Surgery, Gastroenterology and Oncology

|
|
Background: Acute pancreatitis (AP) is a potentially fatal disease that can range from mild and self-limiting to rapidly progressive, leading to multiple organ failure and death. Aim: This study aimed to compare various severity scores in predicting outcomes in AP.
Methods: This is an observational cross-sectional study conducted from June 2018 to December 2019. Adults diagnosed with AP were included in the study.
Results: A total of 57 patients were included, with mean age of patients were 40.07 years. Among them, 36.84% had mild, 35.08% had moderately severe, and 28.07% had severe AP (SAP), with a 10.52% mortality rate in the SAP. Alcohol was the most common cause (59.64%), and abdominal pain was the most frequent symptom (100%). In predicting severity, APACHE II at 48 hours had the highest AUROC of 0.980, with a cut-off score of ³9 offering the best sensitivity and specificity compared to other scores. For predicting mortality, APACHE II at 48 hours also had the highest AUROC of 0.992, with a cut-off score of ³16 providing the best predictive accuracy.
Conclusion: The APACHE II at 48 hours is the best predictor of SAP and mortality and CRP is a reliable parameter, not a score in predicting severity but is an inferior parameter in predicting mortality.
Introduction
Acute pancreatitis (AP) is a rapid-onset inflammation of the pancreas characterized by the early activation of pancreatic enzymes, which causes the pancreas to begin digesting its own tissue. This condition has a general mortality rate of 3-5%. (1-3). In most cases, AP is mild and resolves on its own without complications. However, approximately 20 % of AP patients experience moderate or severe disease characterized by local complications (pancreatic necrosis, fluid collections, splanchnic vein thrombosis, and pseudoaneurysms) and organ failure (respiratory, cardiovascular, and renal). Multi-organ failure (MOF) involving <2 organ systems and persistent (<48 h) organ failure (POF) are associated with a higher risk of death and local complications, especially pancreatic necrosis (4).
Severe AP is associated with mortality rates ranging from 20-40% (3,5,6). Although AP is a common clinical condition, there is a lack of prevalence data from India (3). In contrast, the incidence of AP has been reported to be higher in countries such as the USA, Finland, and Scotland, with rates of 49.3, 46.6, and 41.9 per 100,000 population, respectively (7). In the last ten years, the mortality rate linked to severe AP has not increased despite the rising number of AP cases. This is likely due to improvements such as earlier detection of severe cases, reduced use of invasive procedures early on, implementation of enteral nutrition, and better intensive care management (5). As a result, promptly identifying patients at risk for severe AP is crucial to ensure they receive appropriate care and timely intervention (8-10).
Although numerous prognostic scoring systems have been introduced since the 1970s to forecast severe AP, their clinical usefulness remains limited, as they typically yield low positive predictive values (PPV) between 11% and 23% (11). These scoring systems often require more than 24 hours for a complete assessment of disease severity, potentially delaying appropriate care. Additionally, many of these scoring systems were initially developed to predict mortality, not organ failure. However, with the decline in AP-related mortality over the past decade, organ failure has emerged as a more clinically relevant outcome for assessing disease severity (12).
Initial assessment of AP severity should include an evaluation of fluid status, organ failure (such as cardiovascular, respiratory, or renal), and the calculation of the acute physiology and chronic health evaluation (APACHE II) score and systemic inflammatory response syndrome (SIRS) score (13,14). While the measurement of amylase and lipase levels is useful for diagnosing pancreatitis, serial measurements do not help in predicting disease severity, prognosis, or guiding management (15). Early recognition of cases that may progress to severe pancreatitis is crucial for initiating timely and appropriate treatment (16-18). Although several classification systems, including SIRS, Glasgow, Ranson, and APACHE II, are available for assessing the severity of AP, they are not well-validated for predicting mortality. Early organ failure remains a strong predictor of disease severity and the need for intensive care treatment (16-18).
In light of these challenges, this study aimed to compare various scoring systems in predicting the severity and mortality of AP and to assess the correlation between C-reactive protein (CRP) levels and the severity of the disease.
Materials and Methods
Study Design and Ethical Consideration
This is an observational cross-sectional study conducted over 18 months, from June 2018 to December 2019. The study design and methodology received approval from the institutional ethics committee and review board. Written informed consent was obtained from all the patients prior to the commencement of the study.
Inclusion Criteria and Exclusion Criteria
Patients of either sex, above 18 years of age, diagnosed with of AP were included in the study. Patients with a history of chronic pancreatitis (characterized by chronic abdominal pain, maldigestion with weight loss, or radiological evidence of the condition) and those who are immunocompromised were excluded from the study.
Data Collection
Data was collected from patients, including their demographic profile (age and sex), duration of illness, symptoms at presentation, co-morbid conditions, medications, past history of AP, and history of alcohol use or smoking.
