Surgery, Gastroenterology and Oncology

|
|
Background: Traumatic Brain Injury (TBI) is a major global health concern, frequently leading to cerebral edema, a critical factor in secondary brain injury and poor outcomes. Aquaporin-4 (AQP-4), an astrocytic water channel protein, and Tumor Necrosis Factor-alpha (TNF-a), a proinflammatory cytokine, are well-established mediators in the pathophysiology of cerebral edema and neuroinflammation following TBI. Given the limitations of current treatments, exploring novel adjunctive therapies is crucial. Objectives: This literature review aims to comprehensively investigate the potential therapeutic effects of Trigona honey in ameliorating cerebral edema after TBI, specifically focusing on its influence on AQP-4 mRNA and TNF-a expression.
Methods: We conducted a comprehensive review of existing in vitro and in vivo literature concerning Trigona honey's neuroprotective effects in TBI, with particular attention to its proposed mechanisms involving AQP-4 and TNF-a modulation.
Results: Evidence suggests that Trigona honey, rich in bioactive compounds like phenolic compounds and flavonoids, may reduce cerebral edema in TBI. This effect is hypothesized to be mediated through the downregulation of AQP-4 expression and the attenuation of the inflammatory cascade via TNF-a suppression. While collective findings support its potential, the precise molecular mechanisms underlying these modulations require further elucidation.
Conclusion: This review highlights Trigona honey as a promising natural agent for reducing cerebral edema in TBI, likely by regulating AQP-4 mRNA expression and TNF-a levels. Future research, including randomized clinical trials, is warranted to fully validate its therapeutic efficacy and establish its role as an adjunctive treatment in TBI management.
INTRODUCTION
Head injury can trigger secondary brain injury known as cerebral oedema (1). Cerebral oedema involves the increased accumulation of extracellular and /or intracellular brain fluid (2). This type of oedema results in the swelling of brain tissue, which is related to a progressive rise in brain fluid levels that can occur due to ischemia, (3) trauma, (4) tumors, (5) and inflamation (5). The limited volume of the cranial cavity and progressive cerebral oedema lead to increased intracranial pressure (ICP), subsequently causing reduced cerebral blood flow, cerebral herniation, and even death (1).
The cerebral oedema can be divided into two categories based on measuring tissue fluid levels through diffusion-weighted imaging (DWI) which include: cytotoxic or intracellular oedema and vasogenic or extracellular oedema (2). In cytotoxic oedema, there is an accumulation of fluid within the intracellular compartment, involving neurons, microglia, and astrocytes. Additionally, there is a failure of the Na+/K+-ATPase pump due to ischemic conditions, leading to disrupted Adenosine Triphosphate (ATP) production. This results in an imbalance of intracellular and extracellular ion exchange, causing increased intracellular osmolality and fluid entering the cells, leading to intracellular disturbances. Hence, cytotoxic oedema is also referred to as ionic oedema (2).
Vasogenic oedema involves an increase in extracellular fluid due to blood-brain barrier (BBB) leakage. This condition leads to increased osmotic pressure, causing fluid to exit blood vessels and enter the extracellular compartment. Intravascular fluid then exits through the endothelium via mechanisms like pinocytosis and/or leaky tight junctions (2).
Damage to the Blood Brain Barrier (BBB) is a key factor in the occurrence of cerebral oedema. Both types of oedema, vasogenic and cytotoxic, lead to Intracranial Pressure (ICP) and reduced cerebral perfusion pressure (CPP), ultimately resulting in tissue ischemia.(6) Inflammation in the brain due to injury or disruption prompts neurons, astrocytes, and microglia to release inflammatory factors, including interleukin-1b (IL-1b), interleukin-6 (IL-6), interferon-? (IFNg), tumor necrosis factor-a (TNF-a), and transforming growth factor-b (TGF-b). This inflammatory response disrupts synaptic signaling and plasticity, weakens inhibitory neurotransmission, and increases blood-brain barrier (BBB) permeability (7-9).
The pathophysiology of cerebral oedema is also influenced by several factors, including Aquaporin (AQP) and cytokines. Aquaporin plays a key role in the development of cerebral oedema (10). Several studies have found an increased levels of AQP in post-head injury patients and its involvement in the occurrence of cerebral oedema suggesting that the use of AQP inhibitors may play a role in managing cerebral oedema (10,11). Inhibition of AQP expression has been associated with reduced oedema levels and infarct area, as well as improved functional status in experimental animal studies with cerebral ischemia. These findings showed that AQP is beneficial in suppressing the occurrence of vasogenic oedema (11).
