Surgery, Gastroenterology and Oncology

|
|
Background: nephrotoxicity is one of the significant complications of cisplatin. Aim of study: to evaluate the impact of cisplatin on nephrotoxicity in patients at the National Oncology Teaching Hospital in Al-Najaf Province.
Patients and methods: An observational, cohort study was conducted by 40 patients at the National Hospital for Oncology and Hematology in Al-Najaf Al-Ashraf, Iraq.
Results: The study shows significant differences in urea and creatinine levels before and after cisplatin treatment with p-values of 0.381, 0.036, respectively. Cisplatin treatment
significantly increased urea and creatinine levels in both genders, with females experiencing a more pronounced decrease after four months than men.
Conclusion: Our findings indicate a high incidence of nephrotoxicity in Cisplatin patients, with males at greater risk than females. This is influenced by Urea and Creatinine levels before, 2 months after, and 4 months after Cisplatin treatment.
Introduction
Clinically sophisticated and extremely effective, cisplatin is an anticancer medication used to treat a variety of solid tumors, including ovarian, stomach, and lung cancer (1).
Kidney disease is a silent condition diagnosed by interpreting changes in kidney function or decreased urine output, but these changes are neither sensitive nor specific to acute kidney injury (AKI) (2).
Nonetheless, nephrotoxicity is the primary side effect of cisplatin administration. Clinically, there is a 20% to 35% probability that individuals using cisplatin would experience nephrotoxicity, which can lead to AKI and death (3).
Since over 90% of cisplatin is excreted in the urine and negatively affects the renal transport system, cisplatin-induced nephrotoxicity arises from a greater degree of cisplatin buildup in the kidney than in another organ through mediated transports (4).
The pathogenic processes of cisplatin-induced nephrotoxicity often Ischemia or necrosis may be observed in the proximal renal tubular epithelial cells, as well as reductions in renal blood flow and glomerular filtration rate (5). Renal tubular epithelial cells lose their brush-like morphology because of cisplatin. Proteinaceous casts arise as a result of the necrosis and progressive loss of epithelial cells brought on by an increase in cisplatin accumulation (5).
Nephrotoxicity, a major barrier to cisplatin treatment, is caused by inflammation, autophagy, oxidative stress, and apoptosis, necessitating further investigation into therapeutic options. Investigating nephrotoxicity therapeutic options requires an understanding of the underlying mechanism.
The kidneys are primarily responsible for excreting cisplatin. The concentration of substances in renal tubular epithelial cells is considerably greater than that present in the blood, highlighting the critical role these cells play in renal function and metabolic processes, and it gets concentrated during excretion. A high concentration of cisplatin in the kidneys results from the secretion and reabsorption of cisplatin and its metabolites in the renal tubules during glomerular filtration during excretion (6). Recent research shows renal tubular cells absorb cisplatin through copper transporter CTR1 and organic cation transporter OCT2 (7), and secrete it into the lumen by multidrug and toxin extrusion 1 and solute carrier family 47 member 1 (8). Knocking down the OCT2 gene can significantly decrease nephrotoxicity (9). OCT2 mutations in patients reduce cisplatin transport into renal tubular cells and OCT2 expression, reducing nephrotoxicity (10). Downregulation of CTR1 expression also significantly reduces cisplatin absorption and cytotoxicity (11).
By attaching to DNA and forming adducts that damage DNA, cisplatin mediates its cytotoxic effects. Water molecules take the place of cisplatin's chloride ligand in an aqueous environment to create a positively charged hydrated complex ion, which is then carried to the nucleus by DNA electrostatic attraction. Then, in rapidly growing cells, this complex attaches itself to DNA to create an adduct, which causes DNA cross-linking and stops DNA synthesis and replication (12,13).
Cisplatin induces apoptosis through caspase [as both dependent and independent] pathways. When entering renal tubular epithelial cells, BAX triggers caspase-2, releasing cytochrome c, apoptosis-inducing factor, and SMAC/DIABLO. Also, p53 activates the mitochondrial pathway in cisplatin-induced cytotoxicity, altering proapoptotic protein BAX and mitochondrial membrane integrity, leading to apoptosis and downregulation of antiapoptotic proteins (14).
