Surgery, Gastroenterology and Oncology

|
|
Background: Polypropylene meshes are widely used in hernia repair, with design and placement techniques significantly impacting postoperative outcomes. Merigrow™ Mesh is a microporous, lightweight polypropylene mesh that improves tissue integration while minimizing complications such as seroma, chronic pain and surgical site infections (SSI). This study evaluated its safety profile under real-world surgical conditions. Aim: To evaluate Merigrow™ Mesh safety and performance in elective hernia repair patients in a retrospective observational study.
Methodology: A total of 119 patients underwent elective hernia repair with Merigrow™ Mesh, with different mesh placement techniques. Standardized suture fixation was used in all cases. Patients were followed up to 12 months to assess early post-market safety outcomes, including complications and preliminary recurrence signals. Follow-up data were captured up to 12 months to assess early safety outcomes.
Results: A total of 119 patients, with a mean age of 49.02±13.98 years and predominantly male (84.87%), were enrolled in the study. The average surgical duration was 68.07±34.72 minutes. The SSI rate at 7 days was 1.68%, while seroma incidence was 3.36% of total cases. The highest seroma incidence (14.29 %) not statistically significant). No mesh migration, recurrence, or chronic postoperative pain occurred. All procedures were completed without intraoperative complications.
Conclusion: Merigrow™ Mesh establishes a favourable safety profile in elective hernia repair, with low rates of SSI, seroma, and no recurrence. Findings support its early post-market safety, though long-term recurrence trends require further study.
Introduction
Hernias remain a prevalent surgical condition worldwide, imposing significant morbidity and healthcare costs. Abdominal wall hernia is ~1.7% overall and 4% in those >45 years; globally ~20 million hernia repairs occur annually (1). If these conditions are untreated could lead to complications such as incarceration or strangulation may rise necessitating timely repair. Real-world data are needed to complement randomized controlled trials (RCTs), which often exclude complex patients (2).
Hernia surgery has been revolutionized by mesh-based repairs, which have been found to have lower recurrence rates than traditional suture techniques, up to 15% or less in many studies (3,4). Synthetic hernia meshes developed in the latter decades of the 20th century changed how hernias are surgically repaired. These devices offer more strength to the abdominal wall and provide tension-free healing. Because of their lower recurrence rate, hernia meshes are better than traditional suture repair in the long term (5,6). Despite these advances, the use of mesh is not without its potential for complications, including postoperative infections, seromas, chronic pain, and rare but serious adverse events such as mesh migration or failure, which can compromise patient recovery and long-term outcomes. Consequently, medical device surveillance now emphasizes the importance of post-market clinical follow-up (PMCF) studies, especially observational designs that provide early safety profiles across diverse patient groups and surgical techniques. The occurrence of postoperative complications underscores the ongoing need to evaluate mesh devices for improved safety and clinical effectiveness.
The Merigrow™ Mesh (Meril Endo-Surgery Pvt. Ltd, India), a synthetic prosthetic used for laparoscopic and open hernia repair, is versatile for groin hernias and abdominal soft tissue reinforcement. Designed to adapt to a variety of hernias, including inguinal, umbilical, and incisional hernias, it is intended to improve tissue integration and decrease the likelihood of complications. Currently, the evidence on mesh use in real-world settings remains limited. RCTs, although rigorous, often exclude complex patient populations and lack flexibility in capturing the nuanced performance of surgical devices across different anatomical sites, surgical approaches, or user expertise. Observational studies like the present one are critical to understanding mesh performance in real-world clinical contexts, particularly within Indian healthcare settings. Most existing data are derived from randomized controlled trials or small-scale observational studies, which may not fully represent the wide range of patient characteristics or surgical techniques encountered in routine clinical practice. With the changing landscape of patient demographics and evolving hernia repair techniques, conducting retrospective studies to evaluate the clinical performance and long-term safety of devices like Merigrow™ Mesh in more diverse surgical practices is crucial.
The present study aims to evaluate the safety and performance of Merigrow™ Mesh in a real-world setting by assessing the outcomes across different time points over a 7-day, 1-month, 3-month, 6-month, and 12-month follow-up period. By focusing on real-world hernia repair practice at a high-volume centre, this study fulfils an essential PMCF role in confirming the device's utility in elective surgeries involving lightweight polypropylene mesh. While recurrence remains an important long-term consideration, this study focuses on early safety events and post-market surgical outcomes within the first 12 months. The results provide initial performance insights and help guide real-world practice while longer-term data accrue.
Material and Method
Study Design & Population
This single-arm, retrospective observational study was conducted as part of a post-market clinical follow-up (PMCF) to evaluate the safety and performance of Merigrow™ Mesh in elective hernia repair. Consecutive patients treated at study center between February 2022 and October 2024 were included. Eligible cases involved inguinal, bilateral, umbilical, or mixed hernias treated via open or laparoscopic procedures. Patients with contaminated fields, active infections, gynecologic pathology, or intra-peritoneal interventions were excluded. All procedures used standardized polypropylene fixation techniques (sutures in open repair; tackers /trans-fascial sutures in laparoscopy)Seroma was defined as a palpable fluid collection persisting beyond 7 days, confirmed by examination or ultrasound. SSI was diagnosed per CDC criteria.
