Surgery, Gastroenterology and Oncology

|
|
Introduction: Adjustable gastric banding (AGB) has undergone a significant decline in use over the past decade due to high rates of long-term complications, frequent reoperations, and the superior efficacy of alternative bariatric procedures. Globally, its utilization between 2014 and 2018 averaged 5%. Risk factors contributing to complications include hiatal hernias, severe gastroesophageal reflux, inadequate postoperative follow-up, and suboptimal surgical technique.
Case presentation: We present a case of a 46-year-old woman with intragastric erosion of an adjustable gastric band placed over ten years prior, who presented with abdominal pain and distension. Imaging demonstrated near-complete band migration into the gastric lumen. A combined laparoscopic and endoscopic approach facilitated successful band removal, including sectioning with a lithotriptor and extraction via endoscopy, alongside laparoscopic dissection and repair. Clinical discussion: endoscopic removal of eroded bands has emerged as the preferred first-line approach, yielding technical and clinical success rates exceeding 85%, with low complication rates. Laparoscopic intervention remains reserved for cases unsuitable for endoscopy, such as those involving dense adhesions or fistulae.
Conclusion: Combined endoscopic-laparoscopic management is safe and effective in selected cases, and long-term follow-up is essential for timely detection and management of complications in patients with gastric bands.
INTRODUCTION
Over the past decade, adjustable gastric banding (AGB) has witnessed a marked decline in utilization both within the United States and globally. Data from the American Society for Metabolic and Bariatric Surgery (ASMBS) indicate that in 2018, AGB accounted for a mere 1.1% of bariatric procedures in the US, a significant reduction from 35.4% in 2011 (1). Worldwide, between 2014 and 2018, gastric banding comprised only approximately 5% of bariatric interventions. This trend primarily reflects the high incidence of long-term complications, the frequent necessity for reoperations, and the superior efficacy demonstrated by alternative procedures such as sleeve gastrectomy and Roux-en-Y gastric bypass (2). Leading organizations, including the ASMBS, acknowledge that although adjustable bands were once extensively utilized, they presently constitute only a minimal proportion of bariatric surgeries (3).
Longitudinal studies reveal that up to 40% of patients experience complications within 10 years post-AGB, while reoperation rates approach 20%, further curtailing their adoption. Currently, gastric banding represents less than 2% of bariatric surgeries in the US (4).
Risk factors predisposing to complications include the presence of severe gastroesophageal reflux disease (GERD), large hiatal hernias, esophageal motility disorders, inadequate postoperative follow-up, and suboptimal surgical technique. Careful patient selection combined with comprehensive multidisciplinary postoperative management remains pivotal in mitigating risks (5).
The standard technique entails laparoscopic placement of an adjustable silicone band encircling the proximal stomach, thereby creating a small gastric pouch. Band tightness is modulated via saline injections into a subcutaneous access port to maintain a balance between effective restriction and avoidance of obstruction or regurgitation. Management protocols incorporate nutritional counseling, periodic band adjustments, and close monitoring of associated comorbidities (6).
Common complications encompass band slippage, reservoir pouch dilation, intragastric erosion, gastric obstruction, port-site infections, and mechanical device failures. While initial management is predominantly conservative, surgical intervention may be warranted for band repositioning, revision, or explantation. Laparos-copic approaches are the standard both for primary placement and for addressing complications (4).
Prominent scientific bodies such as the American Gastroenterological Association and ASMBS emphasize stringent patient selection criteria and rigorous longitudinal follow-up to optimize clinical outcomes and minimize adverse events.
This article provides a current overview of the endoscopic and laparoscopic strategies employed in the management of eroded gastric bands, grounded in the most robust evidence available, to facilitate informed clinical decision-making in complex scenarios.
CASE REPORT
We report the case of a 46-year-old female patient with a history of laparoscopically implanted adjustable gastric banding over 10 years prior in a foreign country, without documented postoperative follow-up. She presented to the Emergency Department complaining of a several-day history of right iliac fossa abdominal pain accompanied by abdominal distension and difficulty in passing flatus. The patient was afebrile and denied nausea, vomiting, or alterations in bowel habits. Physical examination revealed a soft, depressible abdomen with focal tenderness localized to the right iliac fossa; no signs of peritoneal irritation were evident.