Cisplatin can trigger the endoplasmic reticulum stress-mediated apoptotic pathway by affecting the cytochrome P450 enzymatic system, causing oxidative stress and activating caspase-12, leading to apoptosis. As anticipated, cytochrome P450, family 2, subfamily E, polypeptide 1 [Cyp2e1]-knockout animals and anti-caspase-12 antibody transfections have shown significant reductions in cisplatin-induced apoptosis (15-18).
Recent research indicates oxidative and nitrosative stress are key factors in cisplatin-induced nephrotoxicity, with antioxidants and reactive oxygen species [ROS] scavengers showing strong protective effects against this toxicity (19).
Cisplatin-induced nephrotoxicity is linked to inflammatory response, with elevated tumor necrotic factor [TNF-?] expression in a nephrotoxic mouse. Dimethyl thiourea, a hydroxyl radical scavenger, protects against cisplatin-induced kidney damage by inhibiting p38 MAPK activation and TNF-? mRNA expression in murine kidneys (20).
By limiting increases in autophagy and apoptosis in NRK-52E cells treated with cisplatin, autophagy is shown to be essential for preserving cellular homeostasis and avoiding cisplatin-induced nephrotoxicity (21).
This study evaluates cisplatin's impact on nephrotoxicity in patients at the National Oncology Teaching Hospital, aiming to quantify its effects on renal function and uncover underlying mechanisms. Analyzing clinical data, serum creatinine levels, and urine output will improve patient management strategies.
Patients and methods
Study Design
The observational cohort study was conducted based on the laboratory analyses of patients at the National Hospital for Oncology and Hematology in Al-Najaf Al-Ashraf. The study took place from October to December 2023. The patients included in the study had various types of tumors, such as metastatic bladder cancer, head and neck cancer, non-small cell lung cancer, ovarian cancer, and testicular cancer. All patients were treated with cisplatin as one of the anti-tumor medications.
Renal Function Markers
The laboratory analysis results focused on serum creatinine and blood urea nitrogen (BUN) to observe the incidence of nephropathy among patients using cisplatin.
Data Collection
The results of this study were obtained from the hospital's laboratory, with contributions from the consultant doctor, pharmacists, and medical analysts. Our analysis included laboratory results from approximately 40 patients, evenly divided between 20 male and 20 female patients.
Statistical Analysis
All data were analyzed using the Statistical Package for the Social Sciences (SPSS) software, version 26, and Microsoft Excel 2019. Results are expressed as mean ± standard deviation (SD). The significance of differences was evaluated at [p-value less than 0.05].
Results
In the table presented (table 1), there is a noticeable difference in urea and creatinine levels before and after the administration of cisplatin. The first pair indicates a significant increase in urea levels in 40 patients following cisplatin treatment, suggesting a strong correlation with nephrotoxicity. In contrast, the second pair shows the changes in creatinine levels, which, while elevated, do not demonstrate as pronounced a change as the urea levels. The p-value associated with these differences indicates that the changes in creatinine levels are not as statistically significant as those observed in urea levels. This suggests that while both markers indicate kidney function, urea levels may provide a more sensitive indication of nephrotoxic effects following cisplatin administration in this patient cohort.
Table 1 - Comparison of creatinine and urea levels before, 2 months after, and 4 months after cisplatin administration.
