Surgery, Gastroenterology and Oncology

|
|
Background: Rectal cancer is a common malignancy worldwide. Management of rectal cancer is multimodal. Preoperative chemoradiotherapy, particularly for low rectal cancer, plays an important role in downstaging and facilitating surgery. Objective: This study aimed to evaluate the outcomes of preoperative chemoradiotherapy in patients with low rectal
cancer at Hanoi Medical University Hospital.
Materials and Methods: A retrospective study of 56 patients with low rectal cancer who received preoperative chemoradiotherapy at Hanoi Medical University Hospital from January 2021 to June 2023.
Results: The mean age was 57.62±10.02 years, with a male-to-female ratio of 1.67:1. Rullier type I low rectal cancer had the highest proportion, at 69.4%. Before radiotherapy, tumors were predominantly stage II and III in 14.3% and 83.9% of cases, respectively; after radiotherapy, there was a marked shift to stages I and II with rates of 26.8% and 25.0%, respectively. T-stage downstaging occurred in 53.6% and nodal (N) downstaging in 58.9%. The mean tumor size decreased from 37.0±17.36 mm before radiotherapy to 25.3±14.08 mm after radiotherapy (the tumor size reduction rate was 89.3%). Regarding pathological response, 19.6% of patients achieved pCR (TRG=4) and 32.1% had a good response (TRG 3-4). On imaging, 35.7% (mrTRG 1-2) showed substantial tumor regression.
Conclusions: Preoperative chemoradiotherapy is an effective treatment and provides safe surgical outcomes for patients with locally advanced rectal cancer.
INTRODUCTION
Rectal cancer is among the most common gastro-intestinal malignancies and has shown a marked increase in recent years (1). At diagnosis, many patients present with locally advanced disease, with tumors invading the rectal wall or adjacent structures, limiting the possibility of curative resection and sphincter preservation (2). Optimizing treatment strategies - particularly low rectal cancer - to increase response rates, improve local control, and enhance survival outcomes is a key objective of contemporary multimodal therapy (3). Currently, neoadjuvant chemoradiotherapy has become the standard of care for locally advanced rectal cancer. The classical regimen is concurrent chemoradiotherapy (CCRT) with 5-fluorouracil (5-FU) orcapecitabine, which enables tumor downsizing, improves the likelihood of sphincter preservation, and reduces the rate of local recurrence (4). In recent years, multiple studies have demonstrated the advantages of the total neoadjuvant therapy (TNT) strategy - in which patients receive systemic chemotherapy before or after the radiotherapy phase, followed by surgery. Trials such as RAPIDO and PRODIGE-23 have shown that TNT significantly increases the rate of pathologic complete response (pCR), provides better control of micrometastatic disease, and tends to improve long-term survival (5,6). At Hanoi Medical University Hospital, during the early phase of managing locally advanced rectal cancer, most patients were treated with concurrent chemoradiotherapy. However, there is now a clear shift toward wider adoption of total neoadjuvant therapy (TNT). Accordingly, reassessing the real-world effectiveness of these two strategies in the Vietnamese setting is warranted to provide evidence for choosing the optimal treatment approach in routine clinical practice.
MATERIALS AND METHODS
Study Population
Patients with a confirmed diagnosis of low rectal cancer (JSCCR classification) who received preoperative (neoadjuvant) chemoradiotherapy followed by sphincter-preserving laparoscopic surgery at Hanoi Medical University Hospital between January 2021 and June 2023.
Inclusion criteria
• Confirmed low rectal cancer (JSCCR classification).
• Received neoadjuvant chemoradiotherapy prior to sphincter-preserving laparoscopic surgery.
• Medical records contained all variables required for the study.
Exclusion criteria
• Not operated on after neoadjuvant chemoradiotherapy.
• Underwent non-sphincter-preserving surgery after neoadjuvant chemoradiotherapy.
• Incomplete medical records lacking required study information.
• The patient discontinued chemoradiotherapy due to toxicity.
• Rectal cancer with acute complications.
Study Design
Retrospective descriptive study.