Laboratory investigations were unremarkable and contrast-enhanced abdominal computed tomography (CT) showed no evidence of acute intraabdominal pathology. The gastric band appeared appropriately positioned without radiological signs of complication. Given the persistence of symptoms and surgical history, diagnostic upper gastrointestinal endoscopy was performed, revealing near-complete intragastric migration of the band, tethered solely by a thin mucosal bridge.
Following informed patient counseling, a planned surgical removal of the device was undertaken via a combined laparoscopic and endoscopic approach in collaboration with the Digestive Endoscopy unit. Exploratory laparoscopy identified an inflammatory phlegmon involving the greater omentum extending into the supramesocolic compartment (fig. 1).
Figure 1 - Gastric inflammatory phlegmon
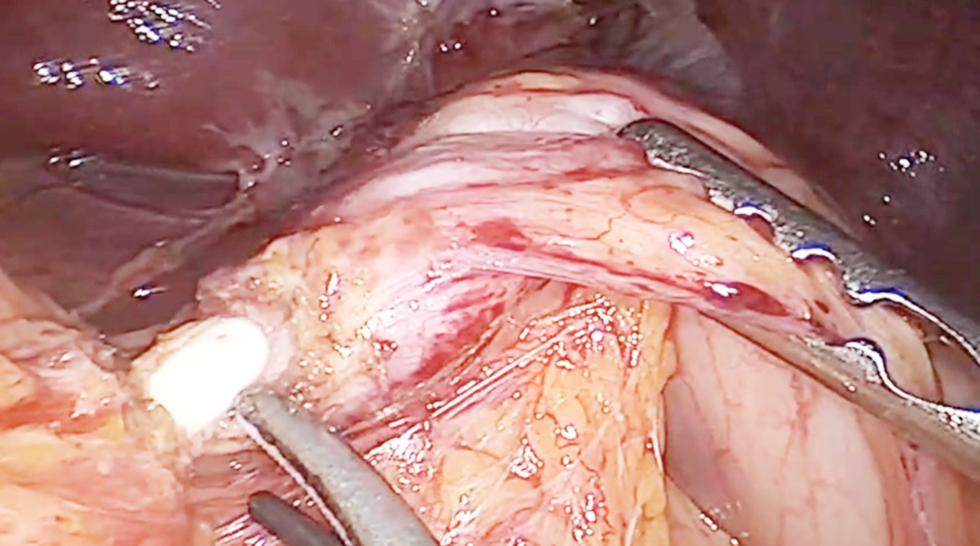
Palpation localized the migrated band within the gastric body, with the connecting catheter penetrating the gastric wall at the lesser curvature. Careful dissection of the prepyloric antrum was performed, and a compression tape was applied proximally to control gastric outflow and prevent contamination during the procedure (fig. 2).
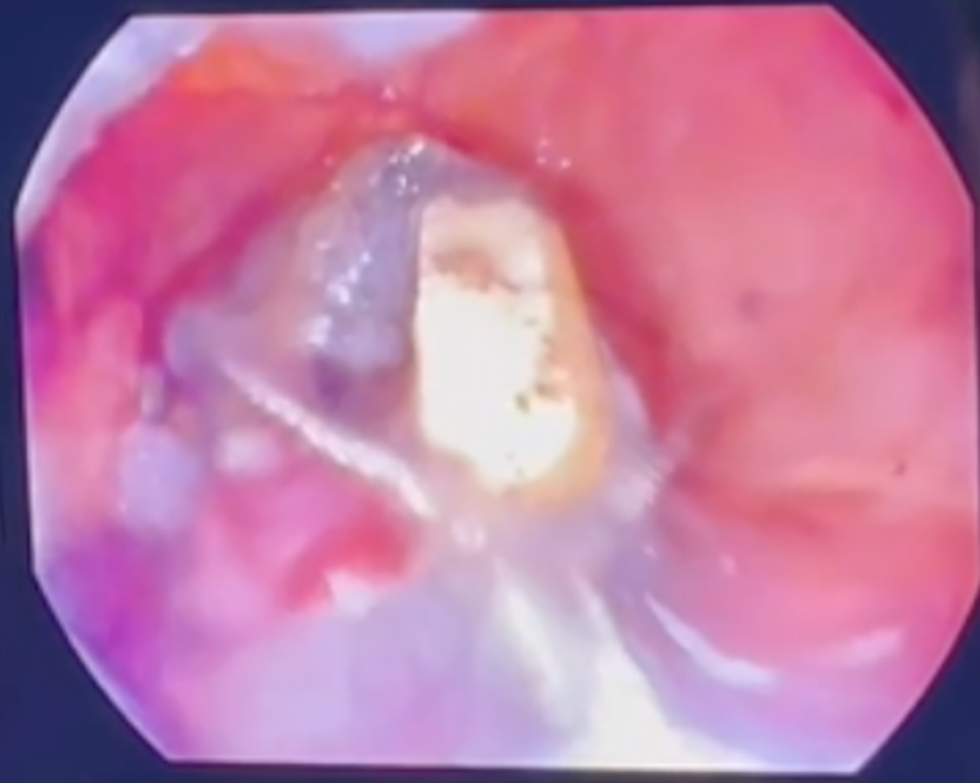
Concomitant intraoperative endoscopy confirmed near-complete migration of the band into the gastric lumen, with only a residual mucosal bridge. Endoscopic sectioning of the band was achieved using a lithotriptor device, followed by uneventful extraction through the endoscope channel (fig. 3 and fig. 4).