Study Endpoints
The study endpoints were to investigate the etiology and clinical profile of patients with AP, and to assess and analyze different severity scores and their correlation with patient outcomes.
Evaluation of Severity Scores
To evaluate the performance of different severity scores, the area under the receiver operating characteristics (AUROC) curve was computed for each score. The AUROC provides a measure of the ability of the score to discriminate between patients with different outcomes (e.g., survivors vs. non-survivors). Optimal cut-off scores were determined based on the AUROC analysis. From these cut-off points, the positive predictive value (PPV), negative predictive value (NPV), sensitivity, and specificity of each score were calculated.
Statistical Analysis
Data were analyzed using SPSS V20 software. Descriptive analysis of the continuous variables was done. The mean values of variables among Mild/moderate pancreatitis were compared with those of severe pancreatitis, non-survivors were compared with those of survivors and the results were expressed as mean (SD). Comparison of two independent variables with normal distribution was performed using Students test (t) test. Independent variables with non- normal distribution was performed using Mann Whitney U test. Multivariate analysis of the variables was performed to know the importance of each variable in predicting the outcome. p<0.05 was considered statistically significant. Area under receiver operating characteristics (AUROC) curve was computed to determine optimal cut-off scores for analyzing the performance of different scores.
Results
A total of 57 patients were included in the study, with mean (SD) age of the patients were 40.07 (12.18) years. Patients in this study had age between 18 -70 years, of which 71.93% were male and 28.07% were female. Among the study population, 36.84% patients had mild, 35.08% had moderately severe and 28.07% patients had severe pancreatitis, 10.52% patients with severe pancreatitis expired. The mean (SD) age of patients was 41.98 (12.401) years (Range: 18- 70 years) among mild/moderately severe pancreatitis and 35.19 (10.41) years (Range: 20-59 years) among severe pancreatitis.
Figure 1 - Etiological factors according to the severity
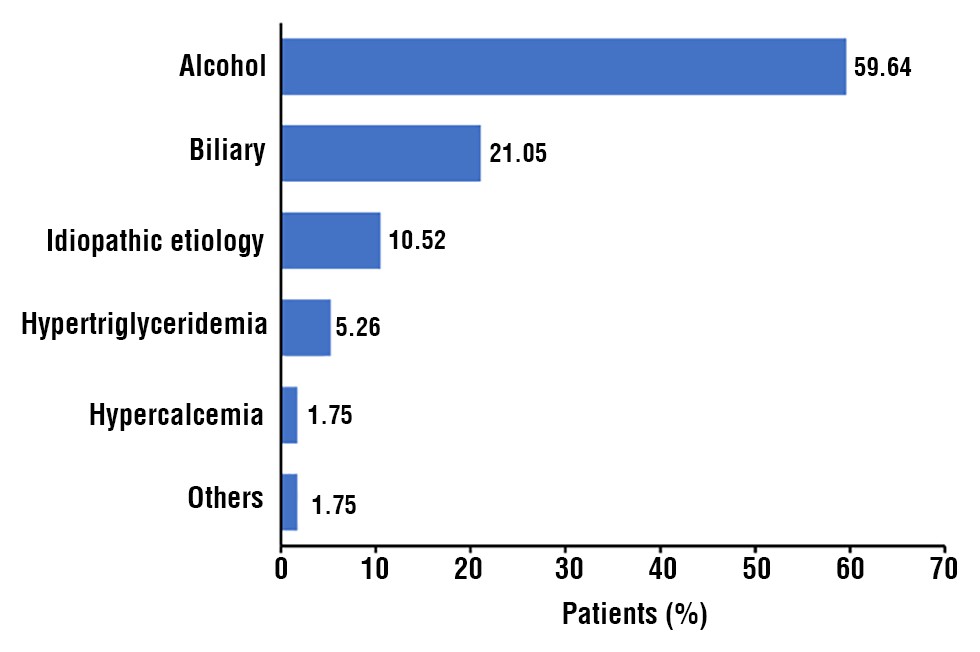
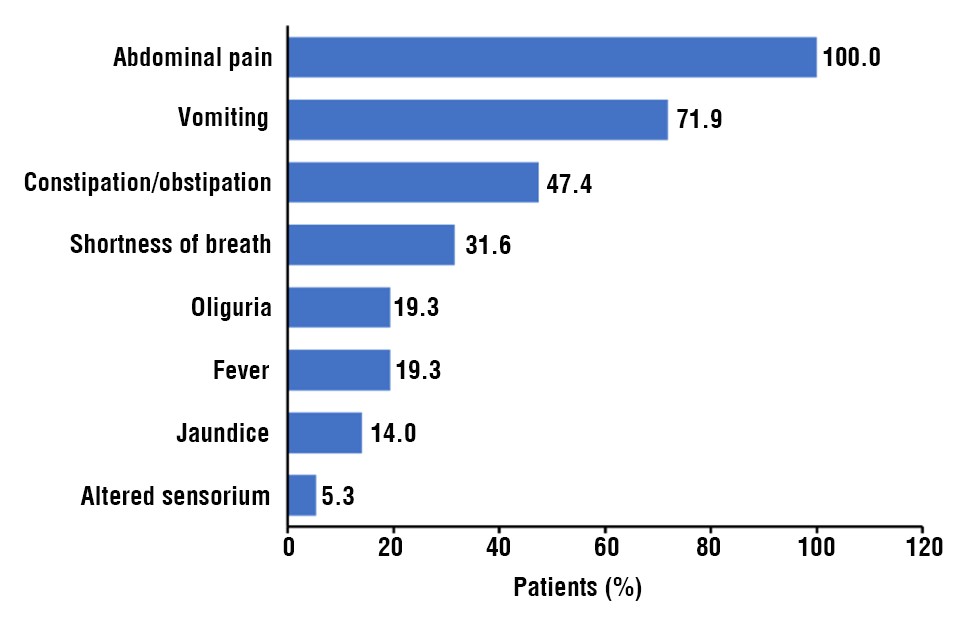
Alcohol (59.64%) was the most common etiology in patients, followed by biliary (21.05%), hyper-triglyceridemia (5.26%), idiopathic etiology (10.52%), and hypercalcemia (1.75%) (fig. 1). Pain in abdomen (100.00%) was the commonest symptom at presentation seen in all the patients followed by vomiting (71.9%), constipation/obstipation (47.4%), shortness of breath (31.6%), fever (19.3%), jaundice (14.0%), oliguria (9.8%), and altered sensorium (5.3%) (fig. 2).
Figure 2 - Distribution of symptoms at presentation
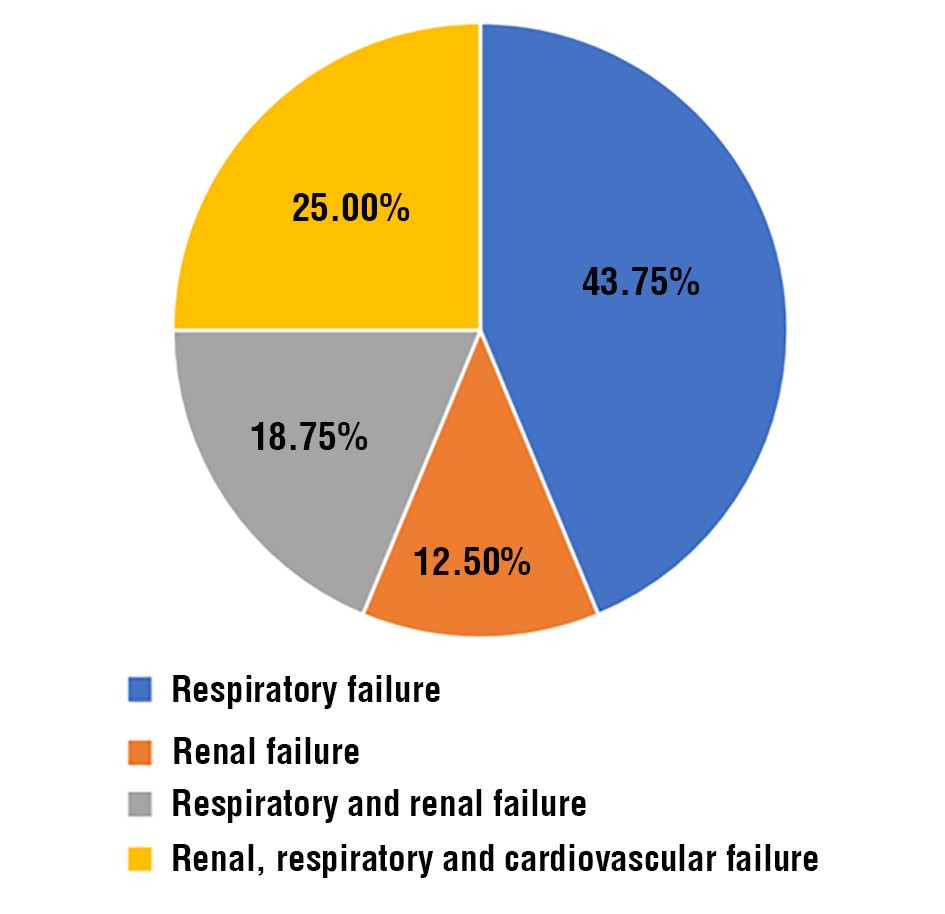
The mean respiratory rate was significantly higher in severe compared to mild/moderately severe (32.31 (6.36) cpm vs 21.66 (4.07) cpm, p<0.001) and non-survivor group compared to survivor 30.67 (3.50) cpm vs 23.94 (6.74) cpm, p=0.002). The laboratory parameter including hemoglobin, hematocrit, total leucocyte count, platelet count, blood urea on day 0, serum creatinine on day 0, serum LDH levels, serum calcium levels were found to be statistically significant between mild/moderate pancreatitis and severe pancreatitis groups (table 1). Among study population, 28.07% patients had persistent organ failure. Respiratory failure (43.75%) was most common, followed by combined renal, respiratory and cardiovascular failure (25%), combined renal and respiratory failure (18.75%), and isolated renal failure (12.5%) (fig. 3).