Data Collection and Statistical Analysis
Data were entered, cleaned, and analyzed using SPSS version 20.0. Statistical methods included:
Comparison of means: Student’s t-test (two-sided; p < 0.05).
Comparison of proportions: chi-square test (p < 0.05); Fisher’s exact test when expected counts were < 5.
Quantitative variables: reported as mean ± standard deviation (SD).
Qualitative variables: reported as counts and percentages.
Statistical significance was set at ? = 0.05.
Ethical Considerations
This retrospective descriptive study used fully anonymized data extracted from electronic medical records, with no collection of identifiable personal information and no intervention in patient care. In accordance with the policies of Hanoi Medical University Hospital, institutional ethics committee/IRB review was not required and informed consent was waived. All procedures complied with institutional data-protection regulations and the ethical principles of the 1964 Declaration of Helsinki and its later amendments.
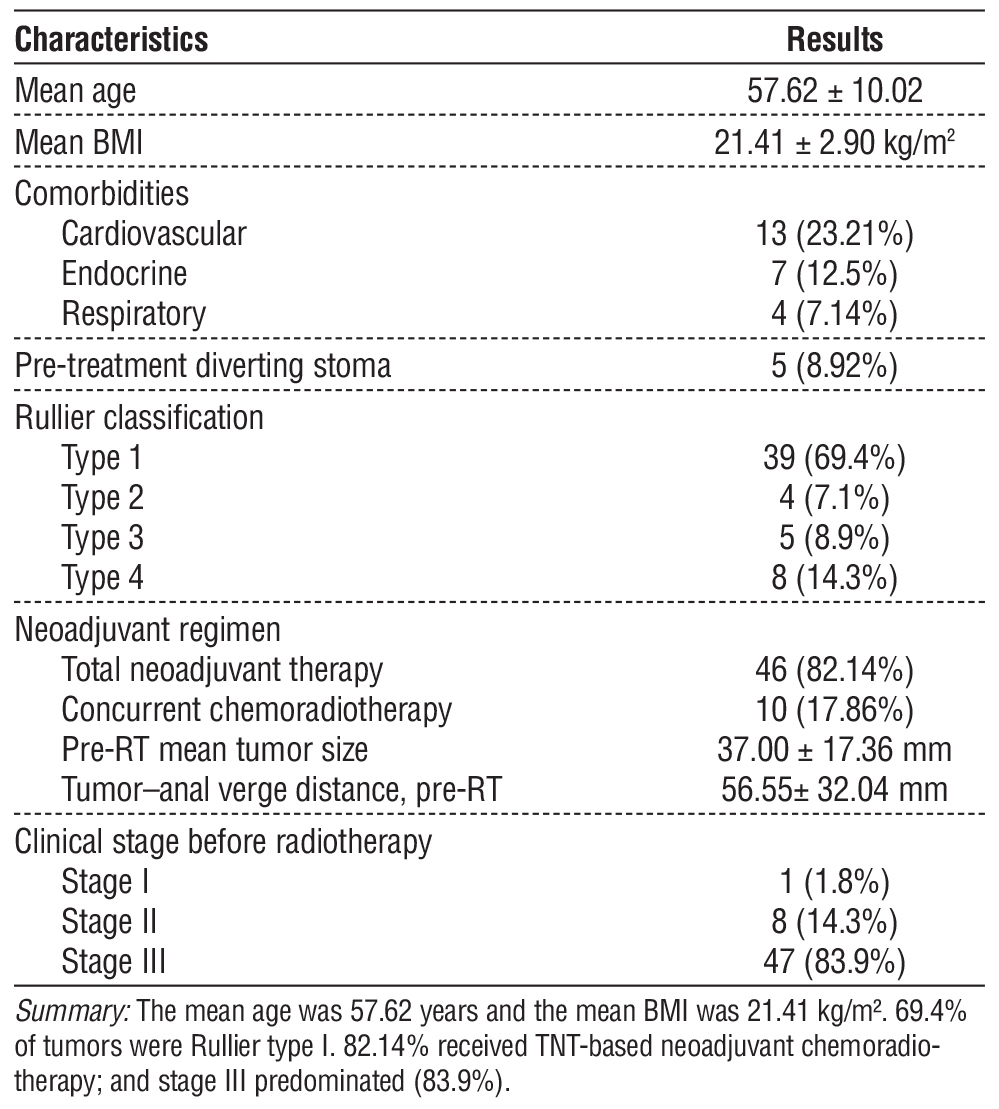
Table 1 - Characteristics of the study population
RESULTS
Characteristics of the Study Population(table 1)
Outcomes of Preoperative Chemoradiotherapy (table 2)
DISCUSSION
Characteristics of the study population
The mean age of patients in this study was 57.62 ± 10.02 years, consistent with the general pattern that rectal cancer is most commonly seen in individuals aged 50-70 years. According to GLOBOCAN 2022, the incidence of colorectal cancer increases with age, peaking in those older than 60 years (1). Another European study by Vuik et al. likewise confirmed the rising incidence in older patients; however, it also documented a substantial increase among individuals under 50 over the past 25 years, particularly in high-income countries (7).
