Surgery, Gastroenterology and Oncology
Official journal of the International Association of Surgeons, Gastroenterologists and Oncologists

|
|
Implications of Bacteriological Study in Perforated Peptic Ulcer Peritonitis
Background: In the setting of perforated peptic ulcer (PPU) peritonitis, empiric antimicrobial
therapy is initiated perioperatively and adapted according to the culture sensitivity. The aim
of the study is to describe the microbiota found in the peritonitis due to a PPU, and to
evaluate the predictors for bacterial or fungal infection.
Material and methods: We performed a single-centre, retrospective observational study of all consecutive patients who presented with PPU peritonitis and underwent emergent surgery in Saint Pierre University Hospital, Brussels, Belgium, between January 2013 and December 2020. The medical history, parameters at admission, bacterial culture, antibiotic resistance and postoperative outcomes were analysed.
Results: A total of 43 patients were included in the study. The microbiological culture rate was positive in 31% (13/43) patients. The bacterial culture revealed that the most frequently isolated bacteria were Klebsiella spp. and Enterobacter in 7 % (3/43) of the patients, while the most prevalent fungus isolation was Candida spp. in 16.7 % (7/43) patients. The most prevalent resistances were against ampicillin (17.1 % [7/43]). The Charlson Comorbidity Index was an independent predictor for bacterial infection.
Conclusion: Candida spp., Klebsiella spp. and Enterobacter were the most common organisms isolated in the setting of PPU peritonitis.
BACKGROUND
Peptic-ulcer disease (PUD) affects 4 million people around the world. One of its leading factors is Helicobacter pylori infection (1). About 50% of the global population presents H. pylori in the gastric mucosa but PUD is present in only 10–20% of people, a prevalence that increases in up to 90% in perforated peptic ulcers (PPU). Other factors include non-steroidal anti-inflammatory drug use, smoking or alcohol (2). Perforations are present in 5% and 20% of complicated ulcers (3). The occurrence of PPU is rare, but remains a life threatening disease. The lifetime prevalence of perforation in patients with PUD is about 5%. In the last decade, more than half of the patients with PPU were older than 70 years and predominantly female (3). The mortality rates vary from 10 40 % (4).
Thirty day mortality rates were reported to be around 20 %, and up to 30% in 90 days (5). The PPU morbidity and mortality can be related to complications like leaks, wound infection, fistula, shock and multiorgan failure (6). Empiric antimicrobial therapy is initiated perioperatively and adapted, if necessarily, accordingly with peritoneal fluid culture sensitivity. This may reduce the incidence of postoperative complications, morbidity, and mortality (7). In the literature, Escherichia coli was the commonest organism isolated from peritonitis due to PPU, and the best bacterial sensibility reported was to piperacillin/tazobactam and cefotaxime (8). The aim of the study is to describe the microbiota found in the peritonitis due to a PPU. The secondary outcome was to evaluate the predictors for bacterial or fungal infection.
MATERIAL AND METHODS
Study design
We performed a single-centre, retrospective observational study of all consecutive patients who presented with peptic-ulcer perforation peritonitis and underwent emergent surgery in Saint Pierre University Hospital, Brussels, Belgium, between January 2013 and December 2020.
The inclusion criteria for the 43 patients analysed were: patients over 18 years old, who underwent surgery related to PPU in an emergency setting with the presence of peritonitis, localisation of the perforation on the stomach or duodenum, and enough quantity of intraabdominal free fluid to allow a bacteriological analysis.
The exclusion criteria were: the presence of peritonitis from a different origin, conservative treatment of PPU, the absence of free fluid, the absence of a bacteriological sample or the absence of bacteriological analysis. The study was approved by the Ethics Committee of our institution.
Intervention
All patients with the diagnosis of PPU peritonitis underwent either an open or laparoscopic emergent surgery for the suture of the perforation. Omentoplasty and drainage placement were performed at surgeons discretion. The intraabdominal free fluid was collected in sterile conditions and sent for bacteriological analysis. Postoperative broad-spectrum antibiotics were prescript postoperatively. The antibiotic treatment was adapted accordingly to the results and sensitivity of the bacterial culture, usually available after 48 hours of treatment. Patients were discharged in the absence of relevant postoperative complications.
