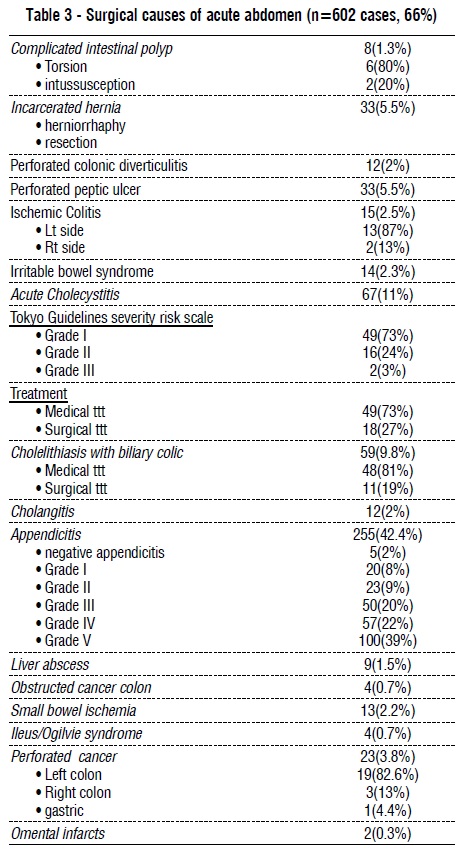
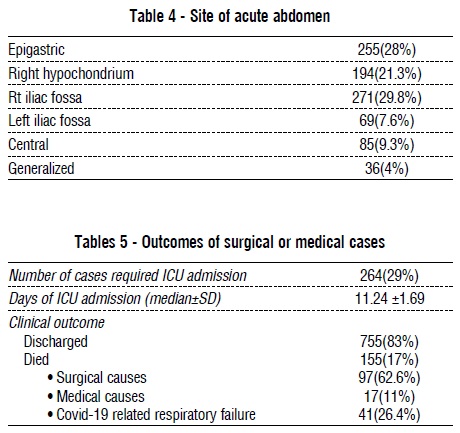
Medical causes of the acute abdomen were in 34% of cases, and 66% were surgical causes. Gastric causes of the acute abdomen were the most common medical causes, with gastritis, gastroesophageal reflux diseases, and peptic ulcer (PU) diseases in 19.2%, 7%, and 17.9% of the total, respectively. Referred pain from COVID-19 pneumonia was the cause of abdominal pain in 20.8% of cases. Acute appendicitis was the commonest surgical cause of acute abdomen (42.4%), mostly Grade V, followed by acute cholecystitis (11%) and biliary colic (9.8%). Acute pancreatitis was the cause in 12.7% of cases, and its causes were mainly idiopathic (48.7%), followed by diabetic ketoacidosis (25.6%), and gallstone pancreatitis (18%) and 89.5% of pancreatitis was mild. Ischemic colitis was found in 2.5% of cases, mainly in the left colon. Small bowel ischemia was found in 2.2% of cases. Crohn's disease was found in 1.6% of cases and ulcerative colitis in 6% of cases. The commonest site of the acute abdomen was the right iliac fossa (29.8%), followed by epigastric pain (28%) and the right hypochondrium (21.3%) (tables 2–4 and figs. 2-3).
Table 5 presents the outcomes of cases admitted with acute abdomen. 29% of cases required ICU admission, with a median day of ICU admission of 11.24 +- 1.69. The mortality rate was 17%, primarily due to surgical causes (62.6%).
Discussion
Acute abdominal pain in COVID-19 patients is challenging to diagnose and, if treated late, will increase morbidity and mortality. Cholecystitis and pancreatitis cause pain in the right upper quadrant of the abdomen (RUQ), similar to right lower lobe pneumonia caused by COVID-19, especially in the absence of other typical respiratory infection symptoms (fever, oxygen requirement, and tachypnea) (14). As a result, this situation is complicated more by the lower diagnostic value of PCR of the nasal swab (15). As a result of having basal lung COVID-19 pneumonia, 64 patients in our study had upper abdominal pain. We needed to ensure we had the correct diagnosis so we did not risk exposing patients to unnecessary procedures. Radiological, laboratory and clinical diagnoses were essential items in the follow-up of these patients.
Our study has several compelling findings with important implications. We found that gastritis was the commonest medical cause of acute abdomen (59/308, 19.2%) found during the era of COVID-19, while acute appendicitis (42.4%), acute cholecystitis (11%), biliary colic (9.8%) and acute pancreatitis (12.7%) were the most common surgical causes. Furthermore, surgery was the most common cause of acute abdominal pain (66%). On the contrary, a study involving three COVID- 19 patients with acute abdominal pain found that none had any surgical complications from COVID-19.
Gastroenteritis is the commonest cause of afebrile acute abdomen (16). Another study showed 3 cases of acute abdomen diagnosed as right iliac fossa pain, right upper quadrant pain, and epigastric pain, all resolved on medications without surgical intervention (16). Our results showed a more significant number of surgical cases, mostly due to our large sample size.
The diagnosis of gastritis was based on an endoscopic diagnosis with a biopsy sent for pathological examination and PCR for the COVID-19 virus. All cases were positive for COVID-19. Antibody levels for H-pylori in the blood were done in all cases, and all cases revealed negative H-pylori infection. SARS-CoV-2 enters the stomach via saliva and is then exposed to the stomach's acidic environment. A pH of 2 affects the virus's integrity (17). We did not prescribe a proton pump inhibitor (PPI) as it decreases the stomach's acidity, allowing the virus to access the small intestine with a large viral load. All cases resolve spontaneously without complications within two weeks.
Covid-19 causes lymphopenia and neutrophilia, as well as clinical symptoms such as pain, anorexia, nausea, tenderness in the right iliac fossa, and a leftward shift in white blood cell count, mimicking appendicitis, and they concluded that CT is necessary for the diagnosis of acute appendicitis in the era of COVID-19 (18-19).
According to our study, acute appendicitis is the most frequently encountered surgical cause of acute abdomen (42.4%). Our previous study confirmed that appendicitis was more severe in the COVID era (20), possibly due to appendicular artery thrombosis or a delay in diagnosis. We found five cases that revealed negative macroscopic appendicitis during exploration. In these five cases, the only intraoperative findings were straw-coloured exudative fluid in the pelvis and enlarged mesenteric lymph nodes in the ileal mesentery.