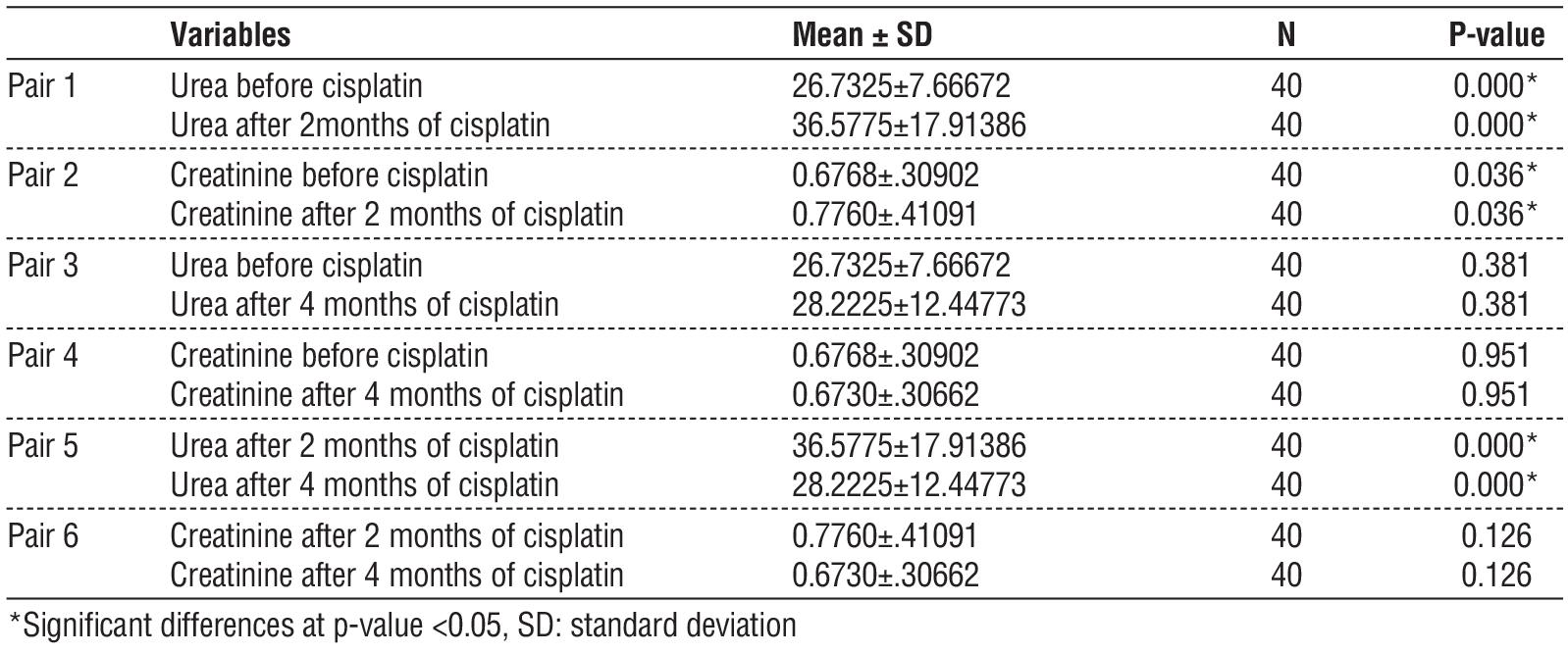
In pairs 3 and 4, when we compared the levels of urea and creatinine, we found an insignificant p-value, indicating a reduced risk of nephrotoxicity after four months of Cisplatin administration due to appropriate monitoring.
In pair 5, when comparing urea levels after two and four months of Cisplatin treatment, we observed a significant p-value, suggesting that urea levels markedly decreased after four months. However, in pair 6, which examined the differences in creatinine levels after two and four months, the p-value was insignificant, indicating that creatinine levels did not show a significant reduction.
The following table (table 2) illustrates the variation in results between male and female patients, highlighting the impact of gender on the outcomes. We observe differences in the levels of creatinine and urea between males and females, even before the administration of cisplatin, as indicated by the p-value. Female patients exhibited lower levels of urea and creatinine compared to male patients.
Table 2 - Comparison of creatinine and urea levels before, after, and 4 months following cisplatin administration based on gender differences.