Experimental studies have discovered an increase in AQP4 levels in glial cells and a decrease in perivascular AQP4 levels at the onset of vasogenic oedema (11). The resolutions in oedema symptoms and functional status improvement are correlated with the restoration of AQP4 to its normal state. There is a direct relationship between cerebral blood flow (CBF) and body metabolism. Reduced CBF is associated with hypoxia and anaerobic glycolysis. Hypoxia can disrupt the blood-brain barrier through various mechanisms mediated by Vascular Endothelial Growth Factor (VEGF), inflammatory, and nitric oxide (NO) responses. Hypoxia triggers increased VEGF production, leading to enhanced vascular permeability and oedema formation. VEGF binds to VEGF receptors in the blood vessels of ischemic areas and contributes to BBB disruption and leakage (12).
Trigona Stingless Bee Honey (SBH) is produced by stingless bees of the Trigona genus, native to tropical areas such as Southeast Asia, Australia, and parts of Central and South America. Valued for its distinctive composition and health benefits, Trigona SBH has attracted interest for its potential medicinal applications (13). Trigona SBH, recognized for its distinct composition and health-promoting qualities, has garnered interest for its possible medicinal benefits. Unlike other types of honey, Trigona SBH is more acidic and has a unique flavor profile. It is also packed with antioxidants, flavonoids, phenolic acids, vitamins, and minerals, which give it strong antimicrobial, anti-inflammatory, and antioxidant properties. These attributes have made Trigona SBH a valued ingredient in both traditional and alternative medicine practices (13,14).
Trigona SBH has antioxidant properties that inhibit NF-kB and MAPK signaling pathways, leading to reduced production of inflammatory factors such as IL-6, TNF-a, and IFN-g. Due to its rich antioxidant content, Trigona SBH is widely studied for its role in managing inflammation and related conditions, where oxidative stress is a key factor influencing the level of inflammation (9,14-16).
The goal of managing cerebral oedema is to maintain both global and regional CBF to meet the brain's metabolic demands and prevent secondary injury from cerebral ischemia. Various compounds or agents are used to inhibit factors that trigger oedema formation. The aim of this review is to discuss Trigona SBH and its potential in reducing brain oedema through the expression of Aquaporin-4 (AQP-4) mRNA and TNF-a in traumatic brain injury patients.
The Blood Brain Barrier (BBB) and Oedema in Traumatic Brain Injury (TBI)
The BBB is a critical structure for maintaining homeostasis in the brain. Leakage of plasma proteins through the BBB is a key factor in the accumulation of fluid in the extracellular space (17,18). Various mechanisms can be associated to the disruption of the BBB, one of which is brain tissue inflammation (17).
The BBB selectively separates the brain tissue from the bloodstream. The key structure of the BBB is the capillary endothelial cells that are interconnected through tight junctions. Unlike extracerebral capillaries, cerebral endothelial cells are nonfenestrated, contain fewer pinocytic vesicles, have many mitochondria, and are covered by astrocytes. The opening of tight junctions is the key mechanism in the formation of vasogenic oedema. The fluid accumulation in oedema can reach 14-78 mL/day (19). Before entering the microvascular bloodstream, oedema fluid enters the brain ventricles through the transependymal flow.
Extracellular proteins such as claudin, occludin and adhesion junction molecules are the tight junction’s molecular composition. Claudins are regarded as the key proteins forming the foundation of tight junctions (TJs) and play a critical role in determining the "tightness" of the paracellular barrier between adjacent endothelial cells of the blood-brain barrier (BBB). The claudin protein family consists of 27 members
identified so far, each with an approximate molecular weight of ~26 kDa (20). These proteins exhibit tissue-specific expression patterns, which contribute to varying barrier properties across different tissues. At the BBB, claudin-5 is known to be the primary tight junction protein. Alterations in tight junction functionality are not solely due to changes in the levels of individual proteins but are typically the result of disruptions in the overall balance of all tight junction proteins (21,22).