The histological examination and PCR for COVID-19 were negative in all resected appendices but positive for mesenteric lymph node biopsy and the aspirated peritoneal fluid. We relied on CT abdomen to diagnose acute appendicitis in cases of vague abdominal ultrasonography findings. However, these negative cases showed vague CT findings, so we operated in all 5 cases. All five of these cases showed negative shifting tenderness. A study consistent with ours suggested that mesenteric lymph node enlargement could cause abdominal pain in COVID-19 patients and mimic acute appendicitis (21). Several studies have examined post-SARS-CoV2 liver disorders. SARS-CoV2 binds to ACE2 on cholangiocytes, causing cholangiocyte dysfunction and liver damage (22–24). 43 out of 99 patients had an abnormal liver function, especially higher ALT or AST levels, and 1 in 99 had severe liver damage (1). In the study of Zhang et al, the incidence of liver damage was 78% of the 82 confirmed deaths from SARS-CoV2 infection (25). We found 11% cholecystitis and 9.8% biliary colic, and most cases improved with medical treatment (73% and 81%, respectively). The remaining cases required either open or laparoscopic cholecystectomy with low-pressure pneumoperitoneum due to decreased pulmonary function associated with COVID-19 infection (26-27). Nine patients (1.5%) developed a suppurative liver abscess, which was solitary and located in the right lobe of the liver in all cases. These abscesses developed one week after developing respiratory pneumonia consistent with COVID-19 (28) with a negative bacterial culture of the aspirate from the abscess and a positive PCR for the COVID-19 virus. A study by Liemarto et al. evaluated one patient who developed two liver abscesses during the COVID-19 era, occupying most of the right lobe of the liver and treated by open drainage. There were no bacteria in the abscess pus culture (29). Acute pancreatitis (AP) is the most common GIT disease that necessitates immediate hospitalisation and is caused by obstruction of the common bile duct, alcohol abuse, hypercalcemia, viral, bacterial, and parasitic infections (30). The aetiology of acute pancreatitis is idiopathic in 15–25% of cases, and this incidence increased to 69% during the COVID-19 pandemic (31). The virus can cause acute pancreatitis through direct spread from the duodenal epithelium to the pancreatic duct and then to acinar and islet cells via ACE2 expression or by causing diffuse severe endotheliitis of the pancreatic sub-mucosal vessels with micro-ischemia damage (32). SARS-CoV can be isolated from pancreatic fluid or tissue (33-34). We found that idiopathic pancreatitis was the most common cause of pancreatitis (19 patients, 48.7%), followed by diabetic ketoacidosis (10 patients, 25.6%) and gallstone pancreatitis (7 patients, 18%). Our study found that 12.7% of patients had acute pancreatitis, mostly mild acute pancreatitis (89.5%), and responded to conservative treatment. Previous studies (35) agreed that most cases of acute pancreatitis during the COVID-19 pandemic were milder.
Four patients developed severe acute pancreatitis (10.5%). Two patients had localised pancreatic necrosis with the collection, which was diagnosed and drained using CT guidance, and the drained fluid showed positive for COVID-19 virus by PCR. The other two cases of severe pancreatitis developed total pancreatic necrosis, diagnosed by CT and treated by surgical debridement. Four patients died two weeks later due to sepsis and organ failure. In the era of the COVID-19 pandemic, the incidence of necrotizing pancreatitis was 5-10%, which is similar to our results (36). In comparison, the mortality rate of acute pancreatitis was 0.8% (37), which was higher than in our study (10% mortality rate). The higher mortality rate in our study may be due to our study's smaller sample size, associated COVID-19 infection, and many cases of chronic pancreatitis in the other study. We relied on clinical data, serum lipase, and CT abdomen to confirm the diagnosis of acute pancreatitis. The median value of serum lipase in cases of acute pancreatitis was not much increased, with a mean value of 89±23.8 IU/L.
A study by Liu and colleagues reported that 16.4% had increased lipase levels, and 7.46% showed pancreatic changes on CT imaging. Surprisingly, the serum markers were mildly elevated without abdominal pain (38).
Gastrointestinal ischemia was significantly increased in COVID-19 patients, caused mainly by adverse pharmacological effects, micro-vascular thrombotic events, and viral entero-neuropathy (39). COVID-19 can cause venous and arterial thrombosis, especially in patients with severe diseases (40). In our study, ischemic colitis, small intestine ischemia, and omental infarcts occurred in 2.4 %, 13%, and 0.3 % of our trial, respectively, with ischemic colitis developing primarily in the left colon (87%) and to a lesser extent in the right colon (13%) due to the lower vascularity of the left colon compared to the right colon. All of the cases presented with rectal bleeding in addition to pain and were diagnosed using CT angiography. Four cases of left colon gangrene (3 splenic flexure gangrene and one descending colon gangrene) necessitating immediate surgical intervention with resection with colostomy with secondary look operation after two days were intended in all cases with no further resection. All resected specimens were sent for histopathological examination and PCR for COVID-19 virus detection and were positive for the virus in all cases. One case developed sepsis and multisystem organ failure in the postoperative course, and the patient died eight days later. In 13 patients (2%), small bowel ischemia developed, and all cases necessitated surgical exploration. 11 patients developed multiple small infarcts of the small intestine, necessitating resection of the gangrenous part of the small intestine, while two patients developed gangrene of the terminal ileum and right colon, necessitating right hemicolectomy. Secondary look operations were intended in all cases, and four cases required another resection. In the postoperative course, four patients developed sepsis with multisystem organ failure at 4, 8, 12, and five days postoperatively and died. All resected specimens were sent for pathological examination and PCR for COVID-19, and all were positive. All patients were started on a therapeutic dose of heparin for systemic anticoagulation peri- and postoperatively. Our explanation for intestinal ischemia in COVID-19 is attributed to reduced venous flow due to prolonged bed rest in critically ill COVID-19 patients. SARS-CoV-2 can cause direct damage to the vessel wall and subsequent coagulopathy by binding to the ACE-2 receptors that are found abundantly on endothelial cells and may be due to hypoxia. These explanations are consistent with other studies (41-43). The acute abdomen in cancer patients is classified broadly. It includes malignant intestinal obstruction (44), typhlitis/neutropenic colitis (45), radiation enteritis, and mesenteric bowel ischemia (46). In our series, perforated colon cancer occurred in twenty-three cases (3.6%). We assumed ischemia from COVID-19 or delayed diagnosis due to patients' fear of contracting an infection or decreased endoscopic screening and diagnosis strictly limited to our centre's emergency or critical GI procedures. Complicated intestinal polyps were the causes of acute abdomen in 8 patients (1.3%), who presented with torsion of polyps (6 patients, 80%) and intussusception (2 patients, 20%). Malignant tumours of the acute abdomen were due to perforation of the left cancerous colon (19 patients), the right colon (3 patients), and gastric cancer perforation (one patient), while an obstructed cancerous colon was the cause of the acute abdomen in 4 patients (0.7%). The increased number of obstructed and perforated colon cancers was due to decreased screening of cancer colon by endoscopic examination and patient fear of contacting the hospital for GIT problems to avoid contact with COVID-19 infection. Mortality in complicated cancer patients occurred in 7 patients (7/27, 26%). One died of an obstructed cancer colon, and six patients died of perforated cancer colon. All deaths were attributed to postoperative sepsis and multisystem organ failure. No cases of acute appendicitis or pancreatitis were due to cancer. A recent study agreed with our explanation for why there are more complicated cancers, and they are getting worse (47). The current study has many advantages. Our analysis was based on a prospective study with a large sample size, eliminating bias and ensuring the findings' reliability and generalizability. Our analysis is a multipleinstitution study, so regional bias is not possible. Our findings are reliable because of rigorous data collection and diagnosis. One problem with this study is that we did not look for SARS-CoV-2 RNA in stool samples from people with COVID-19 to see if there was a link between the number and severity of digestive symptoms and the presence of viral RNA in stool samples.
CONCLUSIONS
The commonest medical causes of the acute abdomen during COVID-19 were gastric causes, while acute appendicitis, gallbladder diseases, and intestinal ischemia were the commonest surgical causes. Idiopathic acute pancreatitis was more common than gallstone pancreatitis, and most cases were mild. The most common cause of death from the acute abdomen is surgery, followed by COVID-19-related respiratory failure, and finally, medical causes of acute abdomen. Ischemia of the colon frequently occurs in the left colon, small intestine, and right colon. It was approved by the institutional review board (IRB) for all parts of this study by ZUMEC 152020-23.All patients agreed to participate and publication. Clinical trial registration: NCT05295251. All authors shared in study design, data collection, statistical analysis, data interpretation, literature search, manuscript writing and all authors shared final approval of manuscript. The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request. none. none.