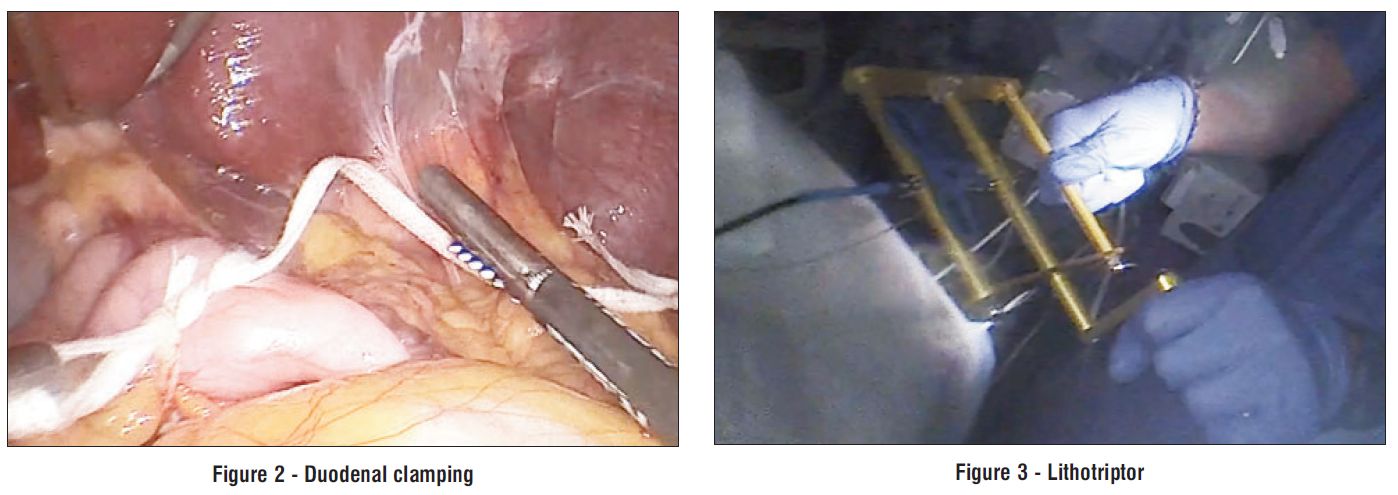
Figure 4 - Endoscopic band removal
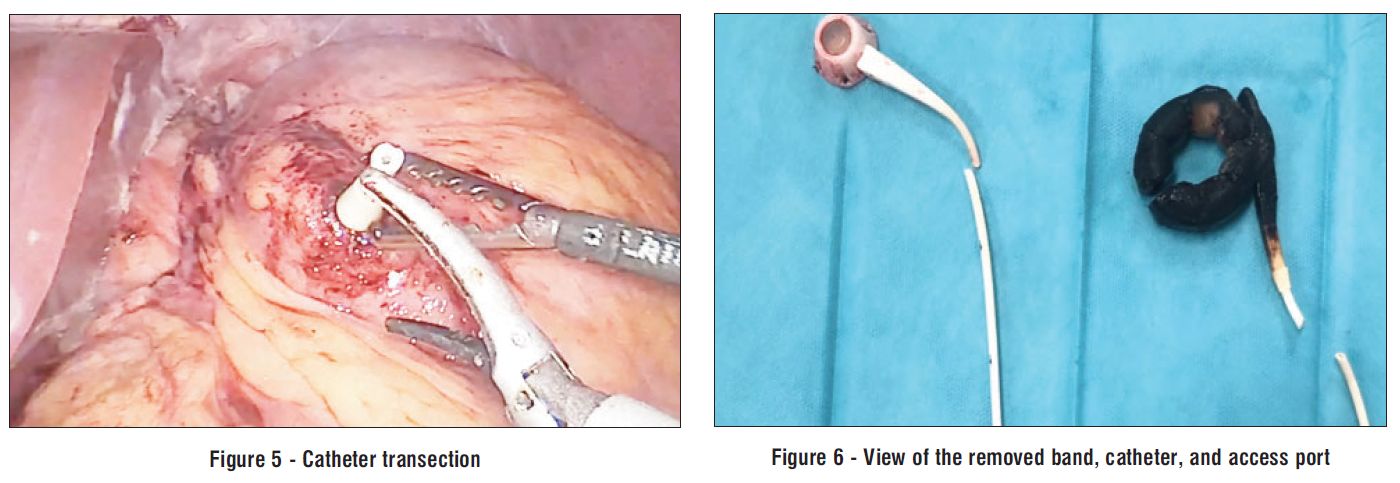
The catheter was transected at its gastric entry point (fig. 5), and the resulting fistulous tract was resected with two linear endoscopic staple loads. Subsequently, both the catheter and the subcutaneous reservoir - located in the left hypochondrium - were completely removed (fig. 6).
Final laparoscopic inspection showed no evidence of leakage or active bleeding, permitting standard
closure. The postoperative course was uneventful. Oral intake was progressively reintroduced from postoperative day two without complications. The patient maintained stable clinical status, with normal bowel function and absence of infectious or local adverse events, and was discharged on postoperative day four in good condition.
DISCUSSION
Intragastric erosion of the gastric band, occurring in up to 3% of patients, necessitates removal to circumvent risks of infection, gastric perforation, or therapeutic failure. The endoscopic approach, currently favored in the majority of cases, is indicated when at least one third of the band has migrated into the gastric lumen. This technique involves endoscopic transection and cautious extraction of the band, aiming to minimize additional tissue injury (8,9).
