Surgery, Gastroenterology and Oncology

|
|
Background: The classification criteria for primary gastrointestinal lymphoma have been recently updated. This study aimed to determine the immunophenotype and its association with the clinical features of common gastrointestinal lymphoma types.
Methods: We investigated 159 patients with primary malignant lymphoma of the gastro-intestinal tract from three tertiary referral hospitals. The patients were categorized according to the WHO (2019) classification of digestive system tumors based on histopathology and immunohistochemistry.
Results: The median age was 56.1 (min 11 - max 99) years, with the men/women ratio of 1.5/1. Diffuse large B-cell lymphoma (DLBCL) was the most common type (n = 99, 62.3%). Among these, there were two subtypes: the activated B-cell-like (ABC) subtype (n=55) and the germinal center B-cell-like (GCB) subtype (n=44). Mucosa-associated lymphoid tissue (MALT) was the second most common type (n = 33, 20.1%). The most frequent complaint was abdominal pain (75.5%). B symptoms (n=18, 11.3%) and complications (n=34, 21.4%) were also observed. Aggressive lymphomas were more commonly detected in stages I and IV, whereas stage IV was very rare (3/47 cases) and stage I was the most common type of indolent lymphoma. The lesions in the aggressive lymphoma group were also larger than those in the indolent lymphoma group.
Conclusions: There was a distinct staging pattern between lymphoma types. The lesion size was notably larger in aggressive lymphomas than in indolent lymphomas, aligning with the more severe clinical presentations observed in advanced cases. These findings may enhance early identification and allow tailored therapeutic interventions.
Introduction
Gastrointestinal (GI) lymphomas are relatively rare malignancies, accounting for 1-4% of all GI tumors (1, 2). However, it is the most common extranodal lymphoma, and responds well to treatment at an early stage. Therefore, the detection and accurate diagnosis of gastrointestinal lymphoma are important (2,3). Dawson’s criteria are often used to diagnose primary gastrointestinal lymphoma (2). Currently, the criteria have become less strict, allowing for contiguous involvement of other organs and distant nodal disease, provided that the extranodal tumor is the presenting site and constitutes the predominant disease (WHO). The development of diagnostic techniques, particularly flexible endoscopy, endoscopic ultrasonography, and chromoendoscopy, has increased the detection of gastrointestinal lymphomas. In 2019, the World Health Organization released the 5th Classification of Digestive Tumors in a series of ‘Bluebooks,’ which made the classification of haematolymphoid tumors of the digestive system into a separate category. This classification includes new entities such as monomorphic epitheliotropic Intestinal T-cell lymphoma, duodenal follicular lymphoma, intestinal T-cell lymphoma NOS, and Indolent T-cell lymphoproliferative disorder of the GI tract. The 5th classification of gastrointestinal tumors applies the diagnosis integration of clinical, morpho-logical (cytological and histological), immunopheno-typic, and genetic data. The terms “essential and desirable diagnostic criteria” are introduced, in which, essential criteria are must-have features to establish a diagnosis. The purpose was to encourage medical facilities to take advantage of the available resources to diagnose and classify this disease group. This group of diseases is heterogeneous, with nonspecific clinical manifestations and endoscopic findings, overlapping histopathological characteristics, and epidemiology that vary significantly between geographical regions, making diagnosis challenging. This study aimed to elucidate the immunophenotypes and associations between the clinical features and histopathological types in patients with gastrointestinal lymphoma in Northern Vietnam.
Patients and Methods
Patient Selection and Ethics
We investigated the immunophenotype and clinicopathological characteristics of gastrointestinal lymphomas diagnosed at Vietnam National Cancer Hospital, Hue Central Hospital, and Hue University of Medicine and Pharmacy Hospital between January 2020 and December 2023. Patients with a suspected diagnosis of gastrointestinal lymphoma, based on histopathological evaluation of biopsy or surgical specimens were included. Patients previously diagnosed with gastro-intestinal lymphoma or whose specimens were insufficient for immunohistochemical testing were excluded. We then performed immunohistochemical staining of samples and excluded patients who had a non-gastrointestinal lymphoma diagnosis on immunohistochemistry or samples inadequate for diagnosis. A total of 159 patients were included in this study. Data on age, sex, chief complaint, B symptoms, complications, Ann Arbor Classification stage, lesion location, and tumor size were retrospectively collected from the database of each hospital.
This study was approved by the Institutional Ethics Committee of Hue University of Medicine and Pharmacy (approval number: H2022/024).
Immunohistochemistry and Histopathological Diagnosis
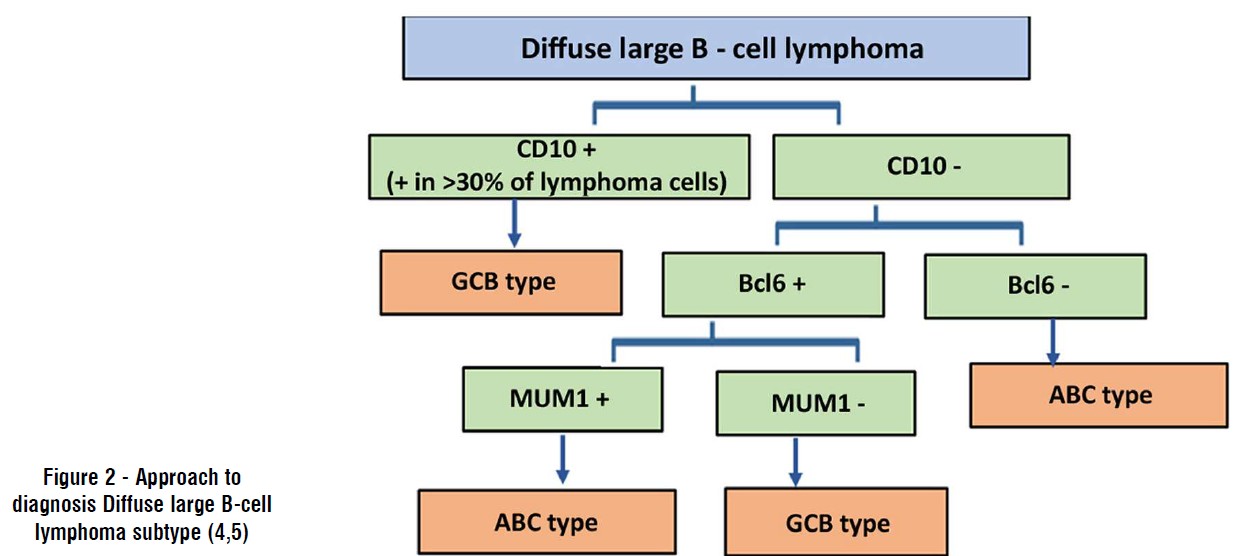
Patients histologically diagnosed with gastro-intestinal lymphoma or suspected gastrointestinal lymphoma underwent immunohistochemistry. The immunohistochemical marker combinations selected were CD3, CD5, CD10, CD20, CD23, CD79a, Bcl-2, Bcl-6, CyclinD1, Ki67, CD4, CD8, and MUM1. In some cases, AE1/3, SOX11, or CD56 were additionally used. Results were assessed by experienced pathologists according to the fifth edition of the WHO classification of digestive system tumors, following M. Pizzi’s approach (4). This approach includes the first step of identifying malignant lymphoid tissue and assessing the morphology and size of lymphoid cells on routine histopathology to select the appropriate immunohistochemical markers for staining. The second step was classification based on the immunohistochemical expression of tumor cells, which were classified according to fig. 1 for lymphomas with small-to-medium-sized cells. Lymphomas with medium-to large-sized cells, diagnosed as diffuse large B-cell lymphoma, were subclassified according to Hans’ formula, as shown in fig. 2. The classification process using immunohistochemistry is always considered compatible with histopathology.