Variables
Baseline characteristics collected were: age, sex, nationality, patients personal history of hypertension, dyslipidaemia, gastritis or PUD, the Charlson Comorbidity Index (which predicts the ten-year mortality for a patient according to their comorbid conditions), and personal history of abdominal surgery or other surgeries. At admission, the physical examination signs analysed were: heart rate (HR), mean arterial pressure (MAP), and temperature; and the C-reactive protein and the white cell blood count from the blood tests. The intraoperative variables collected were the localization of the ulcer, the surgical intervention and the drainage placed.
The bacteriological sample was analysed was to identify the presence of bacteria or fungus, and its resistance to antibiotics. We also evaluated the antibiotic treatment prescribed, in the first, second and third line.
Postoperative variables were the hospital stay, blood tests at 48 and 96 hours after surgery, overall morbidity, its severity according to the Clavien Dindo classification, and the need of re intervention and the specific complications. We also evaluated the gastroscopy findings after discharge.
Statistical analysis
Categorical variables were described with numbers and percentages. Quantitative variables were described with mean and standard deviation if they followed a normal distribution and with median and interquartile range (IQR) if they followed a non-normal distribution. The Shapiro–Wilk test was used as normality test. To explore the predictors for bacterial or fungal infection, all variables were tested in univariate logistic regression. Statistically significant variables in univariate analysis were then included in a multivariable logistic regression model, erasing nonsignificant outcomes until all variables were adjusted to each other in the final model. A p-value < 0.05 in a two tailed statistical analysis was considered statistically significant. Statistical analysis was performed with the statistical software SPSS 23.0 for Windows (IBM SPSS Inc., Chicago, IL).
RESULTS
A total of 43 patients were operated for PPU perforation associated with peritonitis by laparotomy or laparoscopy in the marked period and fulfilled the inclusion criteria.
The median age of the study sample was 46 years (IQR: 32 60). There was a male predominance, with 83.7 % (36/43) male and 16,3% (7/43) female patients. The patient’s personal medical history revealed that 9.3 % (4/43) patients had a documented history of gastritis or previous peptic ulcer, and 18.6 % (8/43) demonstrated history of previous abdominal surgeries. The median score on Charlson Comorbidity Index was 1 (IQR: 0 2). (table 1)
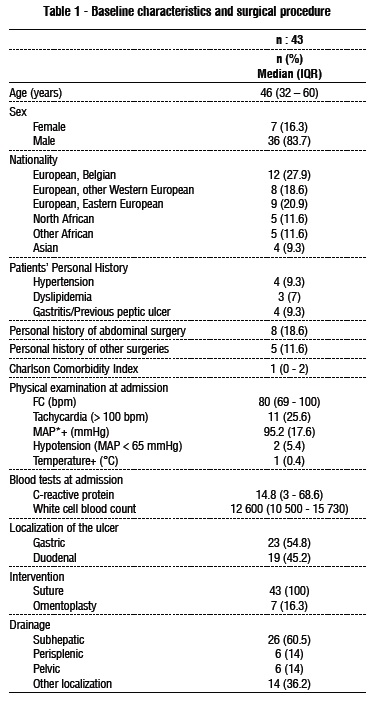
Preoperative assessment
The physical examination at admission revealed that in the median heart rate was 80 bpm (IQR: 69 100) with tachycardia (>100 bpm) in 25.6% (11/43) of the patients. The mean arterial pressure was 95.2 mmHg (SD: 17.6) with hypotension (< 65 mmHg) in 5.4% (2/43) of the patients. The blood test at the admission was performed, and the inflammatory syndrome was evaluated. The medium value of the C-reactive protein was 14.8 g/dL (IQR: 3 – 68.6). The median white cell blood count at admission was 12 600/mm3 (IQR: 10 500 15 730/mm3).
Surgical treatment
The localisation of the perforation was recorded intraoperatively and was predominantly gastric localisation in 54.8 % (23/43) patients, while 45.2 % (19/43) patients presented a duodenal perforation. The surgeries performed consisted on a suture of the perforation for all the patients in the study with omentoplasty in only 16.3 % (7/43) patients. An abdominal drainage was placed in subhepatic area and next to the suture in 60.5 % (26/43) patients, in the pelvis and peri-splenic area for 14 % (6/43) patients, and in other localisations in 36.2 % (14/43) patients.