Table 1 - Lab parameters according to severity of pancreatitis
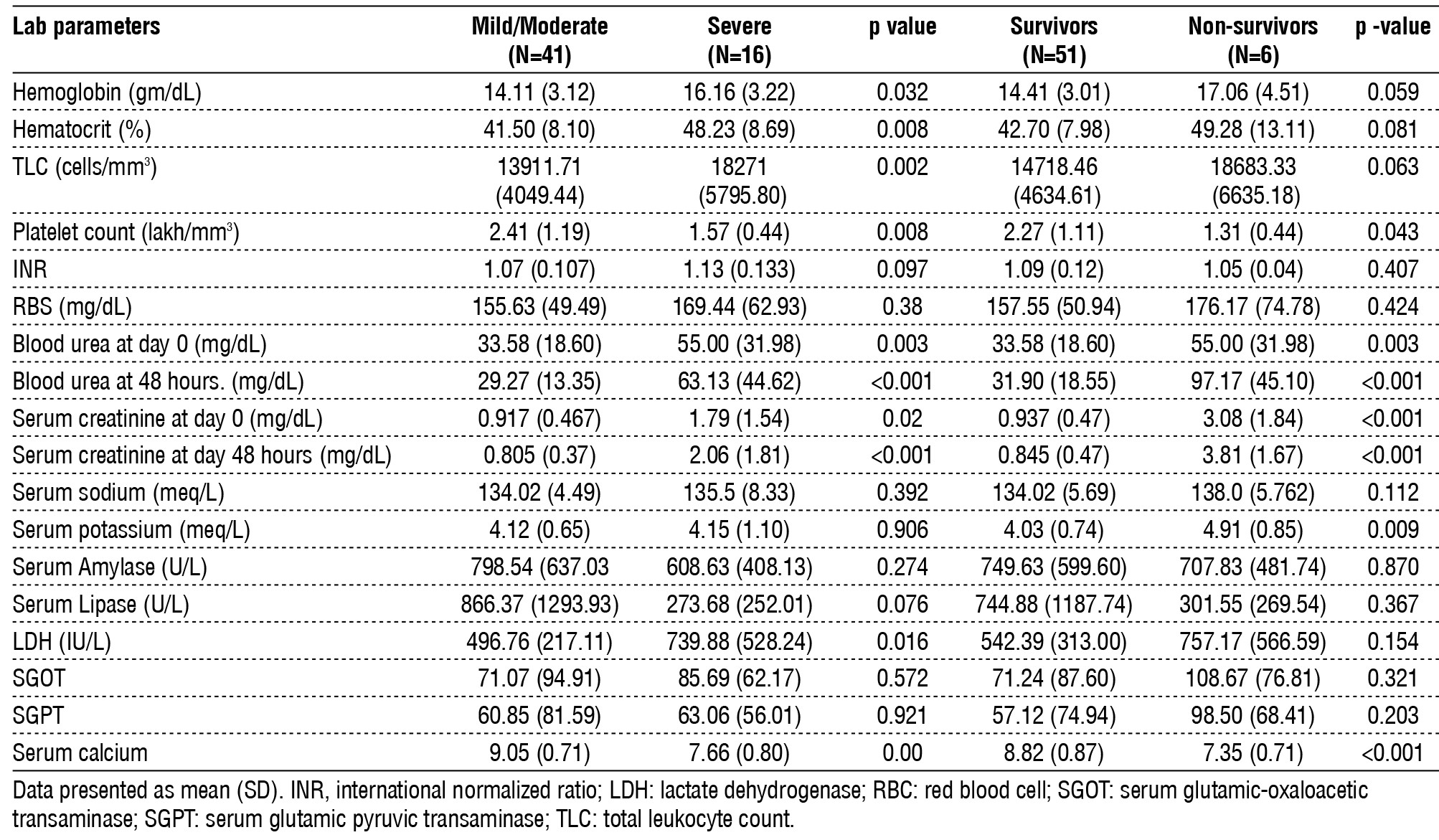
Figure 3 - Distribution of organ failure
At the end of 8 weeks, pseudocyst was seen in 2 patients, WON in 8 patients, new onset diabetes mellitus in 1 patient, splenic and portal vein thrombosis in 1 patient and death in 6 patients. endoscopic interventions were done in 2 patients of WON. The mean (SD) of bedside index of severity in acute pancreatitis (BISAP) [2.63 (0.806) vs 1.39 (0.703), p<0.001], Ranson score [ 4.75 (1.80) vs 1.98 (1.129), p<0.001], APACHE II score at 48 hours [ 15.71 (5.26) vs 4.85 (2.83), p<0.001], Imrie score [ 3.80 (1.014) vs 1.87 (1.105), p<0.001], CRP at 48 hours [ 104.13 (39.75) vs 64.69 (62.35), P=0.027] were significantly higher among patients with severe pancreatitis compared to mild/moderate group (table 2).