REFERENCE
1. Chen N, Zhou M, Dong X, Qu J, Gong F, Han Y, et al. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. The Lancet. 2020; 395(10223):507-13.
2. Gao QY, Chen YX, Fang JY. 2019 Novel coronavirus infection and gastrointestinal tract. J Dig Dis. 2020; 21(3):125-6.
3. Sultan S, Altayar O, Siddique SM, Davitkov P, Feuerstein JD, Lim JK, et al. AGA Institute Rapid Review of the Gastrointestinal and Liver Manifestations of COVID-19, Meta-Analysis of International Data, and Recommendations for the Consultative Management of Patients with COVID-19. Gastroenterology. 2020;159(1):320-34.e27.
4. Cai M, Wang G, Zhang L, Gao J, Xia Z, Zhang P, et al. Performing abdominal surgery during the COVID-19 epidemic in Wuhan, China: a single-centred, retrospective, observational study. British Journal of Surgery. 2020;107(7):e183-e5.
5. Ai T, Yang Z, Hou H, Zhan C, Chen C, Lv W, Tao Q, Sun Z, Xia L. Correlation of Chest CT and RT-PCR Testing for Coronavirus Disease 2019 (COVID-19) in China: A Report of 1014 Cases. Radiology. 2020;296(2):E32-E40.
6. Seeliger B, Philouze G, Cherkaoui Z, Felli E, Mutter D, Pessaux P. Acute abdomen in patients with SARS-CoV-2 infection or coinfection. Langenbeck's Archives of Surgery. 2020;405(6):861-6.
7. Mayumi T, Yoshida M, Tazuma S, Furukawa A, Nishii O, Shigematsu K, et al. The practice guidelines for primary care of acute abdomen 2015. Jpn J Radiol. 2016; 34:80–115.
8. Tenner S, Baillie J, DeWitt J, Vege SS. American College of Gastroenterology Guideline: Management of Acute Pancreatitis. The Am J Gastroenterol. 2013;108(9):1400-15; 1416.
9. Banks PA, Freeman ML. Practice Parameters Committee of the American College of G. Practice Guidelines in Acute Pancreatitis. Am J Gastroenterol. 2006;101(10):2379-400.
10. Kadiyala V, Suleiman SL, McNabb-Baltar J, Wu BU, Banks PA, Singh VK. The Atlanta Classification, Revised Atlanta Classification, and Determinant-Based Classification of Acute Pancreatitis: Which Is Best at Stratifying Outcomes? Pancreas. 2016;45(4):510-5.
11. Mouch CA, Cain-Nielsen AH, Hoppe BL, Giudici MP, Montgomery JR, Scott JW, et al. Validation of the American Association for the Surgery of Trauma grading system for acute appendicitis severity. J Trauma Acute Care Surg. 2020;88(6):839-846.
12. Amirthalingam V, Low JK, Woon W, Shelat V. Tokyo Guidelines 2013 may be too restrictive and patients with moderate and severe acute cholecystitis can be managed by early cholecystectomy too. Surg Endosc. 2017;31(7):2892-2900.
13. Butters T, Grech P, Delisle TG, Ceraldi SS, Riley P, Arnaout A. An unusual presentation of Covid-19 in a patient with acute abdomen. Human Pathology Reports. 2022;28:300631
14. Gahide G, Frandon J, Vendrell J-F. COVID-19 patients presenting with afebrile acute abdominal pain. Clin Med (Lond). 2020;20(3): e4-e6.
15. Woloshin S, Patel N, Kesselheim AS. False Negative Tests for SARSCoV- 2 Infection — Challenges and Implications. N Engl J Med. 2020;383(6):e38.
16. Ahmed AOE, Badawi M, Ahmed K, Mohamed MFH. Case Report: COVID-19 Masquerading as an Acute Surgical Abdomen. Am J Trop Med Hyg. 2020;103(2):841-3.
17. Darnell MER, Subbarao K, Feinstone SM, Taylor DR. Inactivation of the coronavirus that induces severe acute respiratory syndrome, SARS-CoV. J Virol Methods. 2004;121(1):85-91.
18. Pautrat K, Chergui N. SARS-CoV-2 infection may result in appendicular syndrome: Chest CT scan before appendectomy. J Visc Surg. 2020;157(3S1):S63-S64.
19. Habeeb TAAM, Mokhtar MM, Lotfy M, Osman G, Ibrahim A, Riad M, et al. Grey Zone Appendicitis (Intermediate Risk Alvarado Score 5-6): Role of Blood Test Biomarkers to Detect Early Appendicitis and to Decrease the Incidence of Negative Appendectomy. Cost and Effectiveness. Randomized Controlled Trial. Surg Chron 2020;25(4): 339-342.
20. Habeeb TAAM, Hussain A, Schlottmann F, Kermansaravi M, Aiolfi A, Matic I, et al. Recurrent appendicitis following successful drainage of appendicular abscess in adult without interval appendectomy during COVID-19. Prospective cohort study. Int J Surg. 2022; 97:106200.
21. Weng L-M, Su X, Wang X-Q. Pain Symptoms in Patients with Coronavirus Disease (COVID-19): A Literature Review. J Pain Res. 2021;14:147-59.
22. Chau TN, Lee KC, Yao H, Tsang TY, Chow TC, Yeung YC, et al. SARSassociated viral hepatitis caused by a novel coronavirus: report of three cases. Hepatology. 2004;39(2):302-10.
23. Zhang C, Shi L, Wang FS. Liver injury in COVID-19: Management and challenges. Lancet Gastroenterol. Hepatol. 2020;5:428–30. 24. Chai X, Hu L, Zhang Y, Han W, Lu Z, Ke A, et al. Specific ACE2 Expression in Cholangiocytes May Cause Liver Damage After 2019- nCoV Infection. bioRxiv. 2020:2020.02.03.931766.
25. Zhang B, Zhou X, Qiu Y, Song Y, Feng F, Feng J, et al. Clinical characteristics of 82 cases of death from COVID-19. PLOS ONE. 2020;15(7):e0235458.
26. Habeeb TAAM, Abdouyassin M, Habib FM, Baghdadi MA. Emergency Laparoscopic Cholecystectomy with Low-Pressure Pneumo-Peritoneum in Cardiopulmonary Risk Patients: Fundus- Calot Cholecystectomy versus Calot First Cholecystectomy. Randomized Controlled Trial. Surg. Gastroenterol. Oncol. 2021 eCollection September 30 DOI: 10.21614/sgo-eC-370.
27. Yassin MA, Habeeb TAAM, Mohamed AS, Negm SM. Laparoscopic cholecystectomy is no more risky in emergent cases with in cardiopulmonary risk: Fundus-Callot cholecystectomy with low pressure pneumo-peritoneum VS open cholecystectomy. Randomized controlled trial. Surg Chron 2021;26(1):63-68.
28. Merad M, Martin JC. Pathological inflammation in patients with COVID-19: a key role for monocytes and macrophages. Nature Reviews Immunology. 2020;20(6):355-62.
29. Liemarto AK, Budiono BP, Chionardes MA, Oliviera I, Rahmasiwi A. Liver abscess with necrosis in post COVID-19: A case report. Ann Med Surg (Lond). 2021;72:103107.