Figure 1 - Immunohistochemical algorithm for the diagnosis of small to medium-sized B-cell NHLs (4,5)
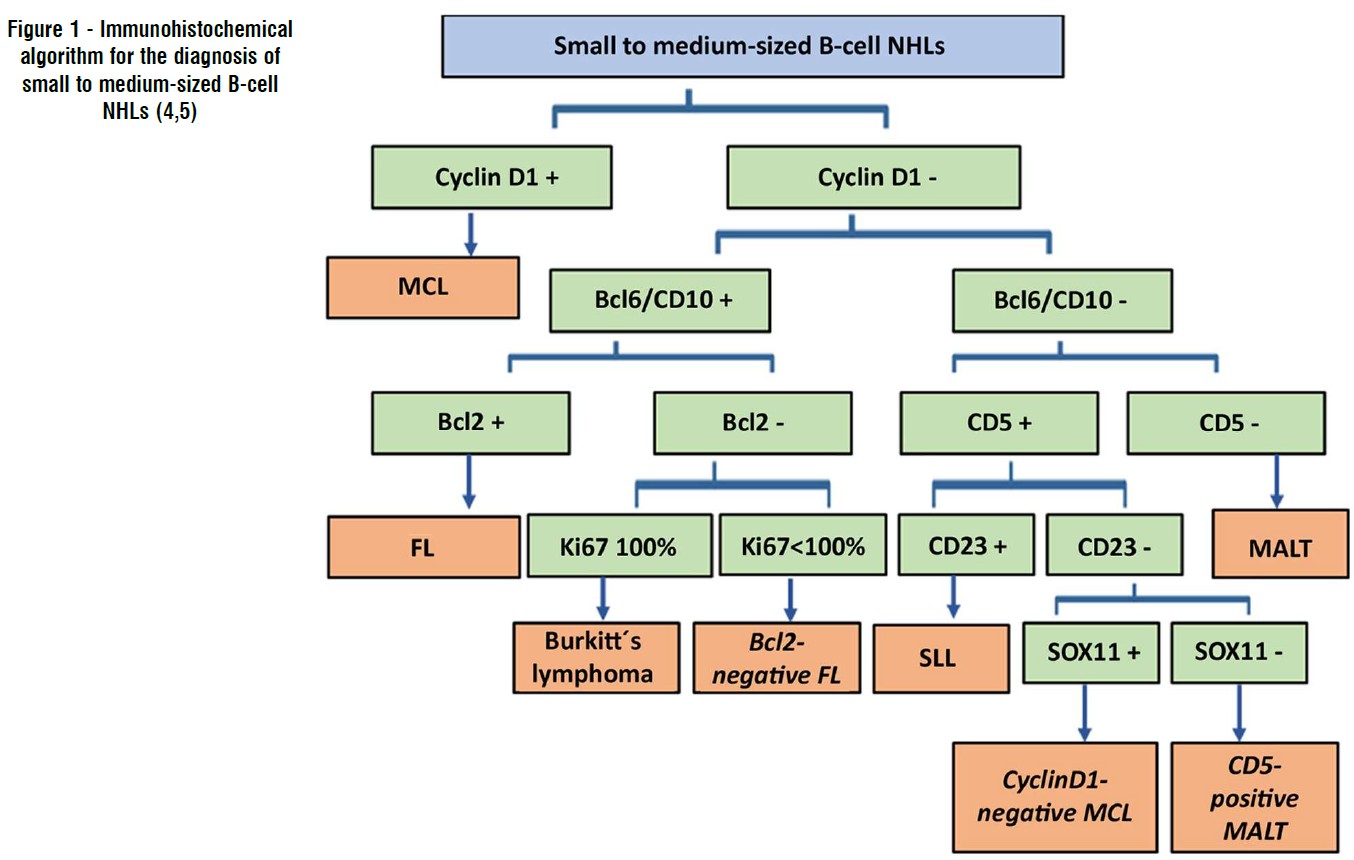
Figure 2 - Approach to diagnosis Diffuse large B-cell lymphoma subtype (4,5)
Statistical Analysis
Statistical calculations and tests were performed using the statistical software RStudio 2024 (R 4.2.1) (5). Categorical and continuous variables were analyzed using descriptive statistics. A significance threshold of 5% was used for all analyses.
Results
Patient Demographics and Clinical Details
The study sample had a mean age of 56.1 ± 15.1 years, ranging from 11 to 99 years, and did not follow a normal distribution (p<0.05) (fig. 3).