Occludin is a tight junction protein and an essential structural and functional component of the blood-brain barrier (BBB). Similar to claudin-5, a reduction of occludin in brain endothelial cells signals a disruption in BBB integrity (23,24). Occludin plays a crucial role in maintaining BBB stability. It is a 65 kDa integral membrane protein consisting of 502 amino acids, featuring two extracellular loops, one intracellular loop, and both carboxyl and amino ends facing the cytoplasm (23,25). Activation of the P2X7 receptor (P2X7R) by ATP triggers the release of interleukin-1b (IL-1b), which subsequently increases the activity of matrix metalloproteinase-9 (MMP-9). This leads to the degradation of tight junction proteins, such as ZO-1 and occludin, ultimately contributing to BBB disruption (23,26).
Junction adhesion molecules belong to the immunoglobulin superfamily and consist of a single membrane-spanning domain, an IgG-like extracellular domain, an extracellular N-terminus essential for dimerization, and a short cytoplasmic C-terminus. This C-terminus is crucial for interacting with the tight junction scaffold protein ZO-1 due to the presence of a zonula occluden binding domain (27). The molecular structure of adherens junctions (AJs) includes the cadherin-catenin complex and the nectin-afadin complex. VE-cadherin is a transmembrane protein at AJs, and its cytoplasmic tail binds with b-catenin, plakoglobin, and p120. b-Catenin and plakoglobin further connect to ?-catenin, which then binds to vinculin, a-actinin, and ZO-1, facilitating anchorage to actin microfilaments. Nectin, another transmembrane protein from the IgG superfamily, promotes homophilic cell-cell adhesion. Intracellularly, nectin binds with afadin, which links to ponsin; ponsin then associates with ?-catenin and vinculin, thereby connecting to actin (20,27,28).
When these extracellular proteins are in excess, they are removed through phagocytic mechanisms by microglia and astrocytes. These extracellular proteins are also known as trans-membrane proteins that bind to intracellular proteins such as ZO-1 and ZO-2. This binding forms attachments between tight junctions and the endothelial cell cytoskeleton. A decrease in the expression and/or function of these proteins will cause the tight junctions to open and lead to the formation of oedema. The main mediator of this mechanism is Vascular Endothelial Growth Factor (VEGF) (29).
VEGF is a primary regulator of both normal and pathological blood vessel growth. VEGF receptors play a significant role in cell-cell adhesion and communication by directly interacting with cell adhesion molecules. VEGF disrupts tight junctions by modifying the phosphorylation of zonula occludens-1 (ZO-1) and occludin via a Src-dependent pathway. This VEGF-mediated increase in vascular permeability contributes to rapid oedema formation within hours, but it also supports revascularization and long-term cell survival (30,31).
Tumor Necrosis Factor-a (TNF-a) in TBI
TNF-a is a pleiotropic cytokine produced by astrocytes, endothelial cells, microglia, and neurons in response to TBI (32). It plays a dual role in the central nervous system (CNS) by inducing both neurotoxic and neuroprotective effects, (33) depending on factors such as cell-specific expression, timing of TNF expression, ligand concentration, and the influence of other signaling pathways (34).
In the brain, astrocytes, endothelial cells, microglia, and also neurons produce TNF (35). TNF-a has a role, namely, inducing apoptosis and necrotic cell death, as well as stimulating cell growth and differentiation (36). The inflammatory cascade following traumatic injury has been documented both clinically and experimentally in patients with TBI, including the influx of leukocytes into injured brain tissue, increased release of cytokines such as TNF-a, IL-1b, IL-10, IL-12, and IL-6 (37,38). Pro-inflammatory and anti-inflammatory cytokines both involved in a complex interaction, yet the full extent of their relationship in the processes of damage and healing is not fully understood (39).
Cytokines, chemokines, and inflammatory growth factors are widely recognized for their significant role in the pathophysiology of TBI (40). Although the initiation of the inflammatory response also contributes significantly to inducing neurorepair mechanisms as a response to physiological stress, excessive or uncontrolled inflammation can lead to neuronal dysfunction and degeneration through pathological neuroinflammatory cascades (41).