Data were collected retrospectively from structured medical records at predefined checkpoints: intraoperative, discharge, and post-discharge (7 days, 1, 6, and 12 months) (fig. 1). Assessors were not blinded but data abstraction was performed independently to reduce bias.
Figure 1 - Flowchart illustrating the retrospective study design and patient follow-up pathway
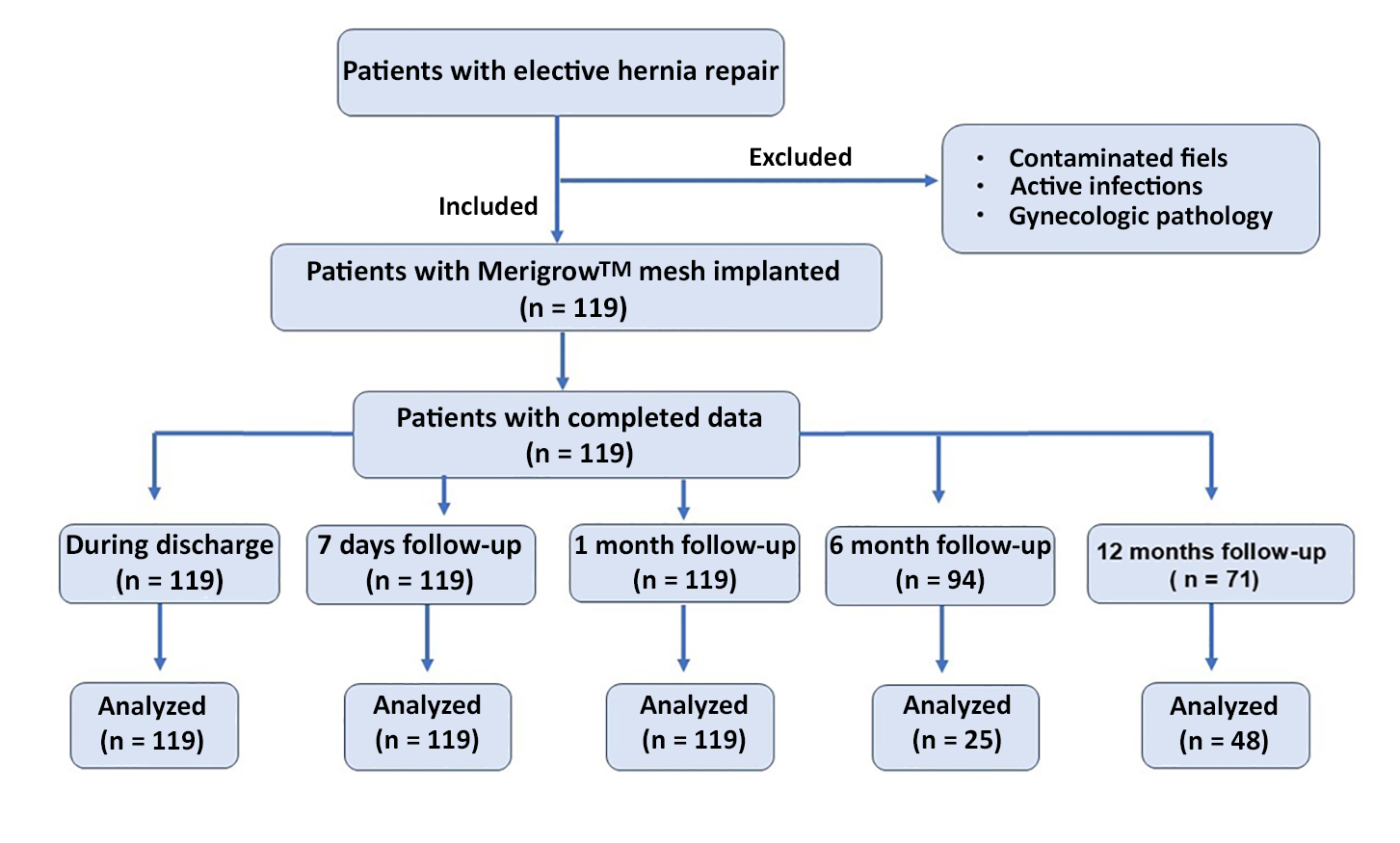
for evaluating Merigrow™ Mesh Knitted Sutures.
Study Endpoints
The primary endpoints were the recurrence of hernia, defined as the reappearance of herniated tissue at or near the site of repair, and the incidence of mesh-related re-intervention, including intra-operative re-interventions. Secondary endpoints included mesh infection, mesh integration as assessed by imaging or histology, length of hospital stay, and device-related adverse events such as mesh erosion, rejection, deep-seated infection, migration, and failure.