Bacterial culture
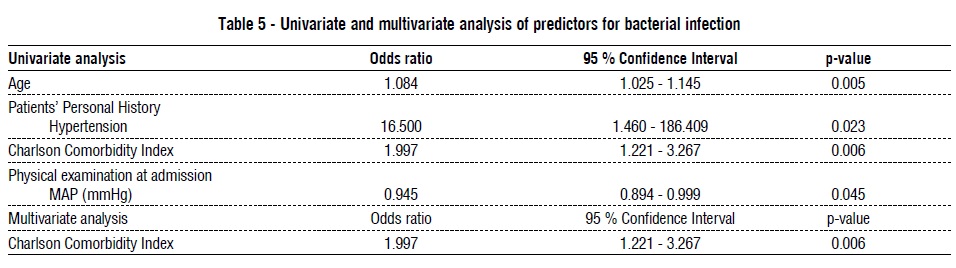
The microbiological culture rate was positive in 31% (13/43) patients. The isolation revealed the presence of bacteria in 20.9 % (9/43) patients and fungal infection in 16.7 % (7/43). The bacterial culture revealed that the most frequently isolated bacteria was Klebsiella spp. and Enterobacter in 7 % (3/43) of the patients, followed by Haemophilus spp. and Streptococcus spp. in 4.7% (2/43) of the patients. The most prevalent fungus isolation was Candida spp., which was positive in 16.7 % (7/43) patients (table 2).
The Enterobacter infection was linked to an older age and more comorbid patients, whereas the Candida spp. cultures were associated with younger patients without significant comorbidities (table 3).

Antibiotic/Antifungal treatment
There was a 17.5 % (7/43) rate of resistance to antibiotics. The most prevalent resistances were against ampicillin (17.1 % [7/43]), amoxicillin/clavulanic acid (9.5 % [4/43]), cefuroxime and trimethoprim/ sulfamethoxazole (4.8% each [2/43]). Other resistances were to cefotaxime, ciprofloxacin, levofloxacin and tigecycline.
Most patients received amoxicillin/clavulanic acid as a first line of treatment (92.9% [39/43]). There was a 7 % (3/43) rate of resistance against the first line of antibiotic treatment. The second line of antibiotics administered were piperacillin/tazobactam and ciprofloxacin ornidazole (2.4% each [1/43]).
Postoperative outcomes
The median hospital stay was 5 days (IQR: 5 7 days). Overall morbidity was 11.6 % (5/43), while 30-day complications according to Clavien Dindo classification revealed a 4.7 % (2/43) grade II complications, 4.7 % (2/43) grade III complications, and one (2.3%) grade V complication, due to irreversible septic shock. Two patients (4.7 %) needed reoperation due to surgical site infection.
A systematic blood test was performed at the second and fourth postoperative days to follow the dynamics of the inflammatory syndrome.
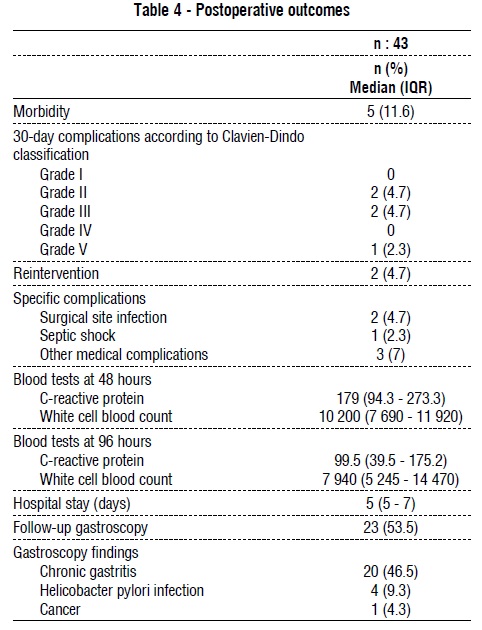
The C-reactive protein at 48 h postoperative presented a median value of 179 mg/dl (IQR: 94.3 - 273.3) and the white cell blood count recorded a median value of 10 200/mm3 (IQR: 7 690 - 11 920). At 96h the tendency to normalisation was more relevant for the white cell blood with a median value of 7 940/mm3 (IQR: 5 245 - 14 470). The C-reactive protein presented also a decreased value of 99.5 mg/dl (IQR: 39.5 - 175.2). A postoperative control gastroscopy was prescribed in 6-8 weeks but only 23 (53.5%) patients underwent the exam. The findings were chronic gastritis (46.5% [20/43]), H. pylori infection after the biopsy analysis (9.3% [4/43]) one case of cancer (4.3%) (table 4).