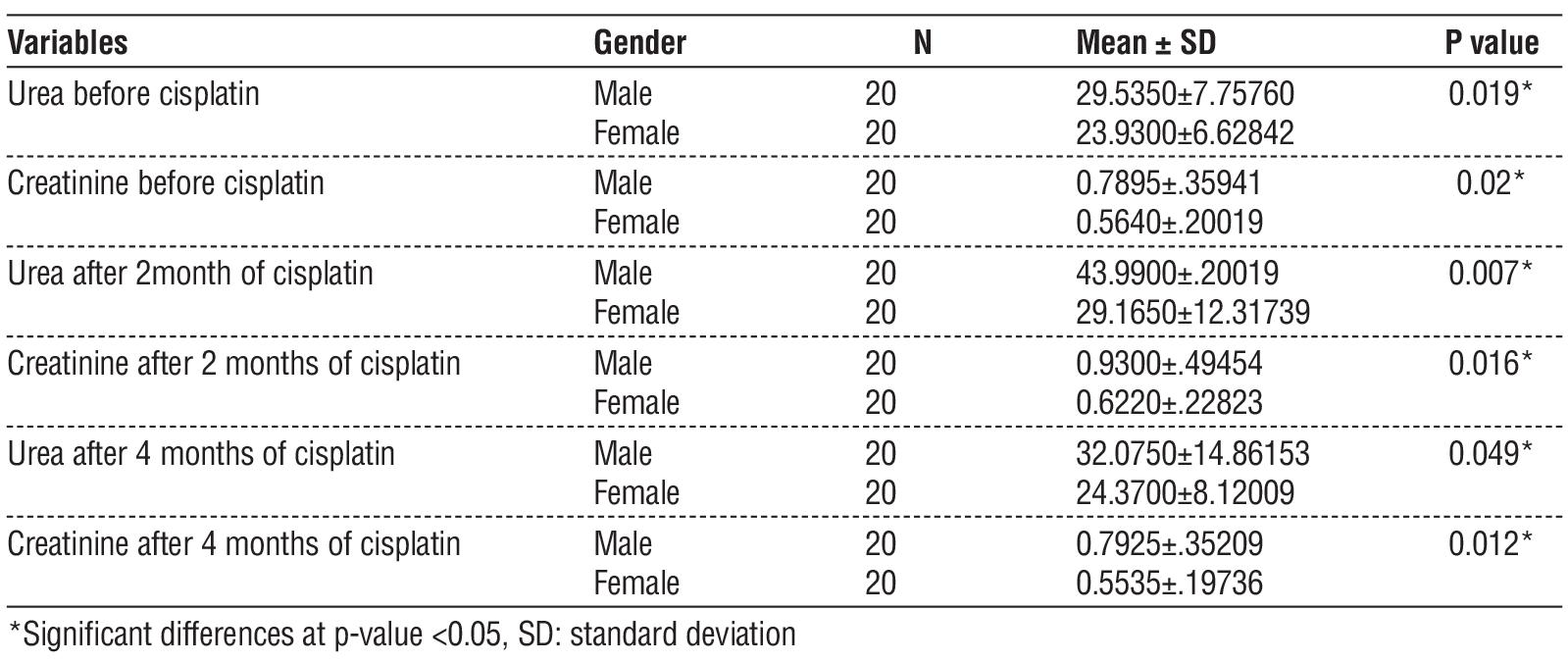
After two months of cisplatin administration, both male and female patients showed an increase in urea and creatinine levels, with the p-value indicating significant differences. Furthermore, after four months, the decrease in urea and creatinine levels in female patients was more pronounced than in male patients, with the p-value again showing significant differences.
Discussion
Michele Peyrone synthesized cisplatin, also known as Peyronie chloride, in 1845. Furthermore, cisplatin (dichlorodiamino platinum) became a widely used inorganic platinum-based chemotherapy agent for treating various malignant solid tumors (22).
Cisplatin is a chemotherapy drug used to treat a variety of cancers, including testicular, ovarian, bladder, head and neck, esophageal, lung, breast, cervical, gastric, and prostate cancers. It is also effective against Hodgkin's and non-Hodgkin's lymphoma, melanoma, and mesothelioma. Cisplatin works by activating the apoptotic pathway, which causes cell damage through oxidative stress and inflammation (22, 23).