Figure 3 - Graph of age distribution among study participants
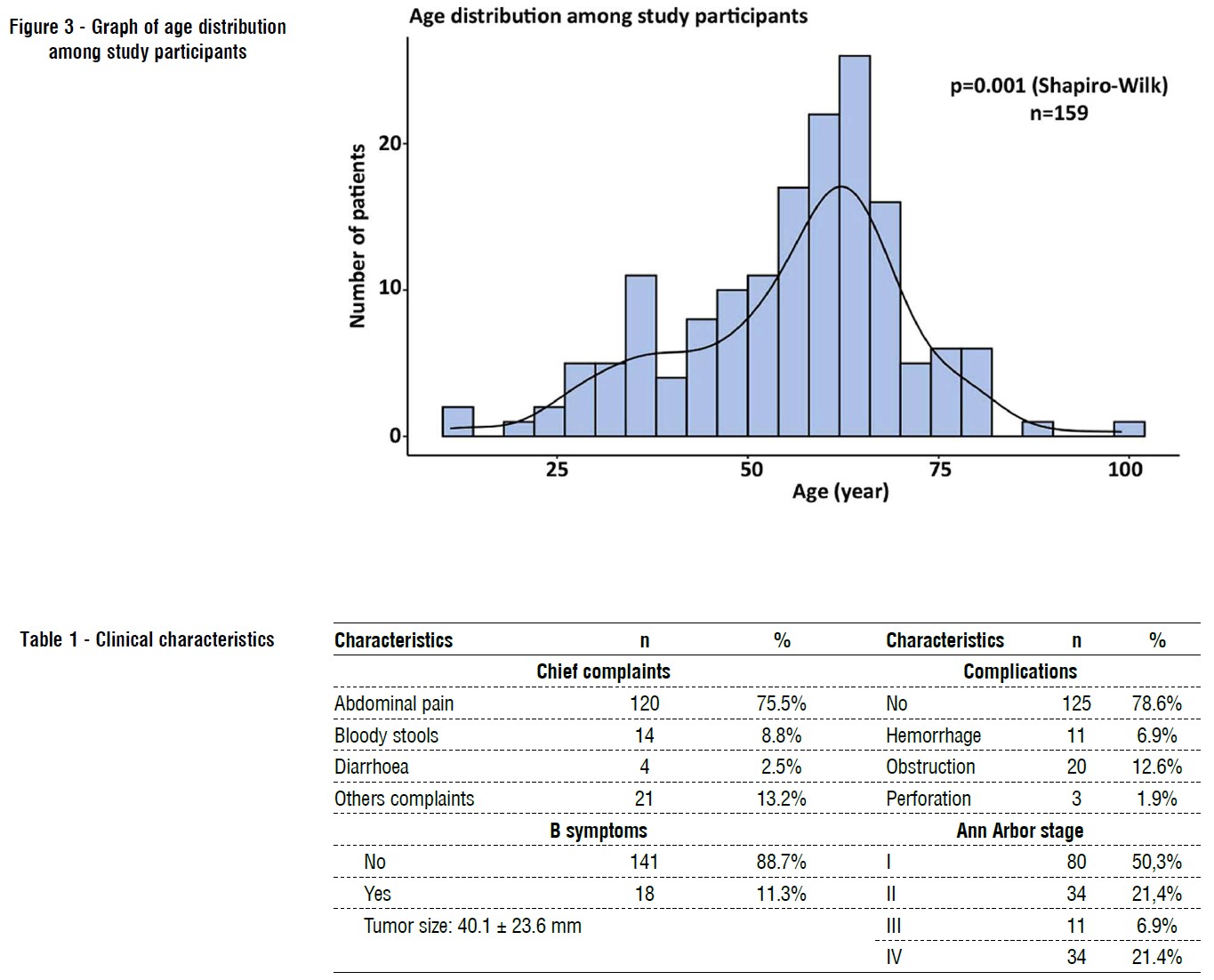
Males were predominant in the study sample compared to females (59.7% males and 40.3% females). The differences were statistically significant (p<0.05). The male to female ratio was 3:2.
Most patients sought medical consultation or were hospitalized because of abdominal pain (75.5%), whereas a smaller proportion presented with bloody stools, diarrhea, obstruction, or other symptoms.
B-symptoms were observed in 11.3% of the total patient population. Complications in patients with gastrointestinal non-Hodgkin lymphoma included gastrointestinal obstruction (12.6%), gastrointestinal bleeding (6.9%), and perforation of the hollow organs (1.9%). The average lesion size was approximately 40.1±23.6 mm. Regarding disease staging, approximately half of the cases were stage I; stages II and IV accounted for 21.4%, while stage III was the least common, comprising only 6.9% (table 1).
Histological Classification and Immunophenotype
The most common type was diffuse large B-cell lymphoma (DLBCL), which was observed in 99 patients. According to Hans Christine P’s algorithm, DLBCL is divided into two subtypes: activated B-cell-like (ABC) (56%) and germinal center B-cell-like (GCB) (44%). The ABC subtype was 100% positive for CD20, 71% positive for CD79a, 85% positive for Mum1, and completely negative for CD3, CD23, and Cyclin D1. The GCB subtype was also 100% positive for CD20, with 64% positive for CD79a, and completely negative for CD3, CD23, and Cyclin D1. The second most common type was mucosa-associated lymphoid tissue (MALT) lymphoma (33 cases). This type showed 100% positivity for CD20, 76% positivity for CD79a, 21% positivity for CD5, 32% positivity for Bcl2, and 20% positivity for Mum1. The cells were almost completely negative for CD10, SOX11, and Cyclin D1 (3% positive staining). Mantle cell lymphoma (MCL) had 15 cases, showing 100% positivity for CD20 and Cyclin D1, 80% positivity for SOX11, 47% positivity for CD5 and Bcl2, and complete negativity for CD3, CD10, and CD23 (table 2).