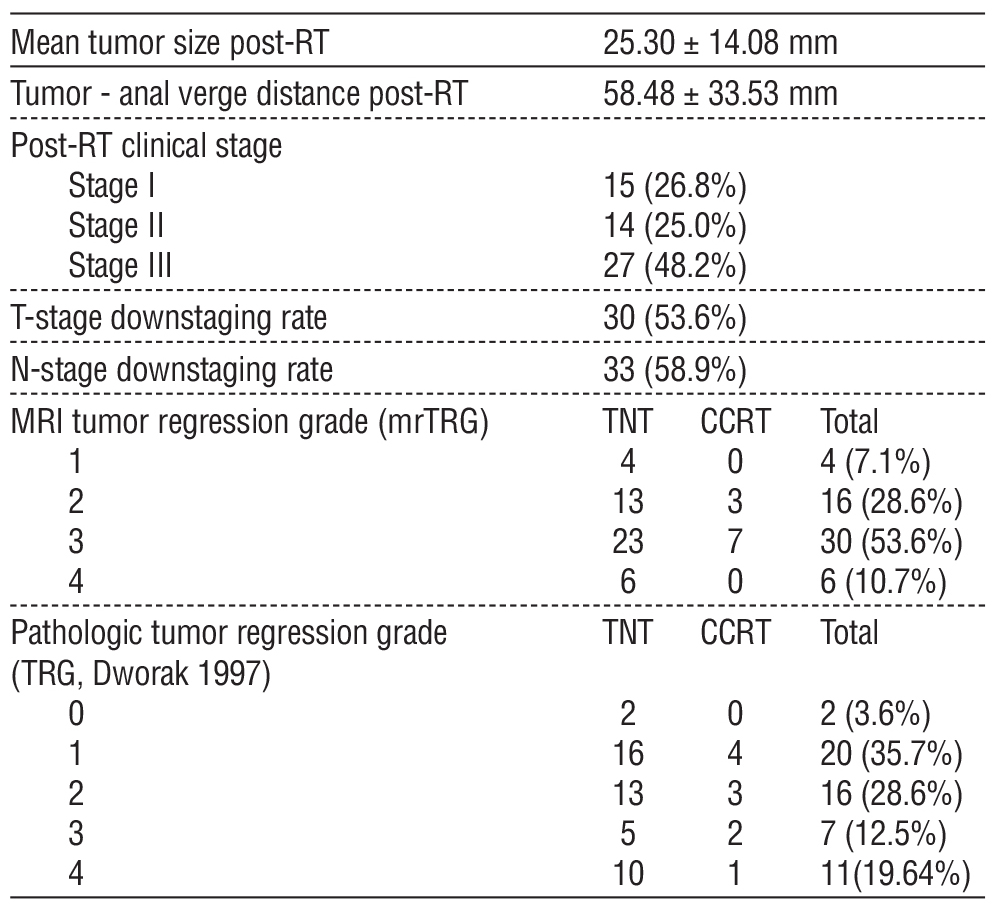
Table 2 - Outcomes of preoperative chemoradiotherapy
The mean BMI of the cohort was 21.41 ± 2.90 kg/m²; 85.18% had normal or good nutritional status, while only 14.82% were underweight (BMI<18.5 kg/m²). These findings indicate a relatively stable pre-treatment nutritional state, which can enhance tolerance to multimodal therapy. In 2021, a study from Taiwan reported that colorectal cancer patients with low BMI (< 18.5 kg/m²) had higher mortality than those with normal BMI, underscoring the prognostic importance of nutritional status (8).