As shown in our findings, a minor portion of filtered creatinine is reabsorbed in the tubules, while a larger amount [up to 10%] is actively secreted into the urine by the tubules. The glomerular filtration rate (GFR) can decrease by 50% before plasma creatinine concentrations rise above the normal range. Moreover, when GFR decreases by an additional 50%, the plasma creatinine concentration doubles. As a result, normal plasma creatinine levels do not necessarily indicate normal renal function; however, elevated creatinine levels typically suggest impaired kidney function (24).
Another study has indicated that kidney damage is evidenced by a significant rise in serum BUN and creatinine levels when compared to the control group. These findings align with those of earlier research that demonstrated cisplatin's nephrotoxic effects. Cisplatin can lead to significant complications, including damage to the tubular structures of the kidneys, inflammation in the surrounding interstitial areas, and injury to the blood vessels. These effects highlight the importance of monitoring renal health when using this powerful chemotherapy agent (25).
The increased levels of BUN (Blood Urea Nitrogen) and creatinine serve as critical markers for assessing kidney function and can help in the early detection of nephrotoxicity, allowing for timely intervention. Understanding the mechanisms underlying cisplatin-induced kidney damage is crucial for developing strategies to mitigate these adverse effects and improve patient outcomes.
Due to variations in hormone levels, men might have a greater risk of experiencing kidney failure at an earlier age compared to women. In men, high testosterone levels may play a role in the deterioration of kidney function. On the other hand, due to higher levels of estrogen in females, women’s kidneys may experience protective effects from this hormone (26,27).
Another study indicated the difference in gender in association on nephrotoxicity induced by cisplatin has yielded differing conclusions about the existence of sex differences. According to some studies, women may have greater resistance to nephrotoxicity because of their more robust antioxidant defenses and the protective properties of estrogen. Other studies, however, have suggested that due to their higher clearance rates of cisplatin, males may be less vulnerable than females (28).
Another hypothesis proposes that the kidneys might actively gather cisplatin, as indicated by its greater concentration in kidney tissue than in blood. Cisplatin penetrates cells via two kinds of membrane transporters: the copper transporter (CTR1) and the organic cation transporter (OCT2). Reports indicate that OCT2 levels are considerably greater in males and show a positive correlation with nephrotoxicity (29).
After four months of cisplatin administration, implementing active hydration with saline and simultaneous administration of mannitol before, during, and after treatment has been shown to significantly reduce nephrotoxicity associated with cisplatin. This strategy is now widely accepted as the standard of care to mitigate the drug's side effects.
Strategies for protecting against cisplatin toxicity include Enhancing drug elimination via intravenous hydration, judicious use of osmotic diuretics, and reducing nephrotoxic medications (30).
Conclusions
Our findings show a high incidence of nephrotoxicity in Cisplatin patients, with males at greater risk than females. Elevated Urea and Creatinine levels are noted in males both before treatment and after two months, with Urea increasing significantly more than Creatinine. After four months, monitoring leads to a decrease in both levels. The decision-making process for patients should be approached cautiously, considering factors such as sex, age, health status, medical history, and other medications that may increase toxicity risk. It is important to assess treatment goals and the patient's susceptibility to cisplatin-related toxicities.
Additionally, it's vital to correct risk factors for nephrotoxicity, ensure adequate hydration during treatment, and option for non-nephrotoxic alternatives when possible.
Conflict of Interests
There is no conflict of interest.
Funds
There is no funding for our study.
References
1. Dasari S, Tchounwou PB. Cisplatin in cancer therapy: molecular mechanisms of action. Eur J Pharmacol. 2014;740:364-78.
2. Kale A, Shelke V, Sankrityayan H, Dagar N, Gaikwad AB. Klotho restoration via ACE2 activation: A potential therapeutic strategy against acute kidney injury-diabetes comorbidity. Biochim Biophys Acta Mol Basis Dis. 2022;1868(12):166532.
3. Pierson-Marchandise M, Gras V, Moragny J, Micallef J, Gaboriau L, Picard S, et al. The drugs that mostly frequently induce acute kidney injury: a case - noncase study of a pharmacovigilance database. Br J Clin Pharmacol. 2017;83(6):1341-9.