Endoscopic removal involves cutting the band within the stomach and extracting it carefully, aiming to minimize injury to the surrounding tissues. Reported clinical and technical success rates of endoscopic removal exceed 85%, with major complications occurring infrequently (4-7%). Conversion to laparos-copic or open surgery is required in fewer than 11% of cases, predominantly due to adhesions, incomplete migration, or technical challenges (10-13).
Procedures are performed under general anesthesia or deep sedation within an operating room setting, enabling immediate surgical intervention if complications arise. Recovery is typically rapid, with discharge achievable within 24 to 48 hours (13).
Laparoscopic management is reserved for scenarios in which endoscopic removal is contraindicated or infeasible, such as in the presence of fibrosis, abscess formation, fistulae, or elevated perforation risk. This approach facilitates direct visualization of the peritoneal cavity, gastric wall repair, and 7 management of associated complications. It necessitates meticulous dissection of the band and its adhesions, controlled removal, and gastric closure as indicated. In experienced hands, morbidity is low, with rare need for conversion to open surgery, albeit with potentially prolonged hospitalization (9).
Potential adverse events include gastric perforation, hemorrhage, pneumoperitoneum, and abscess
formation, most of which are amenable to conservative management. Optimal outcomes require diligent patient selection and a multidisciplinary treatment paradigm (14-16).