TNF-a is produced as a monomeric type-2 transmembrane protein (tmTNF), which is then enter the membrane as a homotrimer and separated by the matrix metalloprotease TNF converting enzyme into a soluble circulating trimer 51kDa (solTNF); both solTNF and tmTNF are biologically active (42). Immediately following TBI, the synthesis, and release of proinflammatory cytokines by astrocytes and microglia, particularly TNF-a with mRNA and protein levels, where these proinflammatory cytokines increase acutely within a short time of about 17 minutes post-injury (43). This process was observed in post-mortem brain examinations of patients after TBI. In a rat research model with TBI, it was found that TNF-a increased prior to the appearance of other cytokines (44). TNF-a is also known to exacerbate trauma and oxidative stress in the brain and plays a role in the release of glutamate and disruption of the BBB, which subsequently leads to an influx of further inflammatory factors (45).
Pharmacological experiments specifically aimed at reducing the production of TNF-a and acutely inhibiting its activity which have been shown to be associated with favorable post-trauma outcomes (32).
Members of the tumor necrosis factor receptor (TNFR) group and their ligands are induced in the early stages following acute central nervous system (CNS) injury and can initiate wide and complex intracellular signaling pathways. TNF-a is a pleiotropic cytokine that transduces signals for cell death and survival through its receptors, TNFR1 and TNFR2 (46).
These actions then activate polymorphonuclear leukocytes which it increase the leukocyte-endothelial adhesion molecules expressions (47). Then the leukocyte adhesion to capillary walls, and infiltrate the brain tissue. Activated leukocytes able to release inflammatory mediators and reactive oxygen species, which both of them have a crucial role in tissue damage following reperfusion or cerebral ischemia (48). Furthermore, TNF-? can activate microglia and macro-phages to induce the production of inflammatory metabolites, sustain and enhance the inflammatory response, and consequently resulted in brain tissue re-injury (49). Additionally, TNF-? has a toxic effect on capillary blood vessels directly, which can lead to endothelial cell damage and subsequently result in microarterial spasms, increased capillary permeability, aggregation of peripheral leukocyte infiltration, and brain oedema (32).
Aquaporin-4 (AQP-4) and Cerebral Oedema
Aquaporins are small integral membrane proteins with a molecular weight of less than 30,000 kDa. They have high permeability and play a role in fluid transport across cell plasma membranes. They are widely found in various tissues of the body (50). An aquaporin molecule consists of six transmembrane protein helices that form a selective water-permeable channel, arranged as a tetramer in the central part of the molecule. Overall, there are 11 types of aquaporins. Two main types of aquaporins, namely AQP-1 and AQP-4 primarily play a role in the homeostasis of fluid in the CNS, which involved in fluid transport in various compartments of brain tissue, including the CSF or cerebral ventricles, the subarachnoid space, the brain parenchyma consisting of intracellular and extracellular spaces, and the intravascular compartmen (33,34). The expression of AQP is limited to astrocytes widely distributed in brain tissue and the spinal medulla, as well as ependymal cells (51).
The expression of AQP-4 is known to have a role in the progression of TBI. Research on animal models has shown that AQP-4 expression increases at the site of brain injury (52). Suppression of the AQP-4 gene has been found to result in reduced oedema symptoms 24 hours after cerebral ischemia. Autopsy studies on human brain tissue have revealed increased AQP-4 immunoreactivity after cerebral infarction (53). A study found that AQP-4 levels increase from 15 hours to eight days after brain injury (54). There is a significant correlation between the increased AQP-4 expression and BBB damage (55). This increase continues for up to 15 hours after head trauma, with the highest increase at eight hours post-trauma (56). This aligns with experiments on animal models, where AQP-4 expression increases four hours after trauma and starts to decrease at 24 hours post-trauma. The severity of injury is associated with increased AQP-4 regulation and interstitial oedema (57).
Research on animal models indicates that AQP inhibitors have clinical potential as diuretics to address cerebral oedema (58). Consistent with this, AQP-4 deletion is known to be protective against vasogenic cerebral oedema (59). Another study suggests that AQP-4 has a dual role in brain fluid transport, regulating both entry and exit (60). Although different results have been obtained for the two types of oedema, some studies suggest that AQP-4 could be targeted for managing cerebral oedema. A study found that AQP-4 is not only involved in oedema formation but also in excess fluid absorption (50). If administered early, AQP-4 inhibitors can reduce cytotoxic oedema which slowing the entry of oedema fluid into brain parenchyma. Late administration of AQP-4 inhibitors in cytotoxic oedema is suspected to worsen cerebral oedema (50,51). Given AQP-4's role in modulating brain fluid, some research suggests that decreasing AQP4 expression could be a new therapeutic option for cerebral oedema (61).