4. Ruggiero A, Rizzo D, Trombatore G, Maurizi P, Riccardi R. The ability of mannitol to decrease cisplatin-induced nephrotoxicity in children: real or not? Cancer Chemother Pharmacol. 2016;77:19-26.
5. Tang C, Livingston MJ, Safirstein R, Dong Z. Cisplatin nephrotoxicity: new insights and therapeutic implications. Nat Rev Nephrol. 2023; 19(1):53-72.
6. Fang C-y, Lou D-y, Zhou L-q, Wang J-c, Yang B, He Q-j, et al. Natural products: potential treatments for cisplatin-induced nephrotoxicity. Acta Pharmacol Sin. 2021;42(12):1951-69.
7. Yonezawa A, Masuda S, Nishihara K, Yano I, Katsura T, Inui K-i. Association between tubular toxicity of cisplatin and expression of organic cation transporter rOCT2 (Slc22a2) in the rat. Biochem Pharmacol. 2005;70(12):1823-31.
8. Iwata K, Aizawa K, Kamitsu S, Jingami S, Fukunaga E, Yoshida M, et al. Effects of genetic variants in SLC22A2 organic cation transporter 2 and SLC47A1 multidrug and toxin extrusion 1 transporter on cisplatin-induced adverse events. Clin Exp Nephrol. 2012;16:843-51.
9. Filipski KK, Mathijssen RH, Mikkelsen TS, Schinkel AH, Sparreboom A. Contribution of organic cation transporter 2 (OCT2) to cisplatin-induced nephrotoxicity. Clin Pharmacol Ther. 2009;86(4):396-402.
10. Ciarimboli G. Membrane transporters as mediators of cisplatin side-effects. Anticancer Res. 2014;34(1):547-50.
11. Pabla N, Murphy RF, Liu K, Dong Z. The copper transporter Ctr1 contributes to cisplatin uptake by renal tubular cells during cisplatin nephrotoxicity. Am J Physiol Renal Physiol. 2009;296(3):F505-F11.
12. Wang D, Lippard SJ. Cellular processing of platinum anticancer drugs. Nat Rev Drug Discov. 2005;4(4):307-20.
13. Fujikawa Y, Kawanishi M, Kuraoka I, Yagi T. Frequencies of mutagenic translesion DNA synthesis over cisplatin-guanine intra-strand crosslinks in lacZ plasmids propagated in human cells. Mutat Res Genet Toxicol Environ Mutagen. 2014;770:23-8.
14. Servais H, Ortiz A, Devuyst O, Denamur S, Tulkens PM, Mingeot-Leclercq M-P. Renal cell apoptosis induced by nephrotoxic drugs: cellular and molecular mechanisms and potential approaches to modulation. Apoptosis. 2008;13:11-32.
15. Boyce M, Yuan J. Cellular response to endoplasmic reticulum stress: a matter of life or death. Cell Death Differ. 2006;13(3):363-73.
16. Liu H, Baliga R. Cytochrome P450 2E1 null mice provide novel
protection against cisplatin-induced nephrotoxicity and apoptosis. Kidney international. 2003;63(5):1687-96.
17. Peyrou M, Hanna PE, Cribb AE. Cisplatin, gentamicin, and p-amino-phenol induce markers of endoplasmic reticulum stress in the rat kidneys. Toxicol Sci. 2007;99(1):346-53.
18. Liu H, Baliga R. Endoplasmic reticulum stress–associated caspase 12 mediates cisplatin-induced LLC-PK1 cell apoptosis. J Am Soc Nephrol. 2005;16(7):1985-92.
19. Jiang M, Wang C-Y, Huang S, Yang T, Dong Z. Cisplatin-induced apoptosis in p53-deficient renal cells via the intrinsic mitochondrial pathway. Am J Physiol Renal Physiol. 2009;296(5):F983-F93.
20. Ramesh G, Reeves WB. p38 MAP kinase inhibition ameliorates cisplatin nephrotoxicity in mice. Am J Physiol Renal Physiol. 2005; 289(1):F166-F74.