Table 2 - Immunophenotypes of gastrointestinal lymphomas
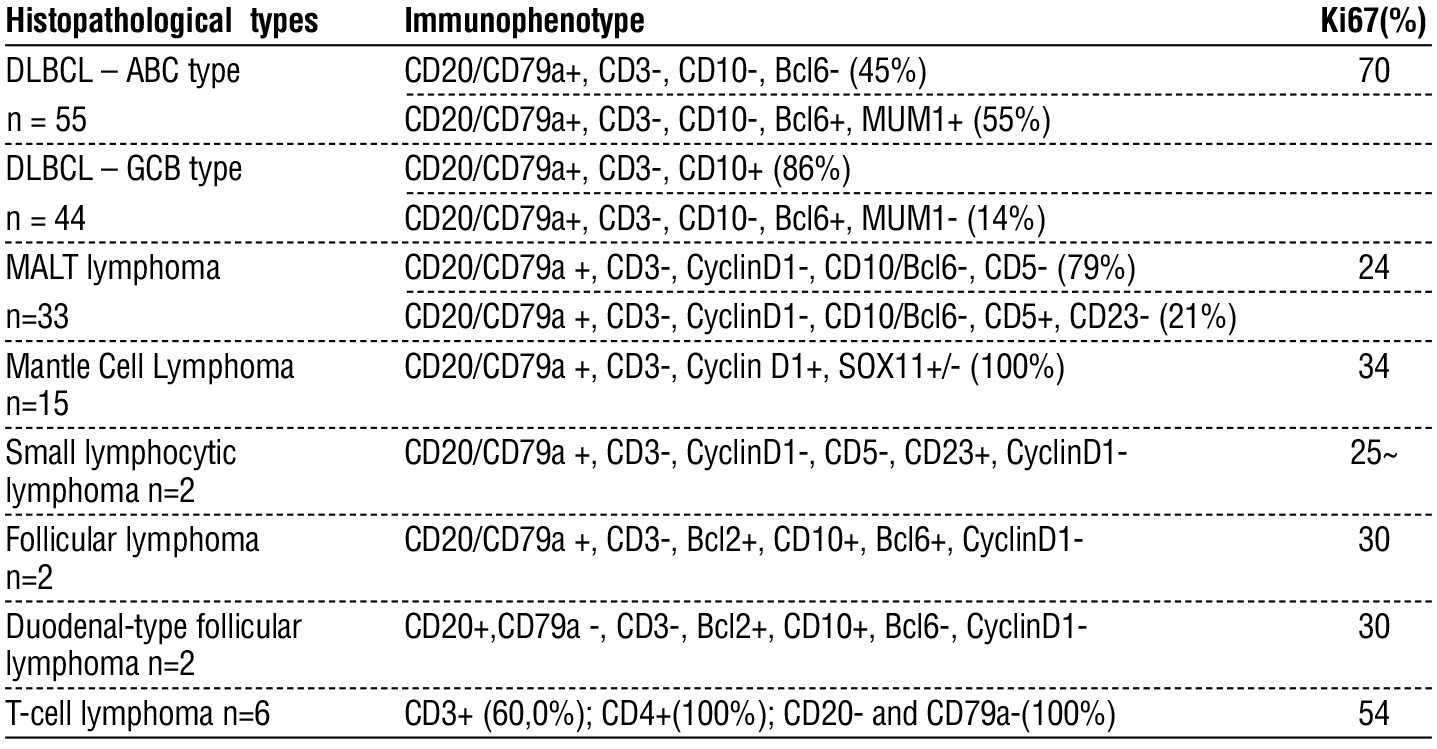
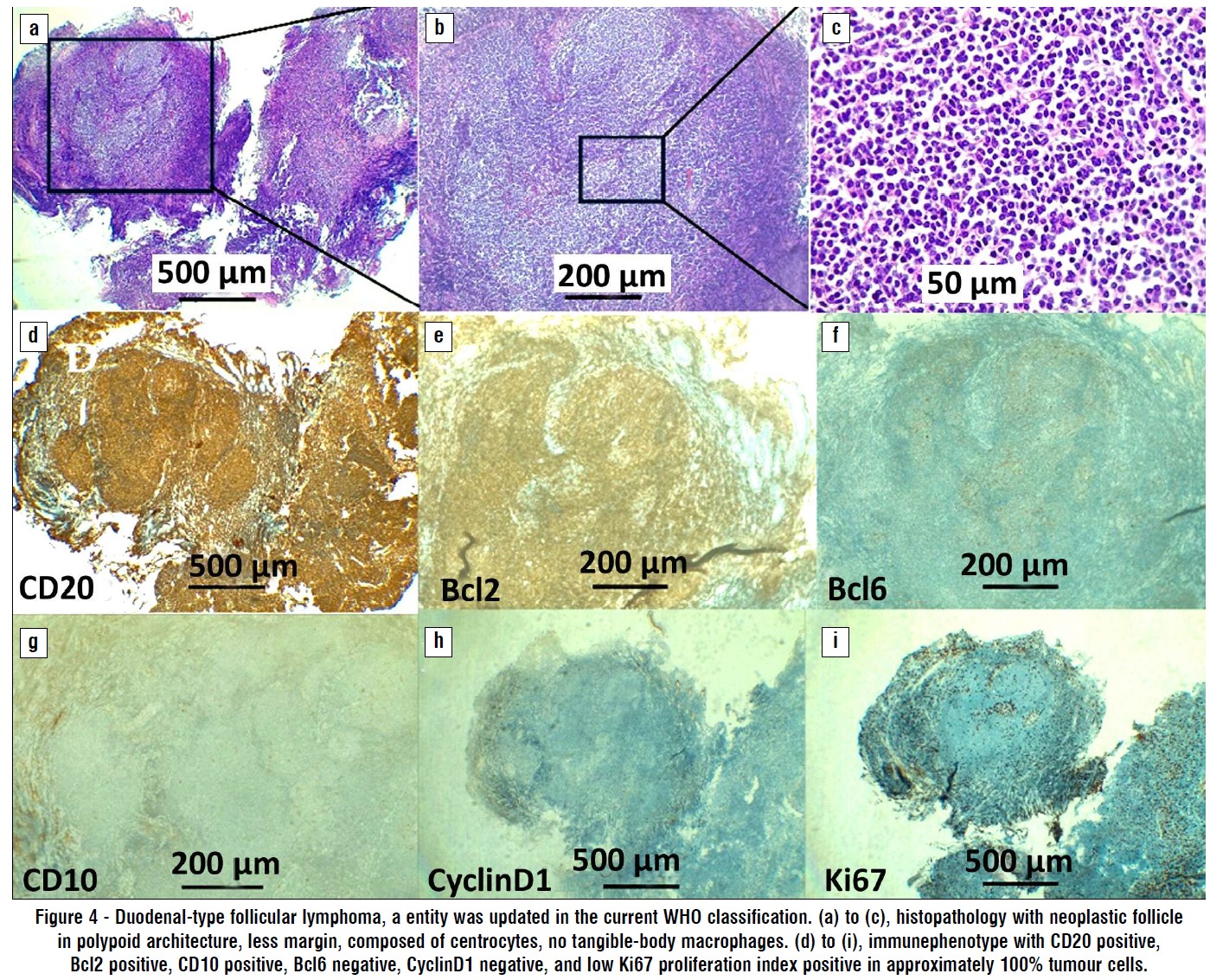
Six cases of T-cell lymphoma were observed, with four demonstrating CD3 positivity. CD3-negative cases exhibited CD4 positivity, and CD8 negativity was observed in 2 of these cases. For follicular lymphoma (FL) and duodenal-type follicular lymphoma (D-FL), each represented by two cases, all were uniformly positive for CD20, CD10, Bcl6, and Bcl2 (fig. 4). Additionally, two cases of small lymphocytic lymphoma (SLL) showed complete positivity for CD20, CD79a, CD23, and CD5, and one case was positive for Bcl2 and Bcl6. All the other assessed markers returned negative results. The Ki67 proliferation index was the highest in DLBCL (70%), followed by TCL (54%); other types range from 24-34%.