Table 2 - Comparison of severity scores and CRP levels in patients with AP

The mean score of BISAP [ 2.67 (1.03) vs. 1.63(0.848), p=0.007], ranson [ 5.33 (1.86) vs. 2.45 (1.59), p<0.001] APACHE II at 48 hours [ 21.75(3.86) vs. 6.51 (4.48), p<0.001], imrie [4.60(1.14) vs. 2.18 (1.20), p<0.001] were significantly higher non- survivors compared to survivors (table 2). The APACHE II at 48 hours has the highest AUROC compared to BISAP, ranson, imrie severity scores (0.980 vs 0.861 vs. 0.880 vs. 0.908). The APACHE II at 48 at score ≥9 has comparable sensitivity and specificity in predicting severity better than other scores. C-reactive protein had very low AUROC and hence is not a good predictor of severity (table 3).
Table 3 - Comparison of BISAP, Ranson Score, APACHE II score (on admission & at 48 hours), Imrie’s score, CRP level (on admission and at 48 hours) in predicting severity.

The APACHE II score at 48 hours, with a cutoff of ≥16, had the highest AUROC of 0.988, demonstrating the best predictive accuracy for mortality with a sensitivity of 100% and specificity of 95%. The imrie score, at a cutoff of ≥ 3, had an AUROC of 0.928, with sensitivity and specificity of 100% and 59%, respectively. Ranson's score, at a cutoff of ≥ 3, had an AUROC of 0.873, with 83% sensitivity and 63% specificity. The BISAP score, at a cutoff of ≥ 2, had an AUROC of 0.783, with sensitivity and specificity of 83% and 44% respectively. C-reactive protein at 48 hours, with a cutoff of ≥ 88, had an AUROC of 0.724, with sensitivity and specificity of 100% and 66%, but was not a strong predictor of mortality due to its lower AUROC compared to the other scores (table 4).
Table 4 - Comparison of BISAP, Ranson Score, APACHE II score (on admission & at 48 hours), Imrie’s score, CRP level (on admission and at 48 hours) in predicting mortality.

Discussion
Acute pancreatitis was an inflammatory condition of the pancreas and may have a variable severity. Most of the patients have mild disease with minimal morbidity, and the rest of the patients have 10%-20% of mortality in severe AP (19). In this study, APACHE II at 48 hours is the best predictor of severe AP and mortality. The BISAP being simple bedside score, is still a reliable predictor of severity compared to other severity scores. The CRP is a reliable marker in predicting severity but is an inferior marker in predicting mortality.
In present study, majority patients were the males. Similar finding demonstrated by Robert et al. and Shakya P et al. (20,21), incidence of AP overall was significantly higher among men than women. In contrast to this, retrospective study showed that there was predominance of AP in females compared to males (22). This might be due increased prevalence of biliary pancreatitis in females.
In current study, alcohol was the most common etiology followed by biliary, hypertriglyceridemia, idiopathic etiology, and hypercalcemia. In line with current study, Mukherjee D et al, reported alcohol as the commonest etiology, followed by idiopathic causes, and obstructive causes (23). In contrast, one study showed that gallstones were the most common cause of AP, followed by alcohol, idiopathic, post- endoscopic retrograde cholangiopancreatography, and miscellaneous causes (24). Another study reported that biliary was the commonest cause, followed by alcoholism, hypertriglyceridemia, and
the others (25). The most frequent cause of pancreatitis, according to a comparable study by Robert
et al., was gallstones. Other prevalent causes were alcohol, hyperlipidemia, abdominal trauma, hypercalcemia, starvation, pancreatic cancers, and cystic fibrosis (20).
Pain in abdomen was the commonest symptom at presentation seen in all the patients followed by vomiting, constipation/obstipation, shortness of breath, fever, jaundice. Other studies (22,26-28) also corroborate that the most common presentation was abdominal pain followed by vomiting, abdominal distension, fever and jaundice.
The present study revealed that 36.84% patients had mild, 35.08% had moderately severe and 28.07% patients had severe pancreatitis, 10.52% patients with severe pancreatitis expired. In contrast, the recent study found that the majority of patients (71.20%) had mild pancreatitis, 23.71% had moderate pancreatitis, and 5.09% had severe pancreatitis (29). Another study reported that 63.7% patients were mild, 25.8% were moderate and 11.0 % severe (30).