Regarding medical history, the prevalences of cardiovascular, endocrine, and respiratory comorbidities were 23.2%, 12.5%, and 7.1%, respectively. Such coexisting conditions may impair tolerance to chemo-radiotherapy, increase the risk of complications, and diminish treatment efficacy. Michalopoupou (2021), in a study of 1,076 patients with colorectal cancer, reported that the presence of non-cancer comorbidities at diagnosis significantly reduced 10-year survival, particularly among those with two or more comorbidities (9). Similarly, Hang Qiu et al. (2023) reported that 75.5% of colorectal cancer patients in China had at least one comorbidity - most commonly hypertension, chronic obstructive pulmonary disease, and chronic liver disease - which influenced treatment strategies and survival outcomes (10).
Additionally, in this study, 8.92% of patients underwent creation of a diverting stoma prior to treatment. In locally advanced rectal cancer, pre-treatment diversion before neoadjuvant chemo-radiotherapy is typically indicated for impending obstruction or other acute complications. Several studies have suggested that a diverting stoma can improve the patient’s general condition and facilitate delivery of chemoradiotherapy. However, diversion has potential downsides: Sandén G. (2023), in a cohort of 105 patients, found that pre-treatment diversion prolonged the waiting time to initiate therapy (median 51 vs 36 days in those without diversion) and was associated with a higher complication rate (75% vs 29.6%) (11). On the other hand, a diverting stoma has proven effective in preventing one of the most serious complications after rectal surgery - anastomotic leak. In the analysis by Myrseth et al. (2022) of 1,018 patients, the reoperation rate due to anastomotic leak decreased from 7.8% to 2.3% in the diverted group compared with the non-diverted group (p < 0.001). There were no differences in overall reoperation rate or overall complications between groups. Multivariable regression showed that the absence of diversion increased the risk of anastomotic leak (adjusted OR 3.77, 95% CI 1.97-7.24) (12). Therefore, the decision to create a diverting stoma prior to neoadjuvant chemoradiotherapy should be carefully individualized to each patient’s clinical status, with the aim of optimizing treatment delivery and minimizing complications
Outcomes of Preoperative Chemoradiotherapy
In this study, most patients were treated with a total neoadjuvant therapy (TNT) regimen (82.14%), while the remaining 17.86% received conventional concurrent chemoradiotherapy. This trend is consistent with current recommendations from multiple oncology societies, particularly following large-scale clinical trials such as RAPIDO and PRODIGE-23, which have shown that TNT significantly increases the pathologic complete response (pCR) rate, improves local control, and reduces the risk of distant metastasis (5,6).
Following completion of neoadjuvant chemoradiotherapy, the mean tumor size decreased from 37.00 ± 17.36 mm to 25.30±14.08 mm, a statistically significant difference (p < 0.05). Radiologic tumor shrinkage was observed in 89.3% of patients. With respect to staging, 53.6% experienced T-stage downstaging and 58.9% had N-stage downstaging, indicating a substantial clinical and imaging response. These findings are highly encouraging, reinforcing the effectiveness of neo-adjuvant therapy in reducing tumor burden, improving the likelihood of curative resection, and contributing to local control and control of micrometastatic disease. Notably, recent trials such as RAPIDO and PRODIGE-23 have shown that total neoadjuvant therapy (TNT) not only improves downstaging but also increases the pathologic complete response (pCR) rate, with reported pCR rates of 25-30% (5,6).