Figure 4 - Duodenal-type follicular lymphoma, a entity was updated in the current WHO classification. (a) to (c), histopathology with neoplastic follicle in polypoid architecture, less margin, composed of centrocytes, no tangible-body macrophages. (d) to (i), immunephenotype with CD20 positive, Bcl2 positive, CD10 positive, Bcl6 negative, CyclinD1 negative, and low Ki67 proliferation index positive in approximately 100% tumour cells.
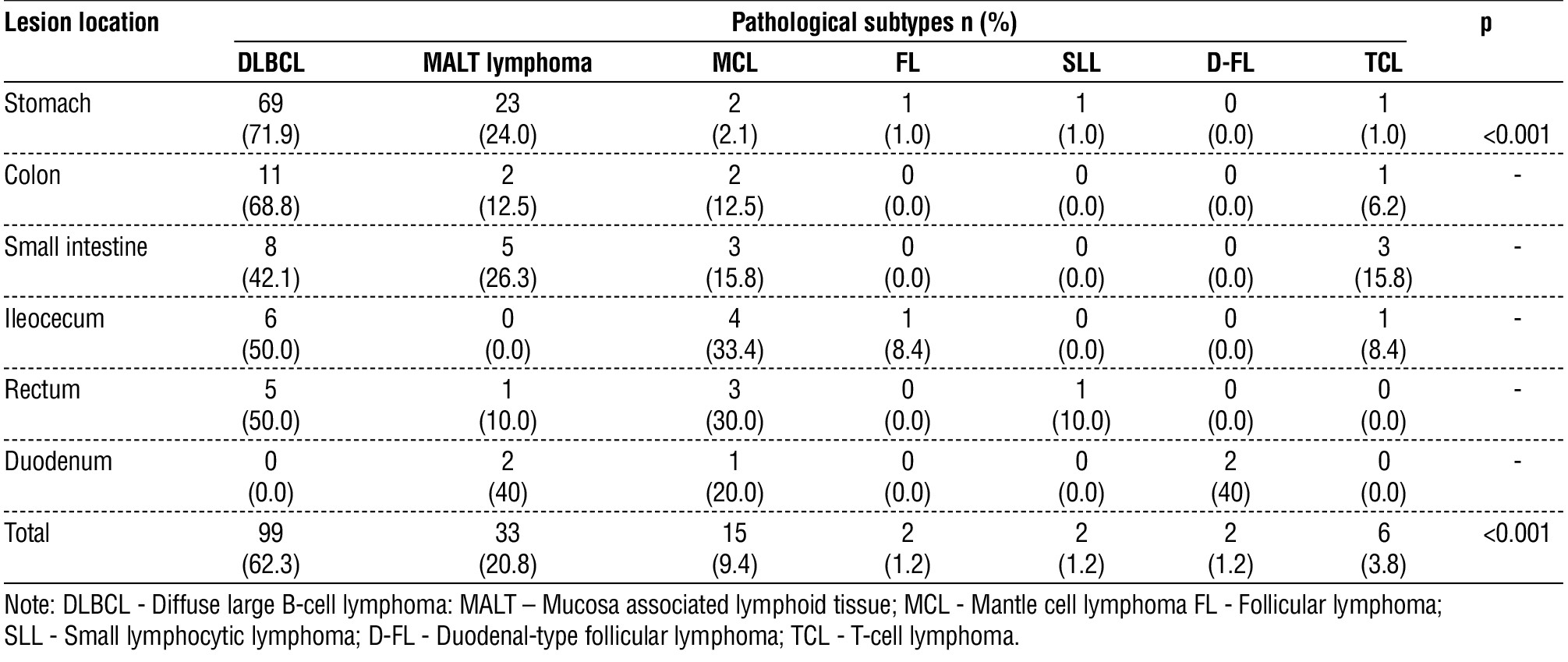
The most frequent anatomical site for DLBCL (69 of 99 cases) and MALT lymphoma (23 of 33 cases) was the stomach. The MCL was observed across nearly all the studied anatomical sites, displaying a broadly distributed pattern, but predominantly appearing in the large intestine. The FL, D-FL, and SLL were limited to the stomach and intestine. TCL was detected in one case in the stomach and in five cases in the intestine. A statistically significant correlation was observed between the lesion location and immunophenotype (p < 0.001), and a statistically significant difference in immunophenotype proportions was found in the stomach (p < 0.001). Due to the limited number of cases in other locations, statistical analysis was not feasible (table 3).