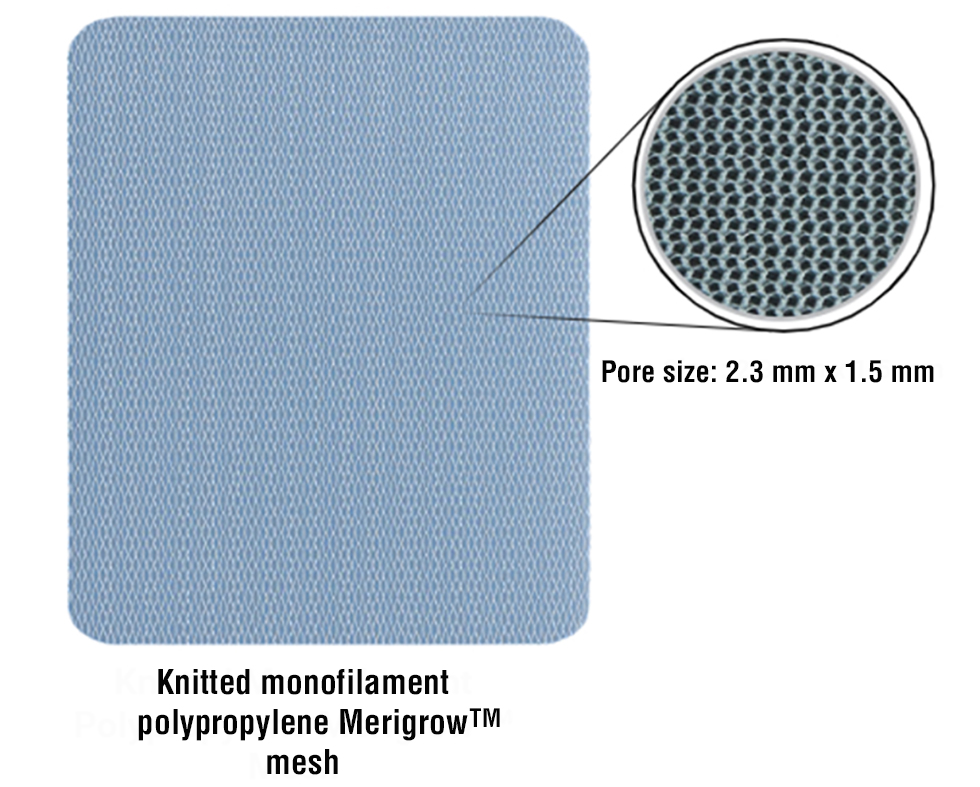
Figure 2 - A representative image of the Knitted Monofilament Polypropylene Merigrow™ Mesh depicting its structural architecture. The magnified inset highlights the uniform pore size (2.3 mm × 1.5 mm), which facilitates optimal tissue integration and host response.
Device Specification
Merigrow™ Mesh is a sterile, nonabsorbable,
knitted monofilament polypropylene mesh designed for hernia repair and soft tissue reinforcement. Made from lightweight polypropylene (55 gsm) and with a macroporous architecture (pore size: 2.3 x 1.5 mm). Its lightweight, macroporous structure allows tissue ingrowth and reduces risk of infection, seroma, or foreign body reaction. The mesh is colourless, knitted, and available in sizes (6×11 cm to 30×30 cm) to suit different types of hernias and surgical techniques, including open and laparoscopic approaches. Its rounded edges also help to reduce tissue irritation and make it easier to handle during placement. Merigrow™ Mesh is intended for permanent implantation, sterilized using ethylene oxide, and offers a high degree of biocompatibility, structural integrity and patient comfort across a wide spectrum of hernia repairs (fig. 2).
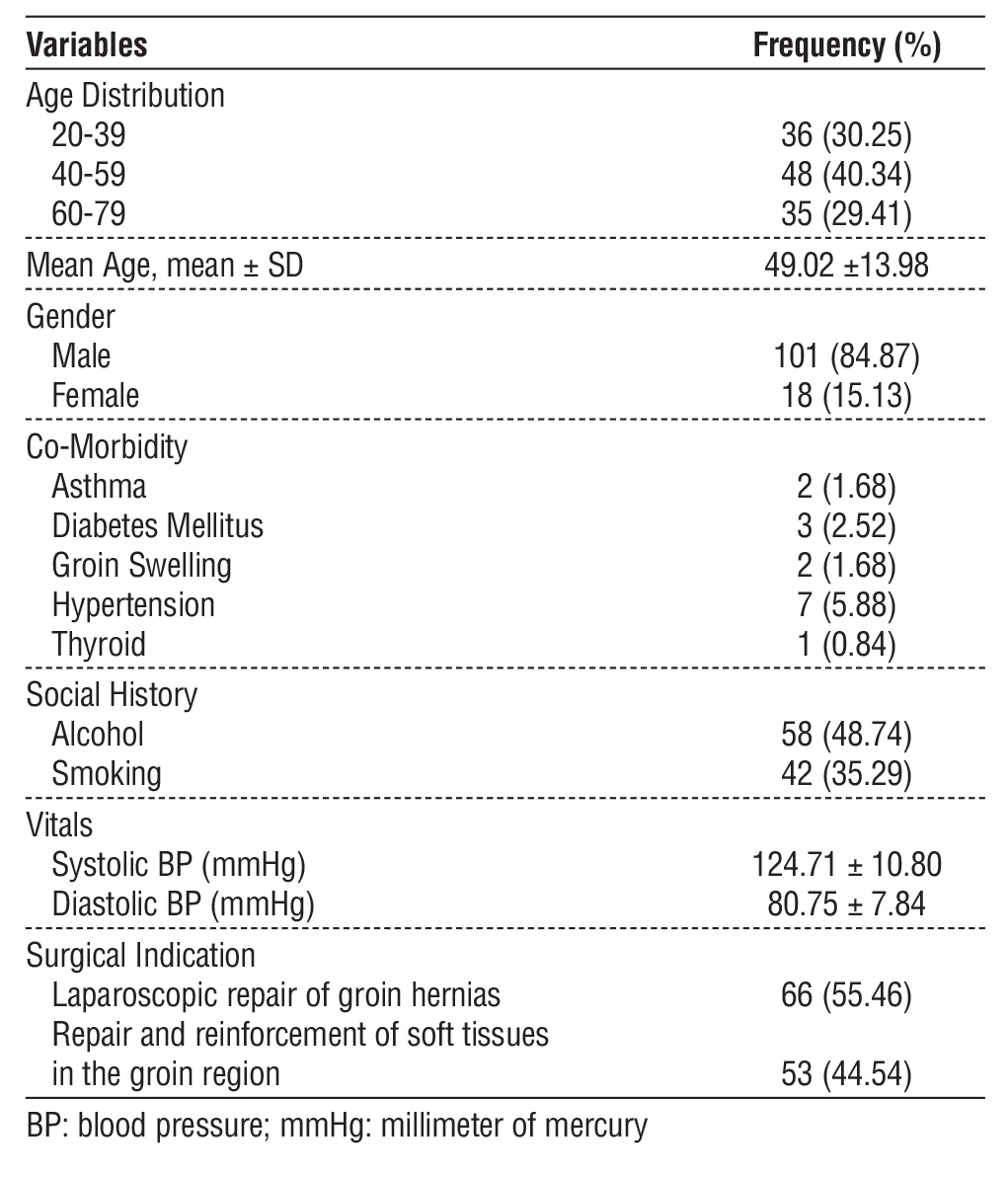
Table 1 - Patient demographics and baseline characteristics
Statistical Analysis
Microsoft Excel and SPSS version 27.0 were used for data cleaning and statistical analysis. Kolmogorov-Smirnov was applied to assess the data normality. Descriptive statistics were used to summarize patient demographics, surgical details, and clinical outcomes. Categorical data were expressed as n (%), continuous as mean ± SD. The analysis was exploratory, aimed at describing real-world safety outcomes.
Results
Demographic and Patient Baseline Details