Organ failure is the main determinant of severity and a major cause of early mortality. In the present study, 36.06% patients developed persistent organ failure, of which respiratory failure was most common, followed by combined renal, respiratory and cardio-vascular failure then by combined renal and respiratory failure, isolated renal failure found in of patients. In line with current study reported 49.6% patients had severe AP, respiratory failure (59%) was the most common organ failure followed by respiratory failure with renal failure (31%) and respiratory and renal with cardiac failure (10%) (24). In contrast to the present study, the other study found that respiratory plus cardiac failure was more common, followed by respiratory and renal failure, and then respiratory, renal, and cardiovascular failure (33).
In predicting severity, APACHE II at 48 hours had the highest AUROC with a cut-off of ≥9 compared to other severity scores. Similarly, Larvin et al. found that the APACHE II score had higher predictive accuracy for severe AP than the Ranson score and could be calculated at any time point (34). In current study, APACHE II (at 48 hours) score had superior predictive value of mortality with an AUROC of 0.988 followed by APACHE II (on admission), Imrie score, Ranson’s score, BISAP and CRP 48 hours & on admission. Similarly, a study by Khanna et al showed that APACHE II was more accurate in predicting mortality in AP with AUC- 0.86 (35). Similarly, one study (36) reported that APACHE II is a useful prognostic scoring system for predicting the severity of AP. In contrast to this, an observational study found that changes in serum procalcitonin levels and APACHE II scores between admission and 48 hours are closely correlated and can effectively predict mortality in acute pancreatitis. Notably, the change in serum procalcitonin levels was a more reliable indicator than the change in APACHE II scores among survivors (37).
C-reactive protein on admission and at 48 hours were a poor predictor of mortality. The calculated AUROC for CRP on admission and at 48 hours for predicting was 0.628 and 0.724 respectively & hence was a poor marker in predicting outcome in AP. Similarly, Aaron D. Stirling et al showed that a CRP rise of >90 mg/dL from admission or an absolute value of >190 mg/dL at 48 h predicts severe disease with the greatest accuracy (38). In line with study, Khanna et al demonstrated that CRP was a good predictor of severity (AUC-0.91) but not a good predictor of mortality (AUC-0.75) (35). Similarly, other studies reported the showed that CRP levels were not statistically significant in non- survivors compared to survivors (39,40).
Limitations
A limitation of study was the small sample size, as it was conducted at a single center. A similar multi-center study would provide a larger sample size, allowing for a better determination of the cut-off values for various severity scores in estimating how severe acute pancreatitis may become and its associated mortality risk. Additionally, other parameters that need to be evaluated include inflammatory markers such as IL-6, which could not assess in this study due to financial constraints and limited resources.
Conclusion
Blood urea, serum creatinine, and serum calcium levels are key indicators to evaluate disease severity and mortality outcomes in AP. Respiratory failure is the most frequent organ failure observed in AP. While CRP is a dependable marker for assessing the severity of the condition, it is less effective in predicting mortality. Scoring systems such as BISAP, Ranson's criteria, APACHE II (both on admission and at 48 hours), and Imrie scores are significantly higher in patients with severe AP and those who do not survive. Of these, APACHE II at 48 hours is the most accurate for predicting both severity and mortality, with BISAP also being a reliable and simple bedside tool for these predictions.
Acknowledgements
Authors would like to thank Shital Kolhe for assistance in manuscript development.
Conflict of Interest
All authors declare no conflicts of interest.
Funding: None
References
1. Heckler M, Hackert T, Hu K, Halloran CM, Buchler MW, Neoptolemos JP. Severe acute pancreatitis: surgical indications and treatment. Langenbecks Arch Surg. 2021;406(3):521-535.
2. Li C-l, Jiang M, Pan C-q, Li J, Xu L-g. The global, regional, and national burden of acute pancreatitis in 204 countries and territories, 1990–2019. BMC Gastroenterol. 2021;21(1):332.
3. Thoeni RF. The revised Atlanta classification of acute pancreatitis: its importance for the radiologist and its effect on treatment. Radiology. 2012;262(3):751-764.
4. Banks PA, Bollen TL, Dervenis C, Gooszen HG, Johnson CD, Sarr MG , et al. Classification of acute pancreatitis--2012: revision of the Atlanta classification and definitions by international consensus. Gut. 2013;62(1):102-111.
5. Sarri G, Guo Y, Iheanacho I, Puelles J. Moderately severe and severe acute pancreatitis : a systematic review of the outcomes in the USA and European Union-5. BMJ Open Gastroenterol. 2019; 6(1): e000248.
6. Paragomi P, Tuft M, Pothoulakis I, Singh VK, Stevens T, Nawaz H, et al. Dynamic changes in the pancreatitis activity scoring system during hospital course in a multicenter, prospective cohort. J Gastroenterol Hepatol. 2021;36(9):2416-2423.