Composition and Benefits of Trigona Honey
Honey is one of known as a natural sweetener used in the food industry worldwide. Honey bees produces these honey, which is a thick liquid containing various molecules such as water (15-17%), glucose and fructose (80-85%), protein, and amino acids (0.1-0.4%). The composition of honey varies widely depending on the type of plants visited by bees or used as a source of nectar (62).
Common compounds found in honey including phenolic acids such as ferulic, ellagic, caffeic, and p-coumaric acids; antioxidants such as superoxide dismutase (SOD), catalase (CAT), tocopherols, ascorbic acid, and reduced glutathione (GSH); as well as flavonoids like galangin, hesperetin, apigenin, chrysin, kaempferol, pinocembrin, and quercetin. Some studies suggest that honey has effects of antioxidant, anti-inflammatory, antibacterial, antiviral, antiulcer, anti-hyperlipidemic, antidiabetic, and anticancer agent (62). Honey works effectively to inhibit the growth of bacteria such as Helicobacter pylori, Shigella spp., Escherichia coli, and Salmonella spp. (62). The osmotic effect, hydrogen peroxide, acidity, and phytochemical factors of honey provides antibacterial activity. The high osmolarity of honey can draw water from micro-organisms, limiting their survival. The acidity of honey ranges from pH 3.2-4.9, which can inhibit pathogens that are not acid-tolerant. Honey can maintain a wound in a sterile condition, allowing molecules that act as antioxidants and antimicrobials to function effectively (63,64).
Stingless bee honey (SBH) is a special type of honey produced by stingless bees, specifically Trigona species. These bees use biting as a defense mechanism instead of stinging, which has been reduced. Trigona SBH is produced by stingless bees known as Trigona species. These bees use biting as a defense mechanism in place of the reduced stinging capability. Klanceng bees belong to the Animalia kingdom, Arthropoda phylum, Insecta class, Hymenoptera order, Apidae family, Trigona genus, and Trigona species (65).
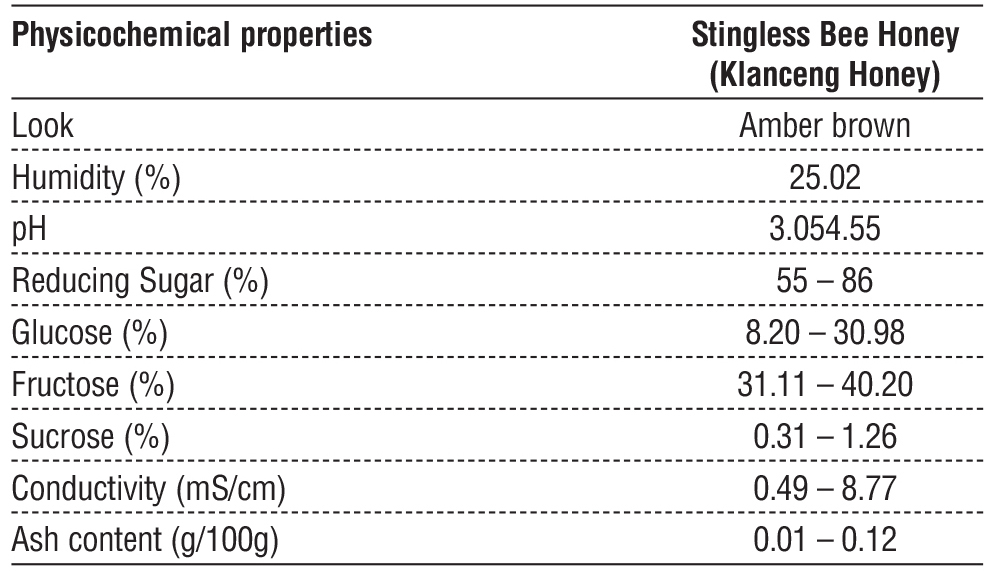
Trigona SBH is characterized by its thinner consistency and amber-brown color. The Trigona SBH taste tends to be more acidic, with a pH ranging from 3.05 to 4.55. The moisture content of klanceng honey is relatively higher, ranging from 30% to 35%. The physicochemical properties of klanceng honey can be observed in (table 1) (66).