A total of 119 consecutive patients with a mean age of 49.02 ± 13.98 years, most being male (84.87%), were enrolled in this real-world study evaluating elective hernia repairs with Merigrow™ Mesh. The inclusion of consecutive cases aimed to reduce selection bias and reflect daily clinical practice. Regarding medical history, hypertension was reported in 5.88 % of patients, diabetes mellitus in 2.52%, and groin swelling was reported in 1.68% of patients, respectively. Moreover, a large proportion of patient populations had a lifestyle of alcohol use (48.74 %) and smoking (35.29 %), which could affect the surgical outcome. The mean systolic and diastolic blood pressures were 124.71 mm/Hg (SD ± 10.80) and 80.75 mm/Hg (SD ± 7.84), respectively (table 1). The primary surgical indications were laparoscopic repair of groin hernias (55.46%) and repair/reinforcement of soft tissues in the groin region (44.54%). These baseline features reflect routine Indian surgical practiceurban practice.
Table 2 - Summary of intraoperative hernia repair characteristics

Operational Details
The procedural characteristics of patients who underwent hernia repair using Merigrow™ Mesh are summarized in table 2. All 119 patients undertook elective surgery, with a mean operative time of 68.07 ± 34.72 minutes. General anaesthesia was administered in most of the cases (79.83%), followed by spinal (18.49%) and local anaesthesia (1.68%). Laparoscopic-based hernia repair was performed in major patients (55.46 %), while the remaining (44.54%) underwent open surgery. The most frequently employed mesh placement method was underlay (43.70%), followed by inlay (30.25%), retro-rectus (20.17%), and overlay (5.88%). The most common clinical indication was inguinal hernia (31.93 %), followed by umbilical (18.49%) and bilateral inguinal hernia (19.33%). A total of 167 meshes were implanted across all cases, among which the most used mesh dimensions were 10×15 cm (24.55%) and 12×15 cm (23.35%). No intraoperative complications or procedural modifications occurred.