21. Inoue K, Kuwana H, Shimamura Y, Ogata K, Taniguchi Y, Kagawa T, et al. Cisplatin-induced macroautophagy occurs prior to apoptosis in proximal tubules in vivo. Clin Exp Nephrol. 2010;14:112-22.
22. Manohar S, Leung N. Cisplatin nephrotoxicity: a review of the literature. J Nephrol. 2018;31(1):15-25.
23. Kunze D, Erdmann K, Froehner M, Wirth MP, Fuessel S. siRNA-mediated inhibition of antiapoptotic genes enhances chemotherapy efficacy in bladder cancer cells. Anticancer Res. 2012;32(10):4313-8.
24. Shrivastava A, Kuzontkoski PM, Groopman JE, Prasad A. Cannabidiol induces programmed cell death in breast cancer cells by coordinating the cross-talk between apoptosis and autophagy. Molecular cancer therapeutics. 2011;10(7):1161-72.
25. Siddik ZH. Mechanisms of action of cancer chemotherapeutic agents: DNA-interactive alkylating agents and antitumour platinum-based drugs. The cancer handbook. 2002;1:1-16.
26. Conte C, Antonelli G, Melica ME, Tarocchi M, Romagnani P, Peired AJ. Role of sex hormones in prevalent kidney diseases. Int J Mol Sci. 2023;24(9):8244.
27. Zhao JV, Schooling CM. The role of testosterone in chronic kidney disease and kidney function in men and women: a bi-directional Mendelian randomization study in the UK Biobank. BMC Med. 2020; 18:1-10.
28. Eshraghi-Jazi F, Nematbakhsh M. Sex Difference in Cisplatin-Induced Nephrotoxicity: Laboratory and Clinical Findings. J Toxicol. 2022;2022(1):3507721.
29. El-Arabey AA. Gender difference in Cisplatin-induced nephrotoxicity in a rat model. Nephrourol Mon. 2015;7(2):e23595.
30. Sikking C, Niggebrugge-Mentink KL, van der Sman AE, Smit RH, Bouman-Wammes EW, Beex-Oosterhuis MM, et al. Hydration methods for cisplatin containing chemotherapy: A systematic review. Oncologist. 2024;29(2):e173-e86.
Full Text Sources:
Abstract:
Views: 5988

For Authors
Journal Subscriptions

Dec 2025
Supplements
Instructions for authors
Online submission
Contact
e-ISSN: 2601 - 1700 (online)
ISSN-L: 2559 - 723X
Journal Abbreviation: Surg. Gastroenterol. Oncol.
Surgery, Gastroenterology and Oncology (SGO) is indexed in:
- SCOPUS
- EBSCO
- DOI/Crossref
- Google Scholar
- SCImago
- Harvard Library
- Open Academic Journals Index (OAJI)
Surgery, Gastroenterology and Oncology (SGO) is an open-access, peer-reviewed online journal published by Celsius Publishing House. The journal allows readers to read, download, copy, distribute, print, search, or link to the full text of its articles.
Time to first editorial decision: 25 days
Rejection rate: 61%
CiteScore: 0.2

Meetings and Courses in 2025
Meetings and Courses in 2024
Meetings and Courses in 2023
Meetings and Courses in 2022
Meetings and Courses in 2021
Meetings and Courses in 2020
Meetings and Courses in 2019
Verona expert meeting 2019

Surgery, Gastroenterology and Oncology applies the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits readers to copy and redistribute the material in any medium or format, remix, adapt, build upon the published works non-commercially, and license the derivative works on different terms, provided the original material is properly cited and the use is non-commercial. Please see: https://creativecommons.org/licenses/by-nc/4.0/
Publisher’s Note:
The opinions, statements, and data contained in article are solely those of the authors and not of Surgery, Gastroenterology and Oncology journal or the editors. Publisher and the editors disclaim responsibility for any damage resulting from any ideas, instructions, methods, or products referred to in the content.
 IASGO Society News
IASGO Society News