Table 3 - Relationship between the pathological types and tumor locations
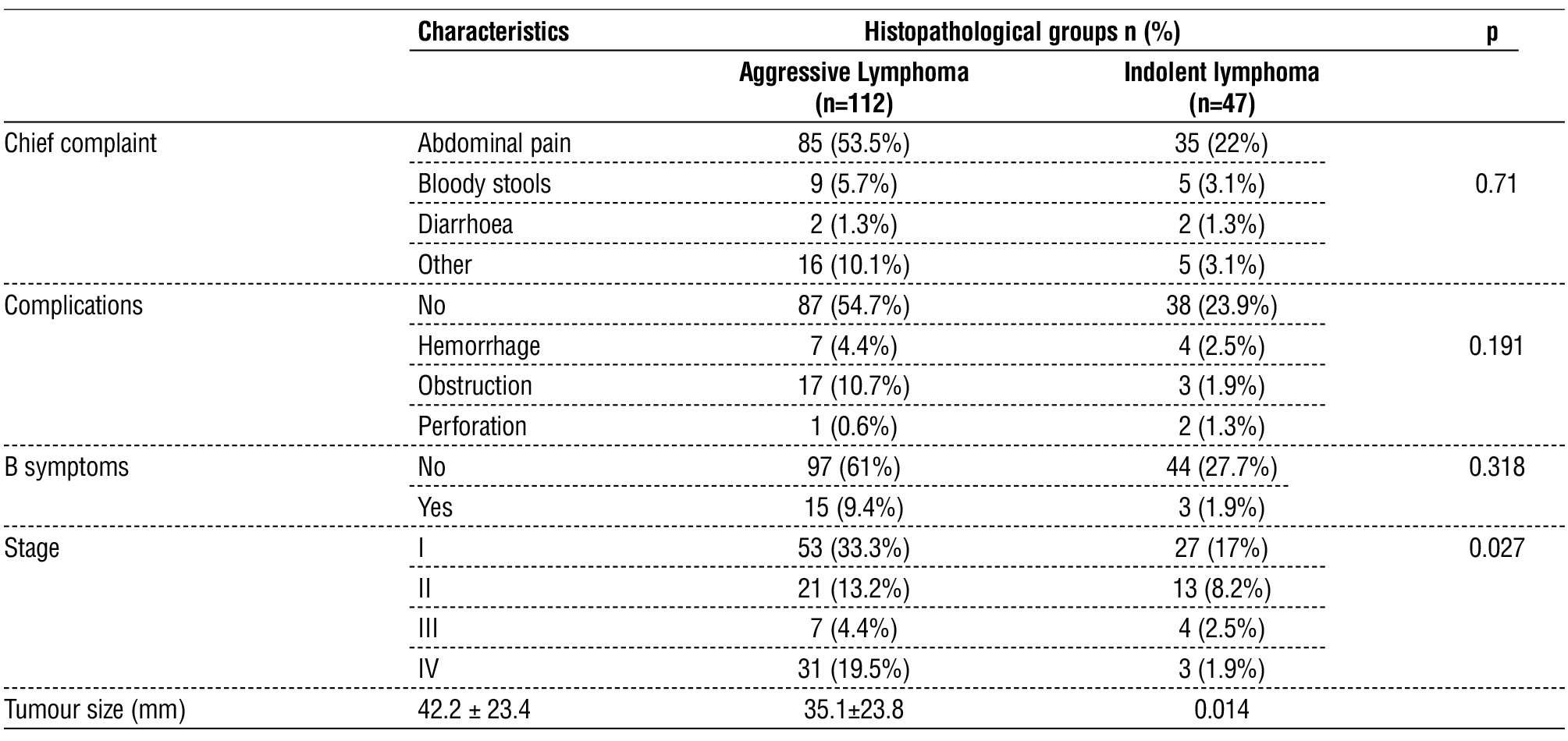
Association between Clinical Characteristics and Histopathological Types (table 4)
Aggressive lymphomas include diffuse large B-cell lymphoma, mantle cell lymphoma, and T-cell lymphoma. Indolent lymphomas include MALT lymphoma, follicular lymphoma, duodenal-type follicular lymphoma, and small lymphocytic lymphoma. There was no association of indolent lymphomas and aggressive lymphomas immunophenotypes in chief complaint, complications, or syndrome B. Differences in disease stage and tumor size between two immunophenotypes were statistically significant. Aggressive lymphomas were detected more frequently at a later stage, and the tumor size was larger than that of indolent lymphomas.
Table 4 - Relationship between clinical characteristics and histopathological groups.
Discussion
Gastrointestinal lymphoma is common in patients aged ≥ 50 years depending on the specific immuno-phenotype. Burkitt's lymphoma is common in children aged 4-7 years. Our study found that the median age was 56.1 ± 15.6 years, ranging from 11 to 99 years. This age also corresponds to the epidemiology of the disease reported in Haematolymphoid tumors of the digestive system in the WHO (2019) Classification of Digestive system tumors (7). Patients with gastrointestinal lymphoma often present with non-specific symptoms, such as common gastrointestinal disorders, abdominal pain or epigastric discomfort, anorexia, weight loss, nausea and/or vomiting, occult gastro-intestinal bleeding, and premature satiety. Systemic B symptoms (fever, weight loss, night sweats) are rarer (8). Our findings are consistent with other studies of non-specific symptoms, the most common of which is abdominal pain (9,10). In our study, most of them were detected in the localized stage, with 71.7% in stages I and II, and 28.3% in stages III and IV. Complication and B symptoms rates were 21.4% and 11.3%, respectively. Other studies have reported rates of B symptoms similar to those in this study (11,12). Wei Wang's study had 27.88% of cases in the early stage (I, II) and 72.12% in the late stage (stage III/IV). In this study, the complication rate was 44.23% with common complications being gastrointestinal bleeding (31.73%), perforation (6.73%), and intestinal obstruction (5.77%), respectively (13). In our study, the complication rate was 21.4%, with the most common complications being obstruction (12.6%), bleeding (6.9%), and perforation (1.9%). In terms of stage and complication rates in our study, it is very similar to Murat Erkut's study with the rate of stages I and II was 75%, and the complication rate was 21% (14). Several reports indicated that the presence of B symptoms and complications does not affect overall survival. A late Ann Arbor stage and aggressive lymphoma type were independent poor prognostic factors for overall survival (15-17). Thus, detecting the disease at an early stage can help significantly reduce the complications of the disease. In the present study, of the 159 cases, 99 were diffuse large B-cell lymphomas, 33 were mucosa-associated lymphoid tissue, 15 were mantle cell lymphoma, 6 were T-cell lymphomas, and other types were scant. Most gastrointestinal lymphomas are B-cell type. The absence of Burkitt lymphoma in the present series was due to the rarity of this lymphoma and the small number of cases studied. DLBCL may be de novo or transformed from MALT lymphoma and often shows significant heterogeneity. Therefore, individualized evaluation of prognostic factors for patients is very meaningful in treatment (18). The frequency of the ABC subtype was higher than that of the GCB subtype, which was similar with other studies (19,20). According Zhang et al study, patients of GCB subtype lived longer than those of ABC subtype (21). Fifteen cases of mantle cell lymphoma had 15 cases, and the tumor cells were completely positive for CD20 and Cyclin D1, 50% for CD5 and Bcl2, and completely negative for CD3, CD10, and CD23. According to literature and previous studies, on immunohistochemistry, mantle cell lymphoma (MCL) is characterized by proliferative lymphocytes that are positive for CD20, CD5 and Cyclin D1. Some samples were negative for Cyclin D1. Tumor cells are usually negative for CD10/Bcl6 and CD23 (7,22). An important differential diagnosis is between mantle cell lymphoma and small lymphocytic lymphoma as both exhibit CD5 markers. With the Cyclin D1 and CD23 markers, the differentiation of mantle cell lymphoma from small cell type lymphoma shows high efficiency. According to a study by Asaad (2005), CD23 is expressed in 64% of small lymphocyte lymphoma cases and only 4% of mantle cell lymphoma cases. Meanwhile, Cyclin D1 was positive in 72% of cases of Mantle cell lymphoma and 4% of cases of Small cell lymphoma (23). Recently, the Ki67 proliferative index has important implications in the diagnosis, prognosis and prediction of the treatment response of gastrointestinal lymphoma (24). The Ki67 proliferation index was found to be 54% in the TCL group. It was much higher in the DLBCL (70%) group than in the other immunohistophenotypes. These findings may be due to DLBCL and TCL being aggressive lymphomas. It is similar to reports by Soumia Zeggai or Lovely George et al (25,26). In our study, the most common locations of the DLBCL (70%) and MALT (68.8%) phenotypes were observed in the stomach. Murat Erkut's study found no