In this study, the mean distance from the inferior tumor edge to the anal verge was 56.55±32.04 mm before treatment, increasing slightly to 58.48±33.53 mm after treatment. Although this change was not statistically significant (p>0.05), a shift in tumor position relative to the anal sphincter complex may facilitate sphincter-preserving procedures instead of abdominoperineal resection (APR). The tumor-anal verge distance is a key factor in surgical decision-making. Historically, low rectal tumors located <5 cm from the anal verge were commonly treated with APR. However, recent studies indicate that, with precise assessment of invasion and distance, sphincter preservation can be performed safely. Specifically, Rullier et al. showed that for tumors 1.5-4.5 cm from the anal verge without external sphincter invasion, curative sphincter-preserving surgery using intersphincteric resection (ISR) combined with total mesorectal excision (TME) is feasible, achieving high local control (local recurrence ~2%) and a 5-year survival of 81%, comparable to classic APR. This evidence supports that, when carefully evaluated, tumor-anal verge distance is no longer an absolute constraint to sphincter preservation (13). Therefore, precise assessment of the tumor -anal verge distance, combined with imaging modalities such as MRI and endorectal ultrasonography, is essential to determine the feasibility of sphincter preservation - particularly in light of the persistent limitations of imaging in accurately evaluating sphincter invasion.
Assessment of post-chemoradiotherapy response by MRI using the mrTRG system and histopathologic response using the TRG system are two key tools for prognostication and treatment planning in rectal cancer. In this study, the rate of good-to-excellent
MRI response (mrTRG 1-2) was 35.7%, whereas the proportion with complete or near-complete pathologic response (TRG 3-4 per Dworak) was 32.14%. The association between MRI response and histopathology was statistically significant (p=0.014), indicating meaningful correlation, though not perfect concordance. This discordance has been noted in prior studies and may be attributable to fibrosis, inflammation, or post-radiation signal changes that confound MRI-based grading. Sclafani et al. (2017) reported that the sensitivity of MRI for detecting pathologic complete response (pCR) is approximately 74.4%, largely due to such post-treatment changes (14). Thus, although MRI is a useful tool for predicting treatment response, MRI-based assessment should be regarded only as an initial guide. Final clinical decisions should integrate clinical evaluation, rectal endoscopy, and biopsy-especially when considering organ-preserving strategies such as a watch-and-wait (W&W) approach.
Figure 1 - MRI images of a mid-rectal tumor before and after treatment in a 53-year-old male patient on high-resolution T2-weighted sequences.
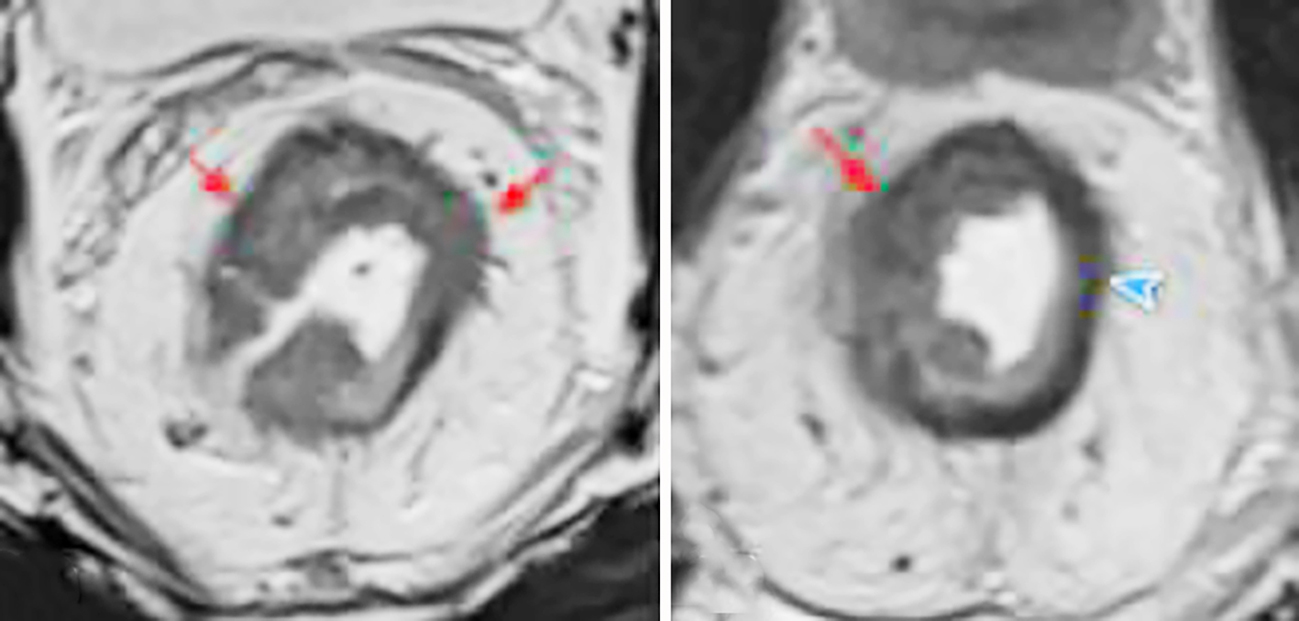
(a) The mid-rectal tumor shows high signal intensity on T2W images (red arrow).