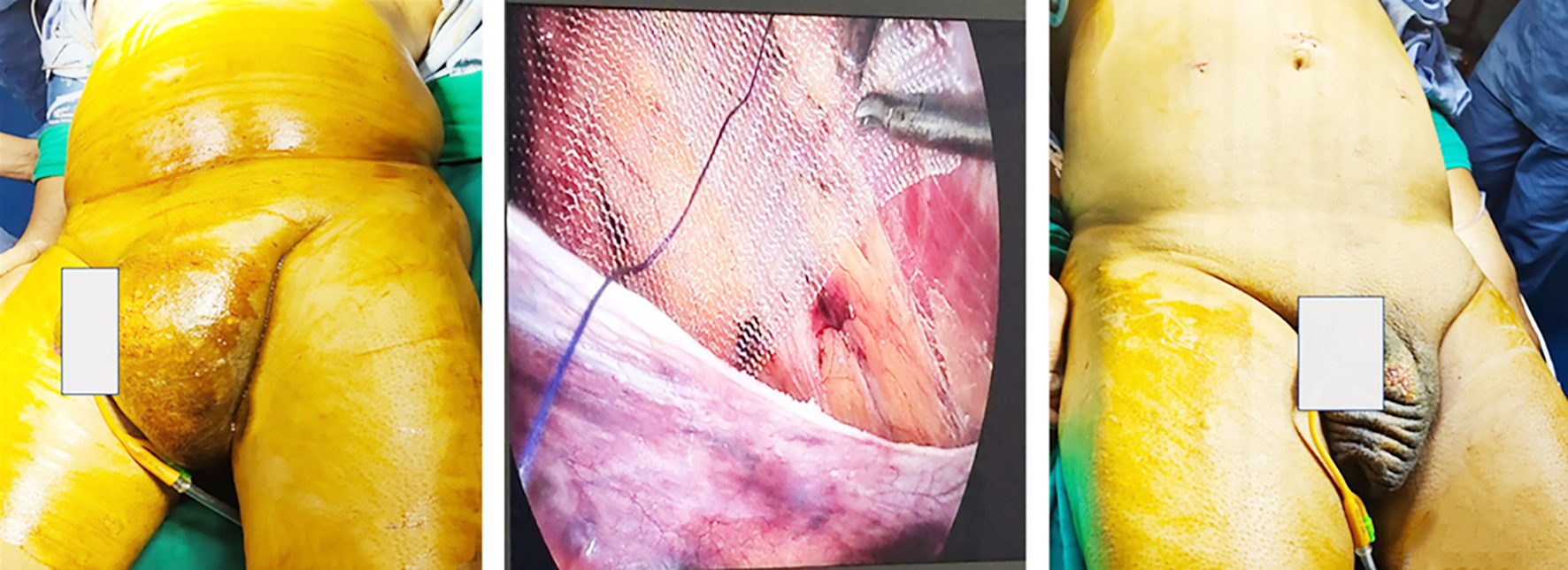
The study represents fig. 3 as a representative case of a patient who received laparoscopic Transabdominal Preperitoneal (TAPP) repair for left inguinal hernia using Merigrow™ Mesh. The patient with a visible groin bulge is seen in the preoperative image (fig. 3 a). During surgery, the mesh was positioned with appropriate coverage over the myopectineal orifice (fig. 3 b). Further, the postoperative image (fig. 3 c) shows the complete resolution of the hernia with no visible postoperative complications.
Discharge Details
No intraoperative complications were observed during the implantation of Merigrow™ Mesh, and no cases of infection, mesh integration failure, or mesh migration were reported at the time of discharge. No AEs or SAEs were recorded from the time of surgery to discharge (table 3).
Table 3 - Discharge outcomes following hernia repair, including discharge rate and frequency of intraoperative or post-discharge complications.

Patients follow-up
The longitudinal follow-up outcomes are presented in table 4. All 119 patients were assessed at 7-day and 1-month follow-up. In the 6-month follow-up, 94 patients (78.99 %) were visited, and in the 12-month follow-up, 71 patients (59.66 %) were visited. However, follow-up completion beyond 1 month was limited 21% at 6 months, 40% at 12 months. No mesh integration failure, mesh migration or mesh-related adverse events were recorded at any point during the follow-up period.
Table 4 - Follow-up outcomes at specified intervals after hernia repair, including visit attendance, completion rates, and post-operative complications.
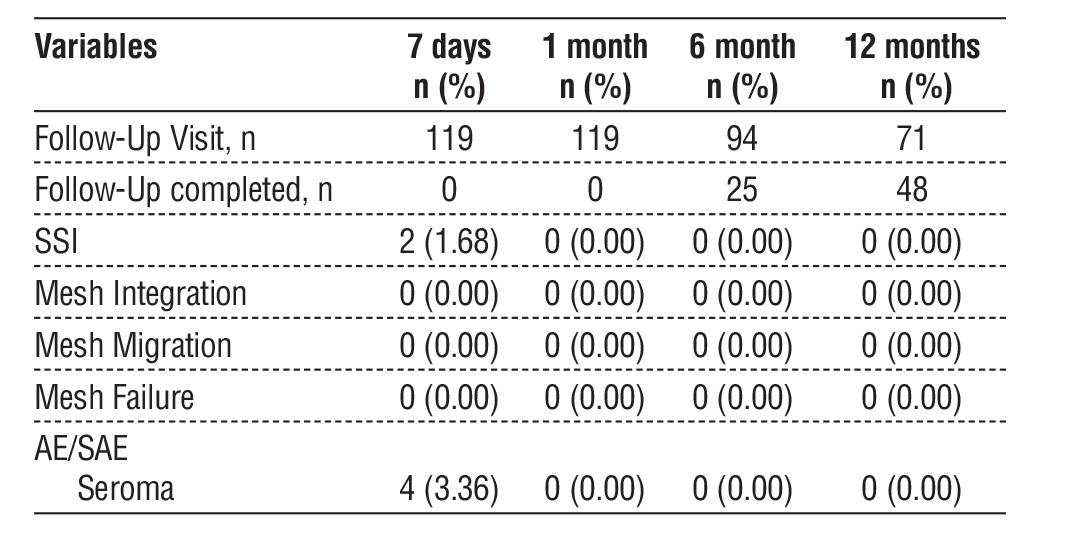
At 7-day follow-up, two patients (1.68%) developed SSI, and four patients (3.36%) presented with postoperative seromas. While beyond the 7-day follow-up time point, no AE/SAE events were reported.