histological differences between gastric and intestinal lymphoma (14). There was a statistically significant proportional difference between gastric immunophenotypes (p<0.001). DLBCL accounted for 70.7% compared to 29.7% of all remaining cases. This distribution was similar to the research of T. Kanno (27). In each type of gastro-intestinal lymphoma, there are no typical symptoms, some only manifest in the late stages, such as Mantle cell lymphoma [28]. In our study, abdominal pain was the most common and found in all immunophenotypes (29). Bleeding complications and gastrointestinal obstruction were not recorded in 3 indolent lymphomas phenotypes: FL, D-FL and SLL. However, there was no significant difference in the association of indolent lymphomas and aggressive lymphomas with the occurrence of complications or as symptom B. Our study also did not record symptom B in T-cell lymphoma, this differs from the study by Murat Erkut et al. that the B symptom in T lymphoma is significantly higher than in gastric and intestinal B lymphoma (14). Diagnosis of primary gastrointestinal T-cell neoplasms are still challenging for pathologist because of its rarity, especially with small biopsy material (30). There was no statistically significant association between the two groups of aggressive and indolent lymphomas, except for the disease stage (p<0.05). Aggressive lymphomas are more commonly detected in stages I and IV. Indolent lymphomas, on the contrary, stage IV is very rare (3/47 cases), and stage I is the most common. The size of lesions in the aggressive lymphoma group was also larger than that in the indolent lymphoma group (42.2 vs. 35.1 mm), a difference of approximately 7.1 mm, and this difference was statistically significant (p<0.05).
Conclusion
In conclusion, this study emphasizes that diffuse large B-cell lymphoma (DLBCL) is the most common lymphoma subtype, with a notable occurrence of both activated B-cell-like (ABC) and germinal center B-cell-like (GCB) subtypes. The findings demonstrate a predominance of aggressive lymphomas in both early (stage I) and advanced (stage IV) stages, while indolent lymphomas are predominantly present at stage I, suggesting a distinct staging pattern among lymphoma types. The lesion size was notably larger in aggressive lymphomas than in indolent lymphomas, aligning with the more severe clinical presentations in advanced cases. These data underscore the importance of subtype-specific diagnostic and staging criteria, which may enhance early identification and allow for tailored therapeutic interventions.
Author’s Contributions
Conceptualization, T.D.C., Q.T.T and T.N.T.; metho-dology, Q.T.T., T.N.D., and B.S.N.T.; software, Q.T.N., N.P.T.T., and V.L.; validation, Q.T.T, B.S.N.T., T.N.T., and T.D.C; formal analysis, Q.T.T., T.N.D., and B.S.N.T.; investigation, T.N.D., B.S.N.T., Q.T.N., N.P.T.T., and V.L; data curation, B.S.N.T., Q.T.N., N.P.T.T., Q.T.T and V.L.; writing-original draft preparation, B.S.N.T., T.N.T., and T.D.C.; writing-review and editing, Q.T.T and T.D.C.; supervision, T.D.C.; project administration, T.D.C.; funding acquisition, T.D.C., Q.T.T. and T.N.T. All authors have read and agreed to the published version of the manuscript.
Acknowledgments
The authors acknowledge Hue University's support through the Core Research Program (Research Group on Regenerative Medicine, NCTB.DHH.2024.02).
Conflicts of Interest
The authors declare no conflict of interest.
Funding
This research was funded by the Research Projects in Science and Technology of the Vietnamese Ministry of Education and Training (B2021-DHH-20).
References
1. Ghimire P. Primary gastrointestinal lymphoma. World J Gastroenterol. 2011;17(6):697.
2. Bautista-Quach MA, Ake CD, Chen M, Wang J. Gastrointestinal
lymphomas: Morphology, immunophenotype and molecular
features. Gastrointest Oncol. 2012;3(3):209-225.
3. Iwamuro M, Tanaka T, Okada H. Review of lymphoma in the
duodenum: An update of diagnosis and management. World J Gastroenterol. 2023;29(12):1852-1862.
4. Pizzi M, Sabattini E, Parente P, Bellan A, Doglioni C, Lazzi S. Gastrointestinal lymphoproliferative lesions: a practical diagnostic approach. Pathologica. 2020;112(3):227-247.
5. Chan JKC, Fukayama M. Haematolymphoid tumours of the digestive system. In: Digestive System Tumours. 5th ed. WHO Blue Books; 2019.
6. Erkut M, Erkut N, Bektas O, Fidan S, Cosar AM, Sonmez M. Effect of Clinical, Endoscopic, Radiological Findings, and Complications on Survival in Patients with Primary Gastrointestinal Lymphoma. Turk J Gastroenterol. 2022;33(11):909-917.
7. Cha RR, Baek DH, Lee GW, Park SJ, Lee JH, Park JH, et al. Clinical Features and Prognosis of Patients with Primary Intestinal B-cell Lymphoma Treated with Chemotherapy with or without Surgery. Korean J Gastroenterol. 2021;78(6):320-327.
8. World Health Organization. Digestive System Tumours. (5th, ed.). WHO; 2019.
9. Aslan A, Ak?n S, Babacan T, Özdemir E. Gastrointestinal Lymphomas. In: Textbook of Gastrointestinal Oncology. Springer International Publishing; 2019. p. 329-349.
10. Khuroo MS, Khwaja SF, Rather A, Hassan Z, Reshi R, Khuroo NS. Clinicopathological profile of gastrointestinal lymphomas in Kashmir. Indian J Med Paediatr Oncol. 2016;37(4):251-255.
11. Shi Z, Ding H, Shen QW, Lu XG, Chen JY, Chen X, et al. The clinical manifestation, survival outcome and predictive prognostic factors of 137 patients with primary gastrointestinal lymphoma (PGIL). Medicine (Baltimore). 2018;97(1):e9583.