30. Parenti DM, Steinberg W, Kang P. Infectious causes of acute pancreatitis. Pancreas. 1996;13(4):356-71.
31. Inamdar S, Benias PC, Liu Y, Sejpal DV, Satapathy SK, Trindade AJ. Prevalence, risk factors, and outcomes of hospitalized patients with COVID-19 presenting as acute pancreatitis. Gastroenterology. 2020;159(6):2226-2228.e2.
32. Pons S, Fodil S, Azoulay E, Zafrani L. The vascular endothelium: the cornerstone of organ dysfunction in severe SARS-CoV-2 infection. Critical Care. 2020;24(1):353.
33. Schepis T, Larghi A, Papa A, Miele L, Panzuto F, De Biase L, et al. SARS-CoV2 RNA detection in a pancreatic pseudocyst sample. Pancreatology. 2020;20(5):1011-2.
34. He L, Ding Y-q, Che X-y, Zhang Q-l, Huang Z-x, Wang H-j, et al. (Expression of the monoclonal antibody against nucleocapsid antigen of SARS-associated coronavirus in autopsy tissues from SARS patients). Di Yi Jun Yi Da Xue Xue Bao. 2003;23(11):1128-30.
35. Szatmary P, Arora A, Raraty MGT, Dunne DFJ, Baron RD, Halloran CM. Emerging phenotype of SARS-CoV2 associated pancreatitis. Gastroenterology. 2020;159(4):1551-1554.
36. anks PA, Bollen TL, Dervenis C, Kinns H. Classification of acute pancreatitis - 2012: revision of the Atlanta classification and definitions by international concensus. Ann Clin Biochemistry. 2013;50(2):182.
37. Krishna SG, Kamboj AK, Hart PA, Hinton A, Conwell DL. The Changing Epidemiology of Acute Pancreatitis Hospitalizations: A Decade of Trends and the Impact of Chronic Pancreatitis. Pancreas. 2017;46(4):482-8.
38. Liu F, Long X, Zhang B, Zhang W, Chen X, Zhang Z. ACE2 Expression in Pancreas May Cause Pancreatic Damage After SARS-CoV-2 Infection. Clin Gastroenterol Hepatol. 2020;18(9):2128-30 e2.
39. Kaafarani HMA, El Moheb M, Hwabejire JO, Naar L, Christensen MA, Breen K, et al. Gastrointestinal Complications in Critically Ill Patients With COVID-19. Ann Surg. 2020;272(2):e61-e2.
40. Helms J, Tacquard C, Severac F, Leonard-Lorant I, Ohana M, Delabranche X, et al. High risk of thrombosis in patients with severe SARS-CoV-2 infection: a multicenter prospective cohort study. Intensive Care Medicine. 2020;46(6):1089-98.
41. Phillippe HM. Overview of venous thromboembolism. Am J Manag Care. 2017;23(20 Suppl):S376-S82.
42. Guo J, Huang Z, Lin L, Lv J. Coronavirus disease 2019 (COVID-19) and cardiovascular disease: a viewpoint on the potential influence of angiotensin-converting enzyme inhibitors/angiotensin receptor blockers on onset and severity of severe acute respiratory syndrome coronavirus 2 infection. J Am Heart Assoc. 2020;9:e016219.
43. Gupta N, Zhao Y-Y, Evans CE. The stimulation of thrombosis by hypoxia. Thrombosis Research. 2019;181:77-83.
44. Tuca A, Guell E, Martinez-Losada E, Codorniu N. Malignant bowel obstruction in advanced cancer patients: epidemiology, management, and factors influencing spontaneous resolution. Cancer Manag Res. 2012;4:159-69.
45. Starnes Jr HF, Moore Jr FD, Mentzer S, Osteen RT, Steele Jr GD, Wilson RE. Abdominal pain in neutropenic cancer patients. Cancer. 1986;57(3):616-21.
46. Riva N, Donadini MP, Dentali F, Squizzato A, Ageno W. Clinical approach to splanchnic vein thrombosis: Risk factors and treatment. Thrombosis Research. 2012;130:S1-S3.
47. Serban D, Socea B, Badiu CD, Tudor C, Balasescu SA, Dumitrescu D, et al. Acute surgical abdomen during the COVID-19 pandemic: Clinical and therapeutic challenges. Exp Ther Med. 2021;21(5):519.
Full Text Sources:
Abstract:
Views: 5251

Results: The mean age of the studied group was 48.13+-11.97 years, with male predominance (66%). Medical causes of the acute abdomen were in 34% of cases, and 66% were surgical causes. Gastric causes of the acute abdomen were the most common medical causes. Referred pain from COVID-19 pneumonia was the cause of abdominal pain in 20.8% of cases. Acute appendicitis was the commonest surgical cause of acute abdomen (42.4%), mostly Grade V, followed by acute cholecystitis (11%) and biliary colic (9.8%). Acute pancreatitis was the cause in 12.7% of cases, and its causes were mainly idiopathic (48.7%).
Conclusions: Surgical causes of acute abdominal pain were more common than medical causes. The commonest medical causes of the acute abdomen during COVID-19 were gastric causes, while acute appendicitis, gallbladder diseases, and intestinal ischemia were the commonest surgical causes. Idiopathic acute pancreatitis was more common than gallstone pancreatitis, and most cases were mild. Surgery is the most common cause of death from the acute abdomen, followed by respiratory failure caused by COVID-19 and then medical causes of acute abdomen.
Trial registration: It was approved by the institutional review board (IRB) for all parts of this study by ZUMEC 152020-23.Clinical trial registration: NCT05295251.Retrospective registered.
INTRODUCTION
Coronaviruses are a large virus family that causes mild to severe respiratory infections. 2019-nCoV is a new coronavirus found in Wuhan, China. Its global spread has been rapid since its late 2019 discovery. Symptoms of exposure include dry cough, fever, and fatigue (1).
It is unclear how common gastrointestinal (GIT) symptoms are. However, as the pandemic progressed, COVID-19's clinical manifestations became more evident, and GIT symptoms more common (2). A recent meta-analysis of 47 studies with 10,890 COVID-19 patients found that 2.7% had abdominal pain, usually accompanied by diarrhoea, nausea, and vomiting (3). Since the beginning of the pandemic, COVID-19's acute abdomen has posed diagnostic and therapeutic challenges (4). Distinguishing between diseases that cause acute abdominal pain can be difficult. To avoid further mortality or morbidity, the examining physician should prioritise life-threatening conditions that may necessitate immediate surgical intervention. This study looked into the commonest causes of acute abdominal pain (medical, surgical, and oncological causes) during the COVID-19 pandemic. The study provides the first comprehensive coronavirus-era assessment of acute abdomen that a deep gap needed to be filled and helps understand this critical aspect during the COVID-19 pandemic.
MATERIALS AND METHODS
Participants and study design
This multi-centre study was conducted at our universities from May 1, 2020, to February 28, 2022, in three departments (surgery, internal medicine, and oncology). In this study, 20222 patients with acute abdominal pain with or without other GIT or respiratory symptoms were admitted to our emergency room, and 910 patients (4%) were COVID-19 positive. This study included adults over the age of 18 of both sexes who had been admitted to the emergency room or were already in the hospital but had an acute abdomen and were diagnosed with COVID-19 by a thoracic computed tomography (CT) scan (5) or reverse transcription-polymerase chain reaction (RT-PCR) of SARS-CoV-2 RNA (6). Patients under the age of 18 years who were COVID-19 negative were excluded. The patient's blood coagulation function, procalcitonin, D-dimer, and CRP (C-reactive protein) levels were measured in addition to routine laboratory tests such as complete blood count and urinalysis.