Figure 3 (a-c) - Representative case from the study showing laparoscopic left inguinal hernia repair using Merigrow™ Mesh. (a) Preoperative left inguinal swelling, while (b) shows Intraoperative mesh placement and (c) depicts the postoperative resolution with healed port sites.
Discussion
The study evaluates Merigrow™ Mesh safety and clinical effectiveness through retrospective data collected from 119 patients who received the lightweight polypropylene mesh during elective hernia surgery. This real-world evaluation offers post-market insight into how Merigrow™ Mesh performs under standard surgical conditions without the constraints of randomized trial protocols. Such observational data is valuable in understanding device safety, especially in diverse patient populations and surgeon preferences.
Our study population showed no signs of hernia recurrence or ongoing groin pain during the twelve-month postoperative period. These results are comparable to or better than those reported in other retrospective studies involving lightweight polypropylene meshes. For example, in a retrospective single-surgeon TEP inguinal hernia study of 150 partially absorbable mesh repairs, Kabaoglu et al. reported a 6.0% seroma rate and a low recurrence rate of 0.67% at a median follow-up of 30 months (7). In contrast, our study showed 0% recurrence and a 3.36% seroma rate at 12 months. These findings are favorable compared with the TULP trial, a large multicenter randomized study involving 950 patients undergoing laparoscopic TEP hernia repair with Ultrapro® mesh. At 1-year follow-up, the Ultrapro® group demonstrated a 2.7% recurrence rate and a 2.9% chronic pain rate (8). Despite technique differences, our findings support Merigrow™ Mesh’s early safety.
These findings align with the long-term results that previous mesh studies. Sajid et al. reviewed 2,189 patients and found that lightweight polypropylene mesh caused less chronic pain than heavyweight mesh without increasing recurrence rates (9). The registry-based study by Melkemichel e.t al. analyzing 76,000 hernia repairs, showed similar recurrence for heavyweight and lightweight polypropylene meshes (10). Although our study did not conduct rigorous chronic pain assessments, the absence of discomfort and reoperations suggests lightweight mesh designs with wider pores and a diminished inflammatory footprint may provide more advantageous long-term outcomes. Krpata et al., compared medium-weight versus heavyweight meshes in open ventral hernia repair and found no significant differences in outcomes, stressing technique and mesh handling over weight (11).
Observational RWE studies, such as the multicenter cohort by Berrevoet et al., highlight the importance of post-market mesh evaluations in routine practice. In their study of laparoscopic ventral hernia repair with Proceed® mesh, a 10.5% seroma/hematoma rate and 3.5% recurrence rate were observed over a 27-month follow-up (12). While our follow-up period was shorter, the absence of recurrence and low complication rates at 12 months in our study support the early safety profile of Merigrow™ Mesh. All cases used standardized suture fixation with no intraoperative complications, conversions, or chronic postoperative discomfort.
Our study showed overlay repairs produced the highest seroma rate at 14.29%, while inlay and retrorectus placements had none. This trend was not statistically significant. Mesh placement position appears critical, as superficial placement leads to dead space (13). Our results agree with Niranjan et al., who reported no seroma, infection, migration, or recurrence using Dolphin® Mesh in 51 patients (14). In our study, The absence of mesh migration, chronic pain, or failure at 12 months supports the early safety advantages of Merigrow™ Mesh’s design.
While no patient complaints of chronic pain were recorded during the 12-month follow-up period, standardized pain or quality-of-life scales were not used, so absence of pain in notes does not equal validated comfort measures. The study did not capture metrics like return to work, satisfaction, or physical activity resumption—parameters that are increasingly recognized as essential for evaluating the true impact of hernia repair. These gaps highlight the need for future prospective studies using validated patient-reported outcome measures (PROMs) to comprehensively assess long-term functional outcomes alongside safety endpoints.
Our early postoperative complication rates are comparable to the literature. Straton et al. reported a 4.7% seroma rate in 212 laparoscopic inguinal hernia repairs, with one SSI (15). The 3.36% seroma rate in our study may reflect Merigrow™ Mesh’s macroporous architecture, which promotes fluid drainage and integration. The SSI rate of 1.68 % also supports safety. Absence of migration or chronic pain supports the design advantages of its lightweight, large-pore structure, similar to TiMesh™ and Parietex™ (16).