Current evidence supports the endoscopic route as the first-line intervention for removal of eroded gastric bands, reserving laparoscopic surgery for complex presentations or when endoscopic retrieval is not feasible. Clinical decision-making should be individuali-zed based on the extent of band migration, concurrent complications, and procedural expertise, with paramount emphasis on patient safety and expedited recovery (14-16).
The risk of gastric injury post-removal correlates with the procedural approach and degree of migration. Endoscopic removal carries an approximately 4.1% rate of intraoperative adverse events, with perforations being exceptionally rare when complete band migration has occurred. In this context, the risk of additional injury remains low, as the band is freely mobile within the gastric lumen (8,11,15).
Conversely, laparoscopic intervention involves risks related to adhesiolysis and gastrotomy, with gastric leaks reported in up to 2% of cases, particularly when inflammation is present or gastric repair is necessary. Nonetheless, laparoscopy affords identification and immediate management of such complications (17).
CONCLUSION
In summary, both modalities confer a low risk of gastric injury when the band is fully eroded, with a slightly lower risk associated with endoscopic removal contingent upon appropriate case selection and execution in specialized centers (8,11,17).
Author’s Contributions
Motos Micó JJ: drafting and writing of manuscript and management of the patient. Sánchez López D: management of the patient. Ferrero Celemin E: management of the patient. Sánchez-Cabezudo Noguera F: revising the manuscript and management of the patient. Gil López JM: management of the patient. Antonio Picardo Nieto: management of the patient.
Conflict of Interests
The authors declare no conflicts of interest regarding the preparation or publication of this case report.
Source of Funding
This case report did not receive any form of funding or grant from public, private or non-profit organizations.
Ethical Approval
In accordance with our institution's policy, case reports do not require Institutional Review Board (IRB) approval, provided a written informed consent has been obtained from the patient.
Consent
A written informed consent was obtained from the patient for publication of this case report and accompanying images.
Declaration of Generative AI and AI-assisted Technologies in the writing process
During the preparation of this manuscript, the authors used OpenEvidence, an artificial intelligence based platform, to support the efficient retrieval, synthesis, and organization of up-to-date scientific evidence. After using this tool/service, the author(s) reviewed and edited the content as needed and take(s) full responsibility for the content of the publication.
REFERENCES
1. English WJ, DeMaria EJ, Hutter MM, Kothari SN, Mattar SG, Brethauer SA et al. American Society for Metabolic and Bariatric Surgery 2018 estimate of metabolic and bariatric procedures performed in the United States. Surg Obes Relat Dis. 2020;16(4): 457-463.
2. Lo Menzo E, Szomstein S, Rosenthal R. Update on Treatment of Morbid Obesity with Adjustable Gastric Banding. Surg Colin North Am. 2016;96(4):795-813.
3. Mechanick JI, Apovian C, Brethauer S, Timothy Garvey W, Joffe AM, Kim J,et al. Clinical Practice Guidelines for the Perioperative Nutrition, Metabolic, and Nonsurgical Support of Patients Undergoing Bariatric Procedures - 2019 Update: Cosponsored by American Association of Clinical Endocrinologists/American College of Endocrinology, The Obesity Society, American Society for Metabolic and Bariatric Surgery, Obesity Medicine Association, and American Society of Anesthesiologists. Obesity (Silver Spring). 2020;28(4):O1-O58
4. Ibrahim AM, Thumma JR, Dimick JB. Reoperation and Medicare Expenditures After Laparoscopic Gastric Band Surgery. JAMA Surg. 2017;152(9):835-842.
5. Acosta A, Streett S, Kroh MD, Cheskin LJ, Saunders KH, Kurian M et al. White Paper AGA: POWER - Practice Guide on Obesity and Weight Management, Education, and Resources. Clin Gastroenterol Hepatol. 2017;15(5):631-649.e10.