7. Kumar N, GT S. Evaluation of patients who are clinically suspected for the case of acute pancreatitis by MCTSI. Int J Radiol Diagn Imaging. 2021;4(1):34-37.
8. Koutroumpakis E, Wu BU, Bakker OJ, Dudekula A, Singh VK, Besselink MG , et al. Admission Hematocrit and Rise in Blood Urea Nitrogen at 24 h Outperform other Laboratory Markers in Predicting Persistent Organ Failure and Pancreatic Necrosis in Acute Pancreatitis: A Post Hoc Analysis of Three Large Prospective Databases. Am J Gastroenterol. 2015;110(12):1707-1716.
9. Mounzer R, Langmead CJ, Wu BU, Evans AC, Bishehsari F, Muddana V , et al. Comparison of existing clinical scoring systems to predict persistent organ failure in patients with acute pancreatitis. Gastroenterology. 2012;142(7):1476-1482; quiz e1415-1476.
10. Langmead C, Lee PJ, Paragomi P, Greer P, Stello K, Hart PA , et al. A Novel 5-Cytokine Panel Outperforms Conventional Predictive Markers of Persistent Organ Failure in Acute Pancreatitis. Clin Transl Gastroenterol 2021, 12(5):e00351.
11. Gurusamy KS, Debray TP, Rompianesi G. Prognostic models for predicting the severity and mortality in people with acute pancreatitis. Cochrane Database of Systematic Reviews. 2018;5:CD013026.
12. Wu BU. Prognosis in acute pancreatitis. CMAJ. 2011;183(6):673-7.
13. Forsmark CE, Baillie J, Practice AGAIC, Economics C, Board AGAIG: AGA Institute technical review on acute pancreatitis. Gastroenterology. 2007;132(5):2022-2044.
14. Tenner S, Baillie J, DeWitt J, Vege SS, American College of G. American College of Gastroenterology guideline: management of acute pancreatitis. Am J Gastroenterol. 2013;108(9):1400-1415; 1416.
15. Vengadakrishnan K, Koushik AK. A study of the clinical profile of acute pancreatitis and its correlation with severity indices. Int J Health Sci (Qassim). 2015;9(4):410-417.
16. Working Group IAPAPAAPG: IAP/APA evidence-based guidelines for the management of acute pancreatitis. Pancreatology. 2013;13(4 Suppl 2):e1-15.
17. Windsor JA: A better way to predict the outcome in acute pancrea-titis? Am J Gastroenterol. 2010;105(7):1671-1673.
18. Wu BU, Banks PA. Clinical management of patients with acute pancreatitis. Gastroenterology. 2013;144(6):1272-1281.
19. Banks PA. Acute Pancreatitis: Landmark Studies, Management Decisions, and the Future. Pancreas. 2016;45(5):633-640.
20. Roberts SE, Akbari A, Thorne K, Atkinson M, Evans PA. The incidence of acute pancreatitis: impact of social deprivation, alcohol consumption, seasonal and demographic factors. Aliment Pharmacol Ther 2013, 38(5):539-548.
21. Shayka P, Singh RK, Negi D. Clinico-epidemiological study of acute pancreatitis in tertiary care center. Int J Acad Med Pharm. 2023; 5(3):349-353.
22. Yadav AP, Raya A, Rai BK, Shah. Clinical Profile of Acute Pancreatitis. Janaki Medical College Journal of Medical Science. 2022;10(2):65-69.
23. Mukherjee D, Bhakta S, Lahiry S, Sinha R. Demographic profile of acute pancreatitis in Eastern India: a single centre experience. Asian Journal of Medical Sciences. 2017;8(6):24-29.
24. Vasudevan S, Goswami P, Sonika U, Thakur B, Sreenivas V, Saraya A. Comparison of various scoring systems and biochemical markers in predicting the outcome in acute pancreatitis. Pancreas. 2018; 47(1):65-71.
25. Zheng Y, Zhou Z, Li H, Li J, Li A, Ma B, et al. A multicenter study on etiology of acute pancreatitis in Beijing during 5 years. Pancreas. 2015;44(3):409-414.
26. Ahmed KU, Ahad MA, Alim MA, Ekram AS. Clinical profile of acute pancreatitis in a teaching hospital. Bangladesh Medical Journal Khulna. 2016;49(1-2):7-12.
27. Das SK, Das S: Clinical profile of patients with acute pancreatitis in a tertiary care centre in Tripura: A retrospective study. Asian Journal of Medical Sciences. 2020;11(6):96-100.
28. Kurrey L, Patel V, Gaharwar A, Jayant V, Pandre S, Kumar S. Clinical study of pancreatitis and its management: A prospective study. IJSS J Surg. 2017;3(3):27-33.