Further,Warren et al. reported 17.9 % SSI using synthetic mesh in contaminated ventral hernia fields, which was reportedly higher than our study finding (17). Despite the elevated baseline infection risks, these data show the role of mesh porosity and surgical environment in infection risk. Our study also agree with Pulikkal et al., found no significant difference in SSI or seroma between open and laparoscopic mesh repairs (18). In summary, this single-arm retrospective study contributes real-world data on Merigrow™ Mesh’s performance in elective hernia repair. The favourable safety signals, low complication rates, and high intraoperative consistency make it an appropriate early evidence source to inform clinical practice and support broader device adoption under post-market surveillance.
Strength
The study's real-world assessment of Merigrow™ Mesh in elective surgery provides practical insights into its performance. The study minimized procedural variability through standardized fixation techniques and uniform operative protocols, which resulted in case consistency. The study including both open and laparoscopic repairs increases the clinical applicability. Merigrow™ Mesh demonstrates safety and reliability in routine hernia repair procedures, as the study shows consistently low complication rates.
Limitation
This study has several limitations. First, its a single-arm retrospective design without a control group, which prevents direct evaluation of mesh types and surgical approaches. Second, the study failed to use standardized patient-reported outcomes, including validated pain and quality-of-life scores, which limited the evaluation of functional recovery. The 12 months duration is too short to capture recurrence, which can develop years after surgery. The 40% dropout at 12 months may have underreported late complications.
Despite these limitations, the study design reflects real-world surgical practice, with standardized fixation protocols and consistent operative techniques that minimize procedural variability. These features enhance internal validity and provide useful early post-market safety insights for elective hernia repair.
Conclusion
This study supports the early safety and intraoperative handling profile of Merigrow™ Mesh in elective hernia repair. While not designed to evaluate comparative effectiveness, the consistent procedural outcomes suggest favourable short-term utility. The low surgical site infection rates, seroma, no recurrence, and absence of chronic postoperative pain support its early performance profile. While longer-term recurrence data are still needed, these findings indicate Merigrow™ Mesh is a reliable option for clean, elective hernia repair.
Authorship
Pushkar Galam and Nikil Jillawar conducted data collection, conceptualization, and overall supervision. Panchhi Malve and Pavan P Goud contributed to methodology, manuscript writing and review. Kiran Kumar Shetty contributed to the technical inputs related to the device and study methodology and provided editorial assistance during manuscript drafting.
Conflicts of Interest
Kiran Kumar Shetty is an employee of Meril, Vapi, India, the manufacturer of the medical device evaluated in this study. His involvement was restricted to providing technical input regarding the device and editorial support during manuscript preparation. The author did not participate in clinical decision-making, data collection, or analysis. Other authors, Pushkar Galam, Nikil Jillawar, Panchhi Malve and Pavan P Goud declare no conflict of interest.
Source of Funding
No external funding was received for the conduct of this study. The medical device used (Merigrow™ Mesh) was part of routine care, and the retrospective study was conducted using de-identified medical records. One of the authors, Kiran Kumar Shetty, is affiliated with Meril (the device manufacturer); the company did not provide specific grants or incentives for this research. No payments were made to the clinical site or investigators for data collection, analysis, or publication.
Ethical Approval
This study was conducted in accordance with the Declaration of Helsinki, the International Council for Harmonization of Technical Requirements for Pharmaceuticals for Human Use (ICH) - Good Clinical Practice (GCP), and ISO 14155:2020 GCP standards. The Institutional Ethics Committee (IEC/1284) approved the study prior to initiation (IEC approval letter; IEC/DEC/ 2024). Due to the retrospective and observational nature of the study, there was no direct intervention and modification of the patient's treatment; therefore, the ethics committee waived the informed consent.
References
1. Pandya B, Huda T, Gupta D, Mehra B, Narang R. Abdominal Wall Hernias: An Epidemiological Profile and Surgical Experience from a Rural Medical College in Central India. The Surgery Journal. 2021;07(01):e41-e46.
2. Bindal V, Pandey D, Gupta S, Agarwal P, Dahiya A, Gupta D, Bindal UD. A Real-World Experience of the Short-Term Clinical Outcomes of Laparoscopic and Robotic-Assisted Ventral Hernia Repairs. Cureus. 2025 Mar 30;17(3):e81480.
3. Madsen LJ, Oma E, Jorgensen LN, Jensen KK. Mesh versus suture in elective repair of umbilical hernia: systematic review and meta-analysis. BJS Open. 2020;4(3):369-379.
4. Zendejas B, Onkendi EO, Brahmbhatt RD, Lohse CM, Greenlee SM, Farley DR. Long-term outcomes of laparoscopic totally extraperitoneal inguinal hernia repairs performed by supervised surgical trainees. The American Journal of Surgery. 2011;201(3):379-384.
5. van Rooijen MMJ, Lange JF. Preventing incisional hernia: closing the midline laparotomy. Tech Coloproctol. 2018;22(8):623-625.