Mesurement of outcome
An acute abdomen is an acute onset of abdominal pain that requires immediate and accurate diagnosis and treatment to prevent morbidity and mortality (7). Acute pancreatitis (AP) was diagnosed according to the American College of Gastroenterology (ACG) guidelines, which requires at least two of the following three criteria: abdominal pain, elevated amylase or lipase levels (three times the upper limit of normal), and unusual imaging findings. The latter could be more important for COVID-19 patients with severe cases that require care in an intensive care unit with ventilatory support and where a clinical evaluation for abdominal symptoms might not be possible (8). Contrastenhanced computed tomography (CT) provides over 90% sensitivity and specificity for diagnosing acute pancreatitis (9). The severity of AP was classified into mild, moderately severe, and severe according to the revised Atlanta classification (RAC). Mild AP has neither organ failure nor local/systemic complications.
Moderate AP is characterised by transient organ failures or local complications, while severe AP is characterised by single or multiple persistent organ failures lasting more than 48 hours (10). The severity of acute appendicitis was based on the emergency surgery grading system of the American Association for Surgery of Trauma (11). The Tokyo Guidelines severity risk scale was used to assess acute cholecystitis (12). All cases underwent appendectomy, resected bowel, pancreatic tissues, aspirated fluid or pus, and excised lymph nodes were exposed to cytological or histopathological examination. The unusual morphological changes raise the possibility of COVID. They include apoptotic necrosis, a prominent histiocytic infiltrate, fibrinoid necrosis of blood vessels, microangiopathy, microthrombi, and increased platelet aggregation with no significant neutrophilic infiltration.
In this situation, PCR was requested on the removed specimens or fluids, including pus, for the possibility of being positive for SARS-Cov-2 RNA (nucleic acid), even if, during admission, three nose and throat swabs for COVID-19 were negative. The assay was performed according to the WHO guidelines (13).
Statistical analysis
For normally distributed continuous variables, the mean ± SD was used; for non-normally distributed continuous variables, the median was used. Percentages were used to represent categorical variables. The laboratory results were assessed to determine if they were within the normal range. SPSS version 20 was used for all statistical analyses (SPSS, Chicago, IL).
RESULTS
A flow diagram of inclusion and exclusion criteria is presented in fig. 1.

Demographic and patient characteristics showed that the mean age of the studied group was 48.13 +- 11.97 years, with male predominance (66%).
The commonest symptoms were fever (88%) and lack of appetite (85%). The associated respiratory symptoms with acute abdomen were present in 36% of patients, with a median respiratory rate of 22.41 ± 3.31/min and O2 saturation of 92.6 ± 66%. Laboratory findings include: WBCs (8.4±1.87/mm), neutrophil count (78%), CRP (41.2±1.21mg/dl), D-Dimer (5.53mg/l), ALT (98±23.7 IU/L) and lipase (89±23.8 IU/L). 37% of people who had COVID-19 had clear contact with someone who had COVID-19 before, with cardiovascular disease the most common co-morbidity (36%). The majority of cases (93%) received antiviral treatment, followed by antibiotics (79%) and glucocorticoids (42%), as shown in table 1.

Medical causes of the acute abdomen were in 34% of cases, and 66% were surgical causes. Gastric causes of the acute abdomen were the most common medical causes, with gastritis, gastroesophageal reflux diseases, and peptic ulcer (PU) diseases in 19.2%, 7%, and 17.9% of the total, respectively. Referred pain from COVID-19 pneumonia was the cause of abdominal pain in 20.8% of cases. Acute appendicitis was the commonest surgical cause of acute abdomen (42.4%), mostly Grade V, followed by acute cholecystitis (11%) and biliary colic (9.8%). Acute pancreatitis was the cause in 12.7% of cases, and its causes were mainly idiopathic (48.7%), followed by diabetic ketoacidosis (25.6%), and gallstone pancreatitis (18%) and 89.5% of pancreatitis was mild. Ischemic colitis was found in 2.5% of cases, mainly in the left colon. Small bowel ischemia was found in 2.2% of cases. Crohn's disease was found in 1.6% of cases and ulcerative colitis in 6% of cases. The commonest site of the acute abdomen was the right iliac fossa (29.8%), followed by epigastric pain (28%) and the right hypochondrium (21.3%) (tables 2–4 and figs. 2-3).
Table 5 presents the outcomes of cases admitted with acute abdomen. 29% of cases required ICU admission, with a median day of ICU admission of 11.24 +- 1.69. The mortality rate was 17%, primarily due to surgical causes (62.6%).
Discussion
Acute abdominal pain in COVID-19 patients is challenging to diagnose and, if treated late, will increase morbidity and mortality. Cholecystitis and pancreatitis cause pain in the right upper quadrant of the abdomen (RUQ), similar to right lower lobe pneumonia caused by COVID-19, especially in the absence of other typical respiratory infection symptoms (fever, oxygen requirement, and tachypnea) (14). As a result, this situation is complicated more by the lower diagnostic value of PCR of the nasal swab (15). As a result of having basal lung COVID-19 pneumonia, 64 patients in our study had upper abdominal pain. We needed to ensure we had the correct diagnosis so we did not risk exposing patients to unnecessary procedures. Radiological, laboratory and clinical diagnoses were essential items in the follow-up of these patients.

Our study has several compelling findings with important implications. We found that gastritis was the commonest medical cause of acute abdomen (59/308, 19.2%) found during the era of COVID-19, while acute appendicitis (42.4%), acute cholecystitis (11%), biliary colic (9.8%) and acute pancreatitis (12.7%) were the most common surgical causes. Furthermore, surgery was the most common cause of acute abdominal pain (66%). On the contrary, a study involving three COVID- 19 patients with acute abdominal pain found that none had any surgical complications from COVID-19.
Gastroenteritis is the commonest cause of afebrile acute abdomen (16). Another study showed 3 cases of acute abdomen diagnosed as right iliac fossa pain, right upper quadrant pain, and epigastric pain, all resolved on medications without surgical intervention (16). Our results showed a more significant number of surgical cases, mostly due to our large sample size.
The diagnosis of gastritis was based on an endoscopic diagnosis with a biopsy sent for pathological examination and PCR for the COVID-19 virus. All cases were positive for COVID-19. Antibody levels for H-pylori in the blood were done in all cases, and all cases revealed negative H-pylori infection. SARS-CoV-2 enters the stomach via saliva and is then exposed to the stomach's acidic environment. A pH of 2 affects the virus's integrity (17). We did not prescribe a proton pump inhibitor (PPI) as it decreases the stomach's acidity, allowing the virus to access the small intestine with a large viral load. All cases resolve spontaneously without complications within two weeks.
Covid-19 causes lymphopenia and neutrophilia, as well as clinical symptoms such as pain, anorexia, nausea, tenderness in the right iliac fossa, and a leftward shift in white blood cell count, mimicking appendicitis, and they concluded that CT is necessary for the diagnosis of acute appendicitis in the era of COVID-19 (18-19).

According to our study, acute appendicitis is the most frequently encountered surgical cause of acute abdomen (42.4%). Our previous study confirmed that appendicitis was more severe in the COVID era (20), possibly due to appendicular artery thrombosis or a delay in diagnosis. We found five cases that revealed negative macroscopic appendicitis during exploration. In these five cases, the only intraoperative findings were straw-coloured exudative fluid in the pelvis and enlarged mesenteric lymph nodes in the ileal mesentery.