29. Saxena R, Kumar S, Nafe Z, Chattoraj A, Chauhan S. Clinical, Biochemical, and Radiological Correlation in the Severity of Acute Pancreatitis: A Retrospective Study. Cureus. 2023;15(2):e34996.
30. Pando E, Alberti P, Mata R, Gomez MJ, Vidal L, Cirera A , et al. Early Changes in Blood Urea Nitrogen (BUN) Can Predict Mortality in Acute Pancreatitis: Comparative Study between BISAP Score, APACHE-II, and Other Laboratory Markers-A Prospective Observational Study. Can J Gastroenterol Hepatol 2021. 2021: 6643595.
31. Wu BU, Johannes RS, Sun X, Conwell DL, Banks PA: Early changes in blood urea nitrogen predict mortality in acute pancreatitis. Gastroenterology. 2009;137(1):129-135.
32. Lin S, Hong W, Basharat Z, Wang Q, Pan J, Zhou M. Blood Urea Nitrogen as a Predictor of Severe Acute Pancreatitis Based on the Revised Atlanta Criteria: Timing of Measurement and Cutoff Points. Can J Gastroenterol Hepatol. 2017;2017:9592831.
33. Parniczky A, Kui B, Szentesi A, Balazs A, Szucs A, Mosztbacher D, et al. Prospective, Multicentre, Nationwide Clinical Data from 600 Cases of Acute Pancreatitis. PLoS One. 2016;11(10): e0165309.
34. Larvin M, McMahon MJ. APACHE-II score for assessment and monitoring of acute pancreatitis. Lancet. 1989;2(8656):201-205.
35. Khanna AK, Meher S, Prakash S, Tiwary SK, Singh U, Srivastava A , et al. Comparison of Ranson, Glasgow, MOSS, SIRS, BISAP, APACHE-II, CTSI Scores, IL-6, CRP, and Procalcitonin in Predicting Severity, Organ Failure, Pancreatic Necrosis, and Mortality in Acute Pancreatitis. HPB Surg. 2013;2013:367581.
36. Cho JH, Kim TN, Chung HH, Kim KH. Comparison of scoring systems in predicting the severity of acute pancreatitis. World J Gastroenterol. 2015;21(8):2387-2394.
37. Choudhuri AH, Duggal S, Biswas PS, Uppal R. A Comparison of Acute Physiology and Chronic Health Evaluation II Score and Serum Procalcitonin Change for Predicting Mortality in Acute Pancreatitis. Indian J Crit Care Med. 2020;24(3):190-194.
38. Stirling AD, Moran NR, Kelly ME, Ridgway PF, Conlon KC. The predictive value of C-reactive protein (CRP) in acute pancreatitis - is interval change in CRP an additional indicator of severity? HPB (Oxford). 2017;19(10):874-880.
39. Jinno N, Hori Y, Naitoh I, Miyabe K, Yoshida M, Natsume M, et al. Predictive factors for the mortality of acute pancreatitis on admission. PLoS One. 2019;14(8):e0221468.
40. De Campos T, Cerqueira C, Kuryura L, Parreira JG, Solda S, Perlingeiro JA, et al. Morbimortality indicators in severe acute pancreatitis. JOP. 2008;9(6):690-697.
Full Text Sources:
Abstract:
Views: 11372

For Authors
Journal Subscriptions

Dec 2025
Supplements
Instructions for authors
Online submission
Contact
e-ISSN: 2601 - 1700 (online)
ISSN-L: 2559 - 723X
Journal Abbreviation: Surg. Gastroenterol. Oncol.
Surgery, Gastroenterology and Oncology (SGO) is indexed in:
- SCOPUS
- EBSCO
- DOI/Crossref
- Google Scholar
- SCImago
- Harvard Library
- Open Academic Journals Index (OAJI)
Surgery, Gastroenterology and Oncology (SGO) is an open-access, peer-reviewed online journal published by Celsius Publishing House. The journal allows readers to read, download, copy, distribute, print, search, or link to the full text of its articles.
Time to first editorial decision: 25 days
Rejection rate: 61%
CiteScore: 0.2

Meetings and Courses in 2025
Meetings and Courses in 2024
Meetings and Courses in 2023
Meetings and Courses in 2022
Meetings and Courses in 2021
Meetings and Courses in 2020
Meetings and Courses in 2019
Verona expert meeting 2019

Surgery, Gastroenterology and Oncology applies the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits readers to copy and redistribute the material in any medium or format, remix, adapt, build upon the published works non-commercially, and license the derivative works on different terms, provided the original material is properly cited and the use is non-commercial. Please see: https://creativecommons.org/licenses/by-nc/4.0/
Publisher’s Note:
The opinions, statements, and data contained in article are solely those of the authors and not of Surgery, Gastroenterology and Oncology journal or the editors. Publisher and the editors disclaim responsibility for any damage resulting from any ideas, instructions, methods, or products referred to in the content.
 IASGO Society News
IASGO Society News