(b) Post-treatment images demonstrate mixed signal intensity: fibrosis predominates, showing markedly low T2 signal (blue arrow), while a residual tumor component with intermediate T2 signal remains (red arrow); corresponding to mrTRG 3.
In our study, the rate of pathologic complete response (pCR; TRG 4 per the Dworak system) after TNT was 21.7% (10/46), higher than 10.0% (1/10) in the CCRT group. Similarly, the proportion achieving mrTRG 1–2 on MRI was 37.0% (17/46) with TNT versus 30.0% (3/10) with CCRT. These findings suggest that TNT confers an advantage in enhancing tumor regression compared with conventional CCRT. Our results are consistent with prior large trials: the RAPIDO trial reported a pCR rate of 28.4% with TNT versus 14.3% with standard CRT, and the PRODIGE-23 trial reported 27.5% with TNT versus 11.7% with CRT (5, 6). However, the pCR (TRG 4) rate in our study was only 19.6%, lower than the pooled average of 22.4% reported in the meta-analysis by Petrelli et al (2020)(15). This discrepancy may be attributable to several factors, including:
Suboptimal interval from neoadjuvant chemo-radiotherapy to surgery: in our cohort the wait was typically 6-8 weeks, whereas many studies recommend ? 10-12 weeks to increase the pCR rate.
Differences in histopathologic processing and TRG assessment criteria: inter-center heterogeneity can influence grading outcomes.
Tumor biological characteristics in Vietnamese patients: potential biological differences compared with Western populations that may affect treatment response.
In summary, our findings show a generally favorable response in the TNT cohort, although the complete response rate remains lower than in other studies. The divergence between mrTRG and pathologic TRG underscores the complementary roles of imaging and histopathology in evaluating neoadjuvant treatment response.
Figure 2 - Histopathological images before and after neoadjuvant chemoradiotherapy in a patient with mid-rectal adenocarcinoma. (a) Preoperative biopsy showed moderately differentiated adenocarcinoma. Tumor cells have large nuclei, basophilic cytoplasm, narrow cytoplasm, and are arranged in a tubular glandular invading the stroma (Hematoxylin & Eosin, 40x).