Table 1 - Physicochemical properties of trigona honey
Trigona SBH is one of the subtype of stingless bee honey from the genus Trigona spp. According to several physicochemical criteria, the composition of SBH differs from that of other species where (SBH) have higher polyphenol content than any other honey. Trigona SBH is high in total phenolic content and exhibits strong antioxidant, anti-inflammatory, and genoprotective effects. In a previous in vitro study, SBH was shown to reduce levels of interleukin (IL)-6, tumor necrosis factor-a (TNF-a), and interferon in LPS-stimulated macropha (16). Additionally, following LPS exposure, SBH improved antioxidant status and inhibited NF-kB and MAPK signaling pathways in various rat tissues (67,68). These findings suggest that SBH may be beneficial for managing various inflammatory conditions.
Trigona honey contains compounds including caffeic acid and ferulic acid, which inhibit pro-inflammatory enzymes like COX-2 and limit cytokine production, including TNF-a and IL-6. This reduces inflammation, a leading cause of brain Oedema. Trigona honey's strong phenolic and flavonoid content reduces oxidative stress, a key element in brain Oedema progression. Trigona honey stimulates Nrf2, a transcription factor involved in antioxidant defense. This increases the production of endogenous antioxidants such as glutathione, which protects neurons and maintains BBB integrity (16,69).
Effects of Trigona Honey on Brain Oedema in TBI
The anti-inflammatory properties or effects of honey are due to its phenolic compounds. Phenolic compounds can inhibit the excessive production of inflammatory mediators such as NO, TNF, and prostaglandin E2 (PGE2). Phenolic compounds also act as free radical scavengers, thereby protecting cells from cytotoxicity induced by inflammatory mediators (70). Trigona honey also reduces oedema that occurs during inflammation and decreases the microvascular hydrostatic pressure in wound tissue, ensuring unimpeded access of oxygen and nutrients for wound healing (63). Anti-inflammatory agents share a similar correlation with antioxidants in terms of addressing reactive oxygen species (ROS). Trigona honey, containing phenolic compounds and antioxidants, enhances its ability to combat the inflammatory process (66).
Caffeic acid and ferulic acid inhibit the activity of cyclooxygenase-2 (COX-2) and inducible nitric oxide synthase (iNOS), enzymes that play critical roles in the inflammatory process, leading to tissue swelling and damage to the blood-brain barrier (BBB) (69,71,72). These phenolic compounds also suppress the release of pro-inflammatory cytokines such as TNF-a, IL-1b, and IL-6, which are key drivers of inflammation and oedema in brain tissues. Additionally, their strong antioxidant properties enable them to neutralize reactive oxygen species (ROS), protecting the BBB and surrounding neural structures from oxidative stress that worsens swelling. By reducing inflammation, preserving the integrity of the BBB, and preventing fluid leakage, these actions effectively combat brain oedema. Ongoing clinical research aims to better understand their potential for neuroprotection (16,69,72).
Kalantari et.al found honey’s ability to suppress TNF-a, IL-1b, and IL-6 mRNA expression in rat model suggests its potential as a complementary therapy for inflammatory and infectious diseases, particularly those involving hyperactivation of the immune response (73). In a study by Sairazi, honey was found to reduce the expression of TNF-a and IL-1b in a rat model subjected to kainic acid-induced status epilepticus (74). These findings suggest its potential as a neuroprotective agent for epilepsy and other neurodegenerative conditions involving inflammation and oxidative stress.
By reducing the levels of TNF-a and IL-1b, honey’s anti-inflammatory effects may counteract this downregulation, helping to stabilize or restore normal aquaporin expression. This restoration could be particularly beneficial in conditions where aquaporin dysregulation contributes to pathological fluid accumulation or oedema, such as in neuroinflammatory diseases or brain injuries. (75) Honey generates hydrogen peroxide (H?O?), which is transported into cells via Aquaporin-3 (AQP3) channels. Once inside the cells, H?O? stimulates an increase in intracellular calcium levels by facilitating the influx of calcium from the extracellular environment. This process is crucial for activating signaling pathways that drive wound healing and reduce inflammatory process (75). Disrupting AQP3 function or neutralizing H?O? significantly reduces calcium influx, highlighting the indispensable role of AQP3 in mediating honey’s effects on cellular processes involved in tissue repair and reduce inflamation. This finding underscores the importance of AQP3 in the biological mechanisms behind honey-mediated wound healing (73,75).