6. Liang MK, Holihan JL, Itani K, et al. Ventral Hernia Management. Ann Surg. 2017;265(1):80-89.
7. Kabaoglu B, Sobutay E, Bilgic C. Postoperative Outcomes and Recurrence Rate in Laparoscopic Tep Inguinal Hernia Repairs Using Partially Absorbable Meshes: A Retrospective Single-Surgeon Study Over a 5-Year Period. Sisli Etfal Hastan Tip Bul. 2024 Sep 30;58(3):276-283.
8. Burgmans JP, Voorbrood CE, Simmermacher RK, et al. Long-term Results of a Randomized Double-blinded Prospective Trial of a Lightweight (Ultrapro) Versus a Heavyweight Mesh (Prolene) in Laparoscopic Total Extraperitoneal Inguinal Hernia Repair (TULP-trial). Ann Surg. 2016 May;263(5):862-6.
9. Sajid MS, Kalra L, Parampalli U, Sains PS, Baig MK. A systematic review and meta-analysis evaluating the effectiveness of lightweight mesh against heavyweight mesh in influencing the incidence of chronic groin pain following laparoscopic inguinal hernia repair. The American Journal of Surgery. 2013;205(6):726-736.
10. Melkemichel M, Bringman SAW, Widhe BOO. Long-term Comparison of Recurrence Rates Between Different Lightweight and Heavyweight Meshes in Open Anterior Mesh Inguinal Hernia Repair. Ann Surg. 2021;273(2):365-372.
11. Krpata DM, Petro CC, Prabhu AS, et al. Effect of Hernia Mesh Weights on Postoperative Patient-Related and Clinical Outcomes After Open Ventral Hernia Repair. JAMA Surg. 2021;156(12):1085.
12. Berrevoet F, Fierens K, De Gols J, Navez B, Van Bastelaere W, Meir E, Ceulemans R. Multicentric observational cohort study evaluating a composite mesh with incorporated oxidized regenerated cellulose in laparoscopic ventral hernia repair. Hernia. 2009 Feb;13(1):23-7.
13. Ghazal T, Hamza E, Meshikhes AWN. Treatment of persistent complex seroma postventral incisional hernia repair by capsulectomy and scarification using argon beam coagulator. BMJ Case Rep. 2022;15(2):e242961.
14. Paramashivaiah N, Anikode Subramanian R, Krishna Kumar M, R Nath K, Jose A. Utility of Non-Absorbable Knitted Surgical Polypropylene Mesh - A Post Market Clinical Follow Up Study. International Journal of Science and Research (IJSR). 2022;11(8):804-809.
15. Chen L, Hu M, Huang S. Analysis of influencing factors of complications after laparoscopic inguinal hernia repair: An observational study. Medicine. 2023;102(49):e36516.
16. Moreno-Egea A, Carrillo-Alcaraz A, Soria-Aledo V. Randomized clinical trial of laparoscopic hernia repair comparing titanium-coated lightweight mesh and medium-weight composite mesh. Surg Endosc. 2013 Jan;27(1):231-9.
17. Warren J, Desai SS, Boswell ND, et al. Safety and Efficacy of Synthetic Mesh for Ventral Hernia Repair in a Contaminated Field. J Am Coll Surg. 2020;230(4):405-413.
18. Pulikkal Reghunandanan R, Ali Usman A, Basheer S, Kuttichi L, Els Jojo J, Abdul Rasheed MF. Laparoscopic Versus Open Inguinal Hernia Repair: A Comparative Study. Cureus. Published online November 10, 2023.
Full Text Sources:
Abstract:
Views: 63

For Authors
Journal Subscriptions

Dec 2025
Supplements
Instructions for authors
Online submission
Contact
e-ISSN: 2601 - 1700 (online)
ISSN-L: 2559 - 723X
Journal Abbreviation: Surg. Gastroenterol. Oncol.
Surgery, Gastroenterology and Oncology (SGO) is indexed in:
- SCOPUS
- EBSCO
- DOI/Crossref
- Google Scholar
- SCImago
- Harvard Library
- Open Academic Journals Index (OAJI)
Surgery, Gastroenterology and Oncology (SGO) is an open-access, peer-reviewed online journal published by Celsius Publishing House. The journal allows readers to read, download, copy, distribute, print, search, or link to the full text of its articles.
Time to first editorial decision: 25 days
Rejection rate: 61%
CiteScore: 0.2

Meetings and Courses in 2025
Meetings and Courses in 2024
Meetings and Courses in 2023
Meetings and Courses in 2022
Meetings and Courses in 2021
Meetings and Courses in 2020
Meetings and Courses in 2019
Verona expert meeting 2019

Surgery, Gastroenterology and Oncology applies the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits readers to copy and redistribute the material in any medium or format, remix, adapt, build upon the published works non-commercially, and license the derivative works on different terms, provided the original material is properly cited and the use is non-commercial. Please see: https://creativecommons.org/licenses/by-nc/4.0/
Publisher’s Note:
The opinions, statements, and data contained in article are solely those of the authors and not of Surgery, Gastroenterology and Oncology journal or the editors. Publisher and the editors disclaim responsibility for any damage resulting from any ideas, instructions, methods, or products referred to in the content.
 IASGO Society News
IASGO Society News