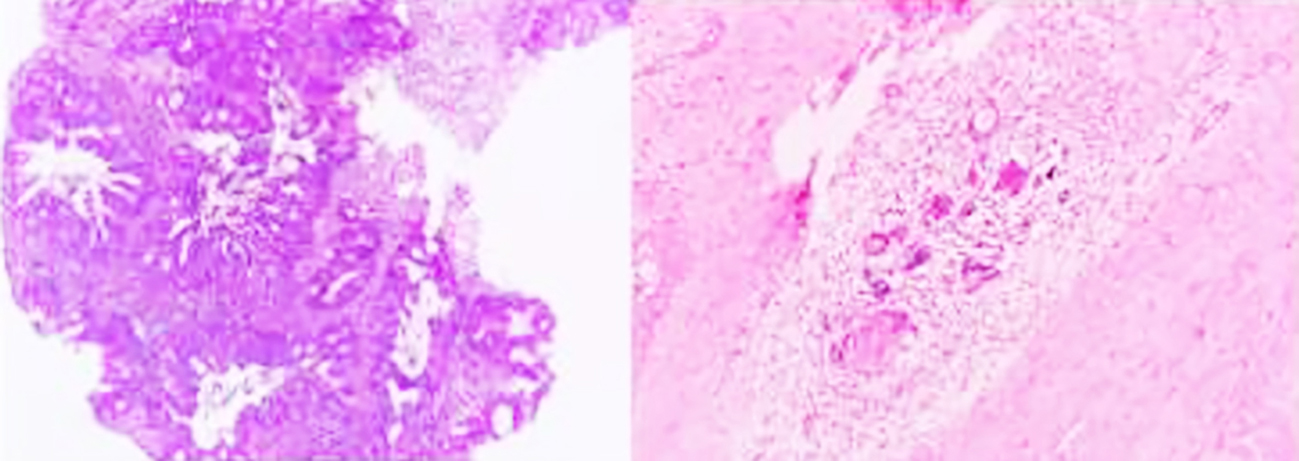
(b) On the surgical specimen after NCRT, adenocarcinoma with stromal inflammatory response, granulomatous inflammatory reaction including multinucleated giant cells, calcificaiton (Hematoxylin & Eosin, 200x).
CONCLUSIONS
Neoadjuvant chemoradiotherapy - particularly total neoadjuvant therapy (TNT) - demonstrates superior effectiveness in shrinking tumors, achieving downstaging, and increasing the likelihood of sphincter preservation without compromising curative oncologic efficacy in patients with locally advanced rectal cancer. As oncologic care continues to shift toward multimodal, function-preserving strategies, the structured and appropriate adoption of TNT can yield further benefits for patients, both in survival outcomes and in post-treatment quality of life.
Acknowledgments
We extend our heartfelt appreciation to the Board of Directors and the Department of Coloproctology & Pelvic Floor Surgery at Hanoi Medical University Hospital for their steadfast support throughout this study. We are also indebted to the physicians, nurses, and allied health professionals whose dedication to data acquisition and coordination made this work possible.
Conflict of Interest
The authors report no conflicts of interest related to the conduct of the research, the preparation of the manuscript, or its publication.
Funding
No external funding was obtained for this study; the work received no financial support from public
agencies, commercial entities, or nonprofit organizations.
REFERENCES
1. Bray F, Laversanne M, Sung H, Ferlay J, Siegel RL, Soerjomataram I, et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2024;74(3):229-263.
2. Sauer R, Liersch T, Merkel S, Fietkau R, Hohenberger W, Hess C, et al. Preoperative versus postoperative chemoradiotherapy for locally advanced rectal cancer: results of the German CAO/ARO/AIO-94 randomized phase III trial after a median follow-up of 11 years. J Clin Oncol. 2012;30(16):1926-33.
3. National Comprehensive Cancer Network - Home. NCCN, https://www.nccn.org.
4. Bosset J-F, Collette L, Calais G, Mineur L, Maingon P, Radosevic-Jelic L, et al. Chemotherapy with preoperative radiotherapy in rectal cancer. N Engl J Med. 2006;355(11):1114-23.
5. Bahadoer RR, Dijkstra EA, van Etten B, Marijnen CAM, Putter H, Meershoek-Klein Kranenbarg E, et al. Short-course radiotherapy followed by chemotherapy before total mesorectal excision (TME) versus preoperative chemoradiotherapy, TME, and optional adjuvant chemotherapy in locally advanced rectal cancer (RAPIDO): a randomised, open-label, phase 3 trial. Lancet Oncol. 2021;22(1): 29-42.
6. Conroy T, Bosset J-F, Etienne P-L, Rio E, François É, Mesgouez-Nebout N, et al. Neoadjuvant chemotherapy with FOLFIRINOX and preoperative chemoradiotherapy for patients with locally advanced rectal cancer (UNICANCER-PRODIGE 23): a multicentre, randomised, open-label, phase 3 trial. Lancet Oncol. 2021;22(5):702-715.