Furthermore, honey’s rich content of flavonoids and phenolic acids not only modulates inflammatory signaling pathways but also reduces oxidative stress, which is another factor that can adversely impact aquaporin function. Thus, through its combined anti-inflammatory and antioxidant actions, honey supports the normalization of aquaporin activity, promoting tissue recovery and fluid regulation in affected areas. This multifaceted mechanism highlights honey’s potential as a therapeutic agent in managing conditions associated with cytokine-mediated aquaporin dysregulation.
CONCLUSION
This review highlights the potential of Trigona honey as a promising adjunctive therapeutic agent for reducing cerebral edema in TBI patients. Its efficacy is posited to stem from the modulation of AQP-4 mRNA expression and suppression of TNF-? levels, critical mediators in cerebral edema pathophysiology. While existing evidence, particularly concerning its rich phenolic compounds, supports its neuroprotective attributes, rigorous randomized clinical trials are imperative to elucidate precise therapeutic mechanisms and validate its clinical efficacy in TBI management. Future research in this domain offers a novel avenue for improving outcomes and accelerating recovery in TBI patients.
Author’s Contributions
Conceptualization: WA, AAI, DW, NAL, WH, ARB, RG, JO, and MF; Data curation: WA, AAI, DW, and NAL; Formal analysis: WA, AAI, and DW; Funding acquisition: WA, AAI, DW, NAL, and WH; Investigation: WA, AAI, DW, NAL, and WH; Methodology: WA, AAI, DW, NAL, and WH; Project administration: ARB, RG, JO, and MF; Resources: ARB, RG, JO, and MF; Software: ARB, RG, JO, and MF; Supervision: WA, AAI, and DW; Validation: ARB, RG, JO, and MF; Visualization: ARB, RG, JO, and MF; Roles/Writing - original draft: WA, AAI, DW, NAL, WH, ARB, RG, JO, and MF; and Writing - review & editing: WA, AAI, DW, and MF. All authors read and have approved the final version of the manuscript.
Conflict of interests
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Ethical statement
Ethics of the research achieved from Universitas Hasanuddin Ethic Committee on April 17th 2023 (Number 249/UN4.6.4.5.31/PP36/2023)
REFERENCES
1. Jha RM, Kochanek PM, Simard JM. Pathophysiology and treatment of cerebral edema in traumatic brain injury. Neuropharmacology. 2019;145(Pt B):230-246.
2. Clément T, Rodriguez-Grande B, Badaut J. Aquaporins in brain edema. J Neurosci Res. 2020;98(1):9-18.
3. Dostovic Z, Dostovic E, Smajlovic D, Avdic O. Brain Edema After Ischaemic Stroke. Med Arch. 2016;70(5):339-341.
4. Winkler EA, Minter D, Yue JK, Manley GT. Cerebral Edema in Traumatic Brain Injury. Neurosurg Clin N Am. 2016;27(4):473-488.
5. Esquenazi D, Alvim IMP, Pinheiro RO, et al. Correlation between Central Memory T Cell Expression and Proinflammatory Cytokine Production with Clinical Presentation of Multibacillary Leprosy Relapse. PLoS One. 2015;10(5):e0127416.
6. Mohanty S, Swain SK, Biswal C. Brain Edema: Newer Concept of Treatment. Ann Natl Acad Med Sci. 2019;55(04):189-192.
7. Vezzani A, Sperk G, Colmers WF. Neuropeptide Y: emerging evidence for a functional role in seizure modulation. Trends Neurosci. 1999;22(1):25-30.
8. Kim SY, Buckwalter M, Soreq H, Vezzani A, Kaufer D. Blood-brain barrier dysfunction-induced inflammatory signaling in brain pathology and epileptogenesis. Epilepsia. 2012;53 Suppl 6:37-44.
9. Arulselvan P, Fard MT, Tan WS, Gothai S, Fakurazi S, Esa Norhaizan M, et al. Role of Antioxidants and Natural Products in Inflammation. Oxid Med Cell Longev. 2016:2016:5276130.