The histological examination and PCR for COVID-19 were negative in all resected appendices but positive for mesenteric lymph node biopsy and the aspirated peritoneal fluid. We relied on CT abdomen to diagnose acute appendicitis in cases of vague abdominal ultrasonography findings. However, these negative cases showed vague CT findings, so we operated in all 5 cases. All five of these cases showed negative shifting tenderness. A study consistent with ours suggested that mesenteric lymph node enlargement could cause abdominal pain in COVID-19 patients and mimic acute appendicitis (21). Several studies have examined post-SARS-CoV2 liver disorders. SARS-CoV2 binds to ACE2 on cholangiocytes, causing cholangiocyte dysfunction and liver damage (22–24). 43 out of 99 patients had an abnormal liver function, especially higher ALT or AST levels, and 1 in 99 had severe liver damage (1). In the study of Zhang et al, the incidence of liver damage was 78% of the 82 confirmed deaths from SARS-CoV2 infection (25). We found 11% cholecystitis and 9.8% biliary colic, and most cases improved with medical treatment (73% and 81%, respectively). The remaining cases required either open or laparoscopic cholecystectomy with low-pressure pneumoperitoneum due to decreased pulmonary function associated with COVID-19 infection (26-27). Nine patients (1.5%) developed a suppurative liver abscess, which was solitary and located in the right lobe of the liver in all cases. These abscesses developed one week after developing respiratory pneumonia consistent with COVID-19 (28) with a negative bacterial culture of the aspirate from the abscess and a positive PCR for the COVID-19 virus. A study by Liemarto et al. evaluated one patient who developed two liver abscesses during the COVID-19 era, occupying most of the right lobe of the liver and treated by open drainage. There were no bacteria in the abscess pus culture (29). Acute pancreatitis (AP) is the most common GIT disease that necessitates immediate hospitalisation and is caused by obstruction of the common bile duct, alcohol abuse, hypercalcemia, viral, bacterial, and parasitic infections (30). The aetiology of acute pancreatitis is idiopathic in 15–25% of cases, and this incidence increased to 69% during the COVID-19 pandemic (31). The virus can cause acute pancreatitis through direct spread from the duodenal epithelium to the pancreatic duct and then to acinar and islet cells via ACE2 expression or by causing diffuse severe endotheliitis of the pancreatic sub-mucosal vessels with micro-ischemia damage (32). SARS-CoV can be isolated from pancreatic fluid or tissue (33-34). We found that idiopathic pancreatitis was the most common cause of pancreatitis (19 patients, 48.7%), followed by diabetic ketoacidosis (10 patients, 25.6%) and gallstone pancreatitis (7 patients, 18%). Our study found that 12.7% of patients had acute pancreatitis, mostly mild acute pancreatitis (89.5%), and responded to conservative treatment. Previous studies (35) agreed that most cases of acute pancreatitis during the COVID-19 pandemic were milder.
Four patients developed severe acute pancreatitis (10.5%). Two patients had localised pancreatic necrosis with the collection, which was diagnosed and drained using CT guidance, and the drained fluid showed positive for COVID-19 virus by PCR. The other two cases of severe pancreatitis developed total pancreatic necrosis, diagnosed by CT and treated by surgical debridement. Four patients died two weeks later due to sepsis and organ failure. In the era of the COVID-19 pandemic, the incidence of necrotizing pancreatitis was 5-10%, which is similar to our results (36). In comparison, the mortality rate of acute pancreatitis was 0.8% (37), which was higher than in our study (10% mortality rate). The higher mortality rate in our study may be due to our study's smaller sample size, associated COVID-19 infection, and many cases of chronic pancreatitis in the other study. We relied on clinical data, serum lipase, and CT abdomen to confirm the diagnosis of acute pancreatitis. The median value of serum lipase in cases of acute pancreatitis was not much increased, with a mean value of 89±23.8 IU/L.
A study by Liu and colleagues reported that 16.4% had increased lipase levels, and 7.46% showed pancreatic changes on CT imaging. Surprisingly, the serum markers were mildly elevated without abdominal pain (38).
Gastrointestinal ischemia was significantly increased in COVID-19 patients, caused mainly by adverse pharmacological effects, micro-vascular thrombotic events, and viral entero-neuropathy (39). COVID-19 can cause venous and arterial thrombosis, especially in patients with severe diseases (40). In our study, ischemic colitis, small intestine ischemia, and omental infarcts occurred in 2.4 %, 13%, and 0.3 % of our trial, respectively, with ischemic colitis developing primarily in the left colon (87%) and to a lesser extent in the right colon (13%) due to the lower vascularity of the left colon compared to the right colon. All of the cases presented with rectal bleeding in addition to pain and were diagnosed using CT angiography. Four cases of left colon gangrene (3 splenic flexure gangrene and one descending colon gangrene) necessitating immediate surgical intervention with resection with colostomy with secondary look operation after two days were intended in all cases with no further resection. All resected specimens were sent for histopathological examination and PCR for COVID-19 virus detection and were positive for the virus in all cases. One case developed sepsis and multisystem organ failure in the postoperative course, and the patient died eight days later. In 13 patients (2%), small bowel ischemia developed, and all cases necessitated surgical exploration. 11 patients developed multiple small infarcts of the small intestine, necessitating resection of the gangrenous part of the small intestine, while two patients developed gangrene of the terminal ileum and right colon, necessitating right hemicolectomy. Secondary look operations were intended in all cases, and four cases required another resection. In the postoperative course, four patients developed sepsis with multisystem organ failure at 4, 8, 12, and five days postoperatively and died. All resected specimens were sent for pathological examination and PCR for COVID-19, and all were positive. All patients were started on a therapeutic dose of heparin for systemic anticoagulation peri- and postoperatively. Our explanation for intestinal ischemia in COVID-19 is attributed to reduced venous flow due to prolonged bed rest in critically ill COVID-19 patients. SARS-CoV-2 can cause direct damage to the vessel wall and subsequent coagulopathy by binding to the ACE-2 receptors that are found abundantly on endothelial cells and may be due to hypoxia. These explanations are consistent with other studies (41-43). The acute abdomen in cancer patients is classified broadly. It includes malignant intestinal obstruction (44), typhlitis/neutropenic colitis (45), radiation enteritis, and mesenteric bowel ischemia (46). In our series, perforated colon cancer occurred in twenty-three cases (3.6%). We assumed ischemia from COVID-19 or delayed diagnosis due to patients' fear of contracting an infection or decreased endoscopic screening and diagnosis strictly limited to our centre's emergency or critical GI procedures. Complicated intestinal polyps were the causes of acute abdomen in 8 patients (1.3%), who presented with torsion of polyps (6 patients, 80%) and intussusception (2 patients, 20%). Malignant tumours of the acute abdomen were due to perforation of the left cancerous colon (19 patients), the right colon (3 patients), and gastric cancer perforation (one patient), while an obstructed cancerous colon was the cause of the acute abdomen in 4 patients (0.7%). The increased number of obstructed and perforated colon cancers was due to decreased screening of cancer colon by endoscopic examination and patient fear of contacting the hospital for GIT problems to avoid contact with COVID-19 infection. Mortality in complicated cancer patients occurred in 7 patients (7/27, 26%). One died of an obstructed cancer colon, and six patients died of perforated cancer colon. All deaths were attributed to postoperative sepsis and multisystem organ failure. No cases of acute appendicitis or pancreatitis were due to cancer. A recent study agreed with our explanation for why there are more complicated cancers, and they are getting worse (47). The current study has many advantages. Our analysis was based on a prospective study with a large sample size, eliminating bias and ensuring the findings' reliability and generalizability. Our analysis is a multipleinstitution study, so regional bias is not possible. Our findings are reliable because of rigorous data collection and diagnosis. One problem with this study is that we did not look for SARS-CoV-2 RNA in stool samples from people with COVID-19 to see if there was a link between the number and severity of digestive symptoms and the presence of viral RNA in stool samples.