Predictors of bacterial and fungal infection
All variables were evaluated in univariate analysis as predictors for bacterial or fungal infection, and variables with statistical significance were evaluated in a multivariate analysis. In univariate analysis, the predictors for bacterial infection were: age, personal history of hypertension, the Charlson Comorbidity Index, and the mean arterial pressure at admission. In multivariate analysis only Charlson Comorbidity Index was an independent predictor for bacterial infection. These results are shown in table 5. No factors could be identified as predictors of fungal infection.
DISCUSSION
The incidence of PUD is about 0.1%-0.3% per year with a prevalence that was about 5.7% in 1998, which has been progressively declining (9). PPU in some studies account for more than 70% of deaths associated with PUD. The incidence of duodenal perforation (DP) is 7–10 cases/100,000 adults per year (10). Factors associated with an adverse outcome in patients with complicated PPU include comorbid disease, poor general and medical status, sign of shock and sepsis (hypotensive shock, metabolic acidosis, acute renal failure, hypoalbuminemia), and delayed treatment (11). Independent risk factors accounted for postoperative morbidity and mortality are elevated levels of serum creatinine and advanced age. Adequate resuscitation, sepsis control, addressing comorbidities and early access to hospital can minimize the risk of morbidity and mortality in patients with PPU (12).
Surgical intervention is the most widespread attitude practiced in cases of PPU. The postoperative outcome after surgical repair is influenced by delayed presentation, presence of pus in the peritoneal cavity, sepsis and shock (13). The choice of the surgical approach (laparoscopy versus laparotomy) has an impact on the immune response in PPU. Schietroma et al., in a study on 119 patients with PPU, showed that 1 hour after the intervention, bacteraemia and the levels of endotoxin was significantly higher for the patients who undergo surgery by laparotomy. More, the laparotomy caused a significant increase in immune neutrophil concentration, neutrophil-elastase, IL-1 and IL-6, CRP and decrease of HLA-DR (14). A conservative, non-operative attitude has equally been described. Asanasak et al., in a 9-year retrospective study on 38 patients with PPU who received non-operative treatment, concluded that for proper selected patients this attitude can be successful, with shorter hospital stay and decrease the number of patients that require an operation (15). Whatever the attitude, the broad-spectrum antibiotics administration is essential for the treatment.
Bacteriology in PPU
The mortality rate in patients in case of intraabdominal sepsis can be as high as 18 55 % when grampositive cocci are present. Along the years, due to the improved care of peritonitis, the mortality decreased from 90 % in 1900 to 15–25 % nowadays. The actual challenge is the increased microbial resistance and the administration of an appropriate empiric antibiotic treatment (16). In case of anaerobic intra-abdominal contamination, there is a significant increase in septicaemia without a significant increase in mortality. Gowda et al., in a study on 275 consecutive patients with PPU, found that the factors that increase the risk of anaerobic infection were: age over 50 years, patients’ comorbidity, peritonitis of more than 48 hours, perforation diameter > 5 mm, peritoneal fluid > 1000 ml and purulent contamination (17).
Alwahed et al., in a study on PPU on 888 patients, found that 48.6% of the studied patients had positive cultures for Bacteroides spp., and that patients with increase age (> 50 years) had a higher prevalence of contamination (83.7 %) (18). The review of Brook et al. sustained that anaerobic contamination in PPU was explainable by the fact that in the gastrointestinal tract flora, the ratio of anaerobe bacteria to aerobe bacteria was of 1,000 - 10,000 to 1 (19). Quantitative analyses of intestinal microbiota of the digestive tube revealed a non-homogeneous distribution. The number of bacterial cells in 1 g chime is lesser in the stomach and duodenum (101 – 103) and increase progressively until reaching their maximum in large intestine (1011-1012) (20). In terms of isolated species in PPU, Bhavin et al. in a study on 200 patients with PPU found that E. coli was the commonest organism isolated, and that