10. Maroli N, Kalagatur NK, Bhasuran B, Jayakrishnan A, Ramalingam Manoharan R, Kolandaivel P, et al. Molecular Mechanism of T-2 Toxin-Induced Cerebral Edema by Aquaporin-4 Blocking and Permeation. J Chem Inf Model. 2019;59(11):4942-4958.
11. Moretti R, Chhor V, Titomanlio L, Fleiss B, Gressens P. Brain Edema in Developing Brain Diseases. In: Brain Edema. Elsevier; 2017. p. 393-429.
12. Adini A, Adini I, Chi ZL, Derda R, Birsner AE, Matthews BD, et al. A novel strategy to enhance angiogenesis in vivo using the small VEGF-binding peptide PR1P. Angiogenesis. 2017;20(3):399-408.
13. Zulkifli NA, Hassan Z, Mustafa MZ, Azman WNW, Hadie SNH, Ghani N, et al. The potential neuroprotective effects of stingless bee honey. Front Aging Neurosci. 2023;14(1):1048028.
14. Iftikhar A, Nausheen R, Muzaffar H, Naeem MA, Farooq M, Khurshid M, et al. Potential Therapeutic Benefits of Honey in Neurological Disorders: The Role of Polyphenols. Molecules. 2022;27(10):3297.
15. Biluca FC, da Silva B, Caon T, Bramorski Mohr ET, Nicácio Vieira G, Valdemiro Gonzaga L, et al. Investigation of phenolic compounds, antioxidant and anti-inflammatory activities in stingless bee honey (Meliponinae). Food Res Int. 2020;129:108756.
16. Ranneh Y, Akim AM, Hamid HA, Khazaai H, Fadel A, Mahmoud AM. Stingless bee honey protects against lipopolysaccharide induced-chronic subclinical systemic inflammation and oxidative stress by modulating Nrf2, NF-kB and p38 MAPK. Nutr Metab (Lond). 2019; 16(1):15.
17. Saraiva C, Praça C, Ferreira R, Santos T, Ferreira L, Bernardino L. Nanoparticle-mediated brain drug delivery: Overcoming blood-brain barrier to treat neurodegenerative diseases. J Control Release. 2016; 235:34-47.
18. Yap KE, Islam AA, Ihwan A, Baan JAB, Hamid F. Comparison of Helsinki CT and Rotterdam CT Scoring Systems As Prognostic Factors of Brain Injury. Nusantara Medical Science Journal. Published
Full Text Sources:
Abstract:
Views: 441

For Authors
Journal Subscriptions

Dec 2025
Supplements
Instructions for authors
Online submission
Contact
e-ISSN: 2601 - 1700 (online)
ISSN-L: 2559 - 723X
Journal Abbreviation: Surg. Gastroenterol. Oncol.
Surgery, Gastroenterology and Oncology (SGO) is indexed in:
- SCOPUS
- EBSCO
- DOI/Crossref
- Google Scholar
- SCImago
- Harvard Library
- Open Academic Journals Index (OAJI)
Surgery, Gastroenterology and Oncology (SGO) is an open-access, peer-reviewed online journal published by Celsius Publishing House. The journal allows readers to read, download, copy, distribute, print, search, or link to the full text of its articles.
Time to first editorial decision: 25 days
Rejection rate: 61%
CiteScore: 0.2

Meetings and Courses in 2025
Meetings and Courses in 2024
Meetings and Courses in 2023
Meetings and Courses in 2022
Meetings and Courses in 2021
Meetings and Courses in 2020
Meetings and Courses in 2019
Verona expert meeting 2019

Surgery, Gastroenterology and Oncology applies the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits readers to copy and redistribute the material in any medium or format, remix, adapt, build upon the published works non-commercially, and license the derivative works on different terms, provided the original material is properly cited and the use is non-commercial. Please see: https://creativecommons.org/licenses/by-nc/4.0/
Publisher’s Note:
The opinions, statements, and data contained in article are solely those of the authors and not of Surgery, Gastroenterology and Oncology journal or the editors. Publisher and the editors disclaim responsibility for any damage resulting from any ideas, instructions, methods, or products referred to in the content.
 IASGO Society News
IASGO Society News