7. Vuik FE, Nieuwenburg SA, Bardou M, Lansdorp-Vogelaar I, Dinis-Ribeiro M, Bento MJ, et al. Increasing incidence of colorectal cancer in young adults in Europe over the last 25 years. Gut. 2019;68(10): 1820-1826.
8. Chiu C-C, Ho C-H, Hung C-M, Chao CM, Lai CC, Chen CM, et al. Correlation of Body Mass Index with Oncologic Outcomes in Colorectal Cancer Patients: A Large Population-Based Study. Cancers (Basel). 2021;13(14):3592.
9. Michalopoulou E, Matthes KL, Karavasiloglou N, Wanner M, Limam M, Korol D, et al. Impact of comorbidities at diagnosis on the 10-year colorectal cancer net survival: A population-based study. Cancer Epidemiol. 2021; 73:101962.
10. Qiu H, Wang L, Zhou L, Wang X. Comorbidity Patterns in Patients Newly Diagnosed With Colorectal Cancer: Network-Based Study. JMIR Public Health Surveill. 2023;9:e41999.
11. Sandén G, Svensson J, Ljuslinder I, Rutegård M. Defunctioning stoma before neoadjuvant treatment or resection of endoscopically obstructing rectal cancer. Int J Colorectal Dis. 2023;38(1):24.
12. Myrseth E, Nymo LS, Gjessing PF, Norderval S. Diverting stomas reduce reoperation rates for anastomotic leak but not overall reoperation rates within 30 days after anterior rectal resection: a national cohort study. Int J Colorectal Dis. 2022;37(7):1681-1688.
13. Rullier E, Laurent C, Bretagnol F, Rullier A, Vendrely V, Zerbib F. Sphincter-saving resection for all rectal carcinomas: the end of the 2-cm distal rule. Ann Surg. 2005;241(3):465-9.
14. Sclafani F, Brown G, Cunningham D, Wotherspoon A, Teixeira Mendes S, Balyasnikova S, et al. Comparison between MRI and pathology in the assessment of tumour regression grade in rectal cancer. Br J Cancer. 2017;117(10):1478-1485.
15. Petrelli F, Trevisan F, Cabiddu M, Sgroi G, Bruschieri L, Rausa E, et al. Total Neoadjuvant Therapy in Rectal Cancer: A Systematic Review and Meta-analysis of Treatment Outcomes. Ann Surg. 2020;271(3): 440-448.
Outcomes of preoperativeFull Text Sources:
Abstract:
Views: 438

For Authors
Journal Subscriptions

Dec 2025
Supplements
Instructions for authors
Online submission
Contact
e-ISSN: 2601 - 1700 (online)
ISSN-L: 2559 - 723X
Journal Abbreviation: Surg. Gastroenterol. Oncol.
Surgery, Gastroenterology and Oncology (SGO) is indexed in:
- SCOPUS
- EBSCO
- DOI/Crossref
- Google Scholar
- SCImago
- Harvard Library
- Open Academic Journals Index (OAJI)
Surgery, Gastroenterology and Oncology (SGO) is an open-access, peer-reviewed online journal published by Celsius Publishing House. The journal allows readers to read, download, copy, distribute, print, search, or link to the full text of its articles.
Time to first editorial decision: 25 days
Rejection rate: 61%
CiteScore: 0.2

Meetings and Courses in 2025
Meetings and Courses in 2024
Meetings and Courses in 2023
Meetings and Courses in 2022
Meetings and Courses in 2021
Meetings and Courses in 2020
Meetings and Courses in 2019
Verona expert meeting 2019

Surgery, Gastroenterology and Oncology applies the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits readers to copy and redistribute the material in any medium or format, remix, adapt, build upon the published works non-commercially, and license the derivative works on different terms, provided the original material is properly cited and the use is non-commercial. Please see: https://creativecommons.org/licenses/by-nc/4.0/
Publisher’s Note:
The opinions, statements, and data contained in article are solely those of the authors and not of Surgery, Gastroenterology and Oncology journal or the editors. Publisher and the editors disclaim responsibility for any damage resulting from any ideas, instructions, methods, or products referred to in the content.
 IASGO Society News
IASGO Society News