12. Koch P, del Valle F, Berdel WE, N A Willich, B Reers, W Hiddemann, et al. Primary Gastrointestinal Non-Hodgkin’s Lymphoma: I. Anatomic and Histologic Distribution, Clinical Features, and Survival Data of 371 Patients Registered in the German Multicenter Study GIT NHL 01/9. J Clin Oncol. 2001;19(18):3861-73.
13. Nakamura S, Matsumoto T, Iida M, Yao T, Tsuneyoshi M. Primary gastrointestinal lymphoma in Japan. Cancer. 2003;97(10):
2462-2473.
14. Wang W, Lin P, Yao H, Jia X, Sun J. Clinical analysis of Primary Gastrointestinal Non-Hodgkin’s Lymphoma. Pak J Med Sci. 2017;33(6).
15. Amer MH, el-Akkad S. Gastrointestinal lymphoma in adults: clinical features and management of 300 cases. Gastroenterology. 1994;106(4):846-58.
16. Tran T, Vu TH, Vo HQ, Thi Nguyen H, Van Nguyen H. Primary
gastrointestinal non-Hodgkin lymphoma: a retrospective study in Vietnam. Ann Med Surg (Lond). 2023;85(6):2390-2394.
17. Gao F, Wang ZF, Tian L, Dong F, Wang J, Jing HM, et al. A Prognostic Model of Gastrointestinal Diffuse Large B Cell Lymphoma. Med Sci Monit. 2021;27:e929898.
18. Ding W, Zhao S, Wang J, Yang Q, Sun H, Yan J, et al. Gastrointestinal Lymphoma in Southwest China: Subtype Distribution of 1,010 Cases Using the WHO (2008) Classification in a Single Institution. Acta Haematol. 2016;135(1):21-28.
19. Hans CP. Confirmation of the molecular classification of diffuse large B-cell lymphoma by immunohistochemistry using a tissue micro-array. Blood. 2004;103(1):275-282.
20. Zhang Z, Shen Y, Shen D, Ni X. Immunophenotype classification and therapeutic outcomes of Chinese primary gastrointestinal diffuse large B-cell lymphoma. BMC Gastroenterol. 2012;12(1):77.
21. Cree IA, Centre international de recherche sur le cancer, Organisation mondiale de la sante?. WHO Classification of Tumours. Digestive System Tumours: WHO Classification of Tumours, Volume 1. 5th ed. International Agency for Research on Cancer Lyon; 2019.
22. Skinnider BF. Lymphoproliferative Disorders of the Gastrointestinal Tract. Arch Pathol Lab Med. 2018;142(1):44-52.
23. Asaad NY, Abd El-Wahed MM, Dawoud MM. Diagnosis and prognosis of B-cell chronic lymphocytic leukemia/small lymphocytic
lymphoma (B-CLL/SLL) and mantle cell lymphoma (MCL). J Egypt Natl Canc Inst. 2005;17(4):279-290.
24. Mao X, Li Y, Liu S, He C, Yi S, Kuang D, et al. Multicolor flow
cytometric assessment of Ki67 expression and its diagnostic value in mature B-cell neoplasms. Front Oncol. 2023;13:1108837.
25. Lovely G, Prithal G, Philipose TR, Sandhya I, Rao PS, Shetty P. Non-Hodgkin lymphomas of the gastrointestinal tract: an institutional experience. Int J Acad Med Pharm. 2022;4(5):384-386.
26. Zeggai S, Harir N, Tou A, Sellam F, Mrabent MN, Salah R. Immuno-histochemistry and scoring of Ki-67 proliferative index and p53 expression in gastric B cell lymphoma from Northern African population: a pilot study. J Gastrointest Oncol. 2016;7(3):462-468.
27. Kanno T, Katano T, Shimura T, Nishigaki R, Kojima Y, Sasaki M, et al. Characteristic endoscopic findings of gastrointestinal malignant lymphomas other than mucosa-associated lymphoid tissue
lymphoma. Acta Gastroenterol Belg. 2022;85(3):477-483.
28. Greenson JK, et al. Neoplastic Adenoma, Stomach. In: Diagnostic Pathology Gastrointestinal. LWW; 2009. p. 233-286.
29. Tian FY, Wang JX, Huang G, An W, Ai LS, Wang S, et al. Clinical and endoscopic features of primary small bowel lymphoma: a single-center experience from mainland China. Front Oncol. 2023; 13:1142133.
30. Nishimura MF, Nishimura Y, Nishikori A, Yoshino T, Sato Y. Primary Gastrointestinal T-Cell Lymphoma and Indolent Lymphoproliferative Disorders: Practical Diagnostic and Treatment Approaches. Cancers (Basel). 2021;13(22):5774.
Full Text Sources:
Abstract:
Views: 2258

For Authors
Journal Subscriptions

Dec 2025
Supplements
Instructions for authors
Online submission
Contact
e-ISSN: 2601 - 1700 (online)
ISSN-L: 2559 - 723X
Journal Abbreviation: Surg. Gastroenterol. Oncol.
Surgery, Gastroenterology and Oncology (SGO) is indexed in:
- SCOPUS
- EBSCO
- DOI/Crossref
- Google Scholar
- SCImago
- Harvard Library
- Open Academic Journals Index (OAJI)
Surgery, Gastroenterology and Oncology (SGO) is an open-access, peer-reviewed online journal published by Celsius Publishing House. The journal allows readers to read, download, copy, distribute, print, search, or link to the full text of its articles.
Time to first editorial decision: 25 days
Rejection rate: 61%
CiteScore: 0.2

Meetings and Courses in 2025
Meetings and Courses in 2024
Meetings and Courses in 2023
Meetings and Courses in 2022
Meetings and Courses in 2021
Meetings and Courses in 2020
Meetings and Courses in 2019
Verona expert meeting 2019

Surgery, Gastroenterology and Oncology applies the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits readers to copy and redistribute the material in any medium or format, remix, adapt, build upon the published works non-commercially, and license the derivative works on different terms, provided the original material is properly cited and the use is non-commercial. Please see: https://creativecommons.org/licenses/by-nc/4.0/
Publisher’s Note:
The opinions, statements, and data contained in article are solely those of the authors and not of Surgery, Gastroenterology and Oncology journal or the editors. Publisher and the editors disclaim responsibility for any damage resulting from any ideas, instructions, methods, or products referred to in the content.
 IASGO Society News
IASGO Society News