CONCLUSIONS
The commonest medical causes of the acute abdomen during COVID-19 were gastric causes, while acute appendicitis, gallbladder diseases, and intestinal ischemia were the commonest surgical causes. Idiopathic acute pancreatitis was more common than gallstone pancreatitis, and most cases were mild. The most common cause of death from the acute abdomen is surgery, followed by COVID-19-related respiratory failure, and finally, medical causes of acute abdomen. Ischemia of the colon frequently occurs in the left colon, small intestine, and right colon. It was approved by the institutional review board (IRB) for all parts of this study by ZUMEC 152020-23.All patients agreed to participate and publication. Clinical trial registration: NCT05295251. All authors shared in study design, data collection, statistical analysis, data interpretation, literature search, manuscript writing and all authors shared final approval of manuscript. The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request. none. none.
REFERENCE
1. Chen N, Zhou M, Dong X, Qu J, Gong F, Han Y, et al. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. The Lancet. 2020; 395(10223):507-13.
2. Gao QY, Chen YX, Fang JY. 2019 Novel coronavirus infection and gastrointestinal tract. J Dig Dis. 2020; 21(3):125-6.
3. Sultan S, Altayar O, Siddique SM, Davitkov P, Feuerstein JD, Lim JK, et al. AGA Institute Rapid Review of the Gastrointestinal and Liver Manifestations of COVID-19, Meta-Analysis of International Data, and Recommendations for the Consultative Management of Patients with COVID-19. Gastroenterology. 2020;159(1):320-34.e27.
4. Cai M, Wang G, Zhang L, Gao J, Xia Z, Zhang P, et al. Performing abdominal surgery during the COVID-19 epidemic in Wuhan, China: a single-centred, retrospective, observational study. British Journal of Surgery. 2020;107(7):e183-e5.
5. Ai T, Yang Z, Hou H, Zhan C, Chen C, Lv W, Tao Q, Sun Z, Xia L. Correlation of Chest CT and RT-PCR Testing for Coronavirus Disease 2019 (COVID-19) in China: A Report of 1014 Cases. Radiology. 2020;296(2):E32-E40.
6. Seeliger B, Philouze G, Cherkaoui Z, Felli E, Mutter D, Pessaux P. Acute abdomen in patients with SARS-CoV-2 infection or coinfection. Langenbeck's Archives of Surgery. 2020;405(6):861-6.
7. Mayumi T, Yoshida M, Tazuma S, Furukawa A, Nishii O, Shigematsu K, et al. The practice guidelines for primary care of acute abdomen 2015. Jpn J Radiol. 2016; 34:80–115.
8. Tenner S, Baillie J, DeWitt J, Vege SS. American College of Gastroenterology Guideline: Management of Acute Pancreatitis. The Am J Gastroenterol. 2013;108(9):1400-15; 1416.
9. Banks PA, Freeman ML. Practice Parameters Committee of the American College of G. Practice Guidelines in Acute Pancreatitis. Am J Gastroenterol. 2006;101(10):2379-400.
10. Kadiyala V, Suleiman SL, McNabb-Baltar J, Wu BU, Banks PA, Singh VK. The Atlanta Classification, Revised Atlanta Classification, and Determinant-Based Classification of Acute Pancreatitis: Which Is Best at Stratifying Outcomes? Pancreas. 2016;45(4):510-5.
11. Mouch CA, Cain-Nielsen AH, Hoppe BL, Giudici MP, Montgomery JR, Scott JW, et al. Validation of the American Association for the Surgery of Trauma grading system for acute appendicitis severity. J Trauma Acute Care Surg. 2020;88(6):839-846.
12. Amirthalingam V, Low JK, Woon W, Shelat V. Tokyo Guidelines 2013 may be too restrictive and patients with moderate and severe acute cholecystitis can be managed by early cholecystectomy too. Surg Endosc. 2017;31(7):2892-2900.
13. Butters T, Grech P, Delisle TG, Ceraldi SS, Riley P, Arnaout A. An unusual presentation of Covid-19 in a patient with acute abdomen. Human Pathology Reports. 2022;28:300631
14. Gahide G, Frandon J, Vendrell J-F. COVID-19 patients presenting with afebrile acute abdominal pain. Clin Med (Lond). 2020;20(3): e4-e6.
15. Woloshin S, Patel N, Kesselheim AS. False Negative Tests for SARSCoV- 2 Infection — Challenges and Implications. N Engl J Med. 2020;383(6):e38.
16. Ahmed AOE, Badawi M, Ahmed K, Mohamed MFH. Case Report: COVID-19 Masquerading as an Acute Surgical Abdomen. Am J Trop Med Hyg. 2020;103(2):841-3.
17. Darnell MER, Subbarao K, Feinstone SM, Taylor DR. Inactivation of the coronavirus that induces severe acute respiratory syndrome, SARS-CoV. J Virol Methods. 2004;121(1):85-91.
18. Pautrat K, Chergui N. SARS-CoV-2 infection may result in appendicular syndrome: Chest CT scan before appendectomy. J Visc Surg. 2020;157(3S1):S63-S64.
19. Habeeb TAAM, Mokhtar MM, Lotfy M, Osman G, Ibrahim A, Riad M, et al. Grey Zone Appendicitis (Intermediate Risk Alvarado Score 5-6): Role of Blood Test Biomarkers to Detect Early Appendicitis and to Decrease the Incidence of Negative Appendectomy. Cost and Effectiveness. Randomized Controlled Trial. Surg Chron 2020;25(4): 339-342.
20. Habeeb TAAM, Hussain A, Schlottmann F, Kermansaravi M, Aiolfi A, Matic I, et al. Recurrent appendicitis following successful drainage of appendicular abscess in adult without interval appendectomy during COVID-19. Prospective cohort study. Int J Surg. 2022; 97:106200.
21. Weng L-M, Su X, Wang X-Q. Pain Symptoms in Patients with Coronavirus Disease (COVID-19): A Literature Review. J Pain Res. 2021;14:147-59.
22. Chau TN, Lee KC, Yao H, Tsang TY, Chow TC, Yeung YC, et al. SARSassociated viral hepatitis caused by a novel coronavirus: report of three cases. Hepatology. 2004;39(2):302-10.
23. Zhang C, Shi L, Wang FS. Liver injury in COVID-19: Management and challenges. Lancet Gastroenterol. Hepatol. 2020;5:428–30. 24. Chai X, Hu L, Zhang Y, Han W, Lu Z, Ke A, et al. Specific ACE2 Expression in Cholangiocytes May Cause Liver Damage After 2019- nCoV Infection. bioRxiv. 2020:2020.02.03.931766.
25. Zhang B, Zhou X, Qiu Y, Song Y, Feng F, Feng J, et al. Clinical characteristics of 82 cases of death from COVID-19. PLOS ONE. 2020;15(7):e0235458.
26. Habeeb TAAM, Abdouyassin M, Habib FM, Baghdadi MA. Emergency Laparoscopic Cholecystectomy with Low-Pressure Pneumo-Peritoneum in Cardiopulmonary Risk Patients: Fundus- Calot Cholecystectomy versus Calot First Cholecystectomy. Randomized Controlled Trial. Surg. Gastroenterol. Oncol. 2021 eCollection September 30 DOI: 10.21614/sgo-eC-370.