6. Kodner C, Hartman DR. Complications of adjustable gastric banding surgery for obesity. Am Fam Physician. 2014;89(10):813-8
7. Owers C, Ackroyd R. A study examining the complications associated with gastric banding. Obes Surg. 2013;23(1):56-9
8. Deshmukh A, Desai PM, Ma C, Kushnir V, Eckhouse S, Dimou FM et al. Endoscopic Management of Gastric Band Erosion: a Systematic Review and Meta-Analysis. Obes Surg. 2024;34(2): 494-502
9. Quadri P, Gonzalez-Heredia R, Masrur M, Sanchez-Johnsen L, Elli EF. Management of laparos-copic adjustable gastric band erosion. Surg Endosc. 2017;31(4):1505-1512.
10. Robinson TJ, Soriano C, Larsen M, Mallipeddi MK, Hunter JA, Chang L. Endoscopic removal of eroded laparoscopic adjustable gastric bands: a preferred approach. Surg Obes Relat Dis. 2020; 16(8):1030-1034
11. Manos T, Nedelcu A, Noel P, Zulian V, Danan M, Vilallonga R, Carandina S et al. Endoscopic Gastric Band Removal. J Clin Med. 2023;12(2):617
12. El-Hayek K, Timratana P, Brethauer SA, Chand B. Complete endoscopic/transgastric retrieval of eroded gastric band: description of a novel technique and review of the literature. Surg Endosc. 2013; 27(8):2974-9
13. Neto MP, Ramos AC, Campos JM, Murakami AH, Falcão M, Moura EH et al. Endoscopic removal of eroded adjustable gastric band: lessons learned after 5 years and 78 cases. Surg Obes Relat Dis. 2010;6(4):423-7.
14. Furbetta N, Gragnani F, Cervelli R, Guidi F, Furbetta F. Technical Details and Result of a Minimally Invasive Management of Gastric Band Erosions: a Series of 47 Patients. Obes Surg. 2019;29(11): 3754-3761
15. Mozzi E, Lattuada E, Zappa MA, Granelli P, De Ruberto F, Armocida A et al. Treatment of band erosion: feasibility and safety of endoscopic band removal. Surg Endosc. 2011;25(12):3918-22
16. Blero D, Eisendrath P, Vandermeeren A, Closset J, Mehdi A, Le Moine O et al. Endoscopic removal of dysfunctioning bands or rings after restrictive bariatric procedures. Gastrointest Endosc. 2010; 71(3):468-74
17. Kohn GP, Hansen CA, Gilhome RW, McHenry RC, Spilias DC, Hensman C. Laparoscopic management of gastric band erosions: a 10-year series of 49 cases. Surg Endosc. 2012;26(2):541-5
Full Text Sources:
Abstract:
Views: 512

For Authors
Journal Subscriptions

Dec 2025
Supplements
Instructions for authors
Online submission
Contact
e-ISSN: 2601 - 1700 (online)
ISSN-L: 2559 - 723X
Journal Abbreviation: Surg. Gastroenterol. Oncol.
Surgery, Gastroenterology and Oncology (SGO) is indexed in:
- SCOPUS
- EBSCO
- DOI/Crossref
- Google Scholar
- SCImago
- Harvard Library
- Open Academic Journals Index (OAJI)
Surgery, Gastroenterology and Oncology (SGO) is an open-access, peer-reviewed online journal published by Celsius Publishing House. The journal allows readers to read, download, copy, distribute, print, search, or link to the full text of its articles.
Time to first editorial decision: 25 days
Rejection rate: 61%
CiteScore: 0.2

Meetings and Courses in 2025
Meetings and Courses in 2024
Meetings and Courses in 2023
Meetings and Courses in 2022
Meetings and Courses in 2021
Meetings and Courses in 2020
Meetings and Courses in 2019
Verona expert meeting 2019

Surgery, Gastroenterology and Oncology applies the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits readers to copy and redistribute the material in any medium or format, remix, adapt, build upon the published works non-commercially, and license the derivative works on different terms, provided the original material is properly cited and the use is non-commercial. Please see: https://creativecommons.org/licenses/by-nc/4.0/
Publisher’s Note:
The opinions, statements, and data contained in article are solely those of the authors and not of Surgery, Gastroenterology and Oncology journal or the editors. Publisher and the editors disclaim responsibility for any damage resulting from any ideas, instructions, methods, or products referred to in the content.
 IASGO Society News
IASGO Society News