27. Yassin MA, Habeeb TAAM, Mohamed AS, Negm SM. Laparoscopic cholecystectomy is no more risky in emergent cases with in cardiopulmonary risk: Fundus-Callot cholecystectomy with low pressure pneumo-peritoneum VS open cholecystectomy. Randomized controlled trial. Surg Chron 2021;26(1):63-68.
28. Merad M, Martin JC. Pathological inflammation in patients with COVID-19: a key role for monocytes and macrophages. Nature Reviews Immunology. 2020;20(6):355-62.
29. Liemarto AK, Budiono BP, Chionardes MA, Oliviera I, Rahmasiwi A. Liver abscess with necrosis in post COVID-19: A case report. Ann Med Surg (Lond). 2021;72:103107.
30. Parenti DM, Steinberg W, Kang P. Infectious causes of acute pancreatitis. Pancreas. 1996;13(4):356-71.
31. Inamdar S, Benias PC, Liu Y, Sejpal DV, Satapathy SK, Trindade AJ. Prevalence, risk factors, and outcomes of hospitalized patients with COVID-19 presenting as acute pancreatitis. Gastroenterology. 2020;159(6):2226-2228.e2.
32. Pons S, Fodil S, Azoulay E, Zafrani L. The vascular endothelium: the cornerstone of organ dysfunction in severe SARS-CoV-2 infection. Critical Care. 2020;24(1):353.
33. Schepis T, Larghi A, Papa A, Miele L, Panzuto F, De Biase L, et al. SARS-CoV2 RNA detection in a pancreatic pseudocyst sample. Pancreatology. 2020;20(5):1011-2.
34. He L, Ding Y-q, Che X-y, Zhang Q-l, Huang Z-x, Wang H-j, et al. (Expression of the monoclonal antibody against nucleocapsid antigen of SARS-associated coronavirus in autopsy tissues from SARS patients). Di Yi Jun Yi Da Xue Xue Bao. 2003;23(11):1128-30.
35. Szatmary P, Arora A, Raraty MGT, Dunne DFJ, Baron RD, Halloran CM. Emerging phenotype of SARS-CoV2 associated pancreatitis. Gastroenterology. 2020;159(4):1551-1554.
36. anks PA, Bollen TL, Dervenis C, Kinns H. Classification of acute pancreatitis - 2012: revision of the Atlanta classification and definitions by international concensus. Ann Clin Biochemistry. 2013;50(2):182.
37. Krishna SG, Kamboj AK, Hart PA, Hinton A, Conwell DL. The Changing Epidemiology of Acute Pancreatitis Hospitalizations: A Decade of Trends and the Impact of Chronic Pancreatitis. Pancreas. 2017;46(4):482-8.
38. Liu F, Long X, Zhang B, Zhang W, Chen X, Zhang Z. ACE2 Expression in Pancreas May Cause Pancreatic Damage After SARS-CoV-2 Infection. Clin Gastroenterol Hepatol. 2020;18(9):2128-30 e2.
39. Kaafarani HMA, El Moheb M, Hwabejire JO, Naar L, Christensen MA, Breen K, et al. Gastrointestinal Complications in Critically Ill Patients With COVID-19. Ann Surg. 2020;272(2):e61-e2.
40. Helms J, Tacquard C, Severac F, Leonard-Lorant I, Ohana M, Delabranche X, et al. High risk of thrombosis in patients with severe SARS-CoV-2 infection: a multicenter prospective cohort study. Intensive Care Medicine. 2020;46(6):1089-98.
41. Phillippe HM. Overview of venous thromboembolism. Am J Manag Care. 2017;23(20 Suppl):S376-S82.
42. Guo J, Huang Z, Lin L, Lv J. Coronavirus disease 2019 (COVID-19) and cardiovascular disease: a viewpoint on the potential influence of angiotensin-converting enzyme inhibitors/angiotensin receptor blockers on onset and severity of severe acute respiratory syndrome coronavirus 2 infection. J Am Heart Assoc. 2020;9:e016219.
43. Gupta N, Zhao Y-Y, Evans CE. The stimulation of thrombosis by hypoxia. Thrombosis Research. 2019;181:77-83.
44. Tuca A, Guell E, Martinez-Losada E, Codorniu N. Malignant bowel obstruction in advanced cancer patients: epidemiology, management, and factors influencing spontaneous resolution. Cancer Manag Res. 2012;4:159-69.
45. Starnes Jr HF, Moore Jr FD, Mentzer S, Osteen RT, Steele Jr GD, Wilson RE. Abdominal pain in neutropenic cancer patients. Cancer. 1986;57(3):616-21.
46. Riva N, Donadini MP, Dentali F, Squizzato A, Ageno W. Clinical approach to splanchnic vein thrombosis: Risk factors and treatment. Thrombosis Research. 2012;130:S1-S3.
47. Serban D, Socea B, Badiu CD, Tudor C, Balasescu SA, Dumitrescu D, et al. Acute surgical abdomen during the COVID-19 pandemic: Clinical and therapeutic challenges. Exp Ther Med. 2021;21(5):519.
Full Text Sources:
Abstract:
Views: 5251

Watch Video Articles
For Authors
Journal Subscriptions

Current Issue
Dec 2025
Supplements
Instructions for authors
Online submission
Contact
ISSN: 2559 - 723X (print)
e-ISSN: 2601 - 1700 (online)
ISSN-L: 2559 - 723X
Journal Abbreviation: Surg. Gastroenterol. Oncol.
Surgery, Gastroenterology and Oncology (SGO) is indexed in:
e-ISSN: 2601 - 1700 (online)
ISSN-L: 2559 - 723X
Journal Abbreviation: Surg. Gastroenterol. Oncol.
Surgery, Gastroenterology and Oncology (SGO) is indexed in:
- SCOPUS
- EBSCO
- DOI/Crossref
- Google Scholar
- SCImago
- Harvard Library
- Open Academic Journals Index (OAJI)
Open Access Statement
Surgery, Gastroenterology and Oncology (SGO) is an open-access, peer-reviewed online journal published by Celsius Publishing House. The journal allows readers to read, download, copy, distribute, print, search, or link to the full text of its articles.
Surgery, Gastroenterology and Oncology (SGO) is an open-access, peer-reviewed online journal published by Celsius Publishing House. The journal allows readers to read, download, copy, distribute, print, search, or link to the full text of its articles.
Journal Metrics
Time to first editorial decision: 25 days
Rejection rate: 61%
CiteScore: 0.2
Time to first editorial decision: 25 days
Rejection rate: 61%
CiteScore: 0.2

Meetings and Courses in 2025
Meetings and Courses in 2024
Meetings and Courses in 2023
Meetings and Courses in 2022
Meetings and Courses in 2021
Meetings and Courses in 2020
Meetings and Courses in 2019
Verona expert meeting 2019

Surgery, Gastroenterology and Oncology applies the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits readers to copy and redistribute the material in any medium or format, remix, adapt, build upon the published works non-commercially, and license the derivative works on different terms, provided the original material is properly cited and the use is non-commercial. Please see: https://creativecommons.org/licenses/by-nc/4.0/
Publisher’s Note:
The opinions, statements, and data contained in article are solely those of the authors and not of Surgery, Gastroenterology and Oncology journal or the editors. Publisher and the editors disclaim responsibility for any damage resulting from any ideas, instructions, methods, or products referred to in the content.
 IASGO Society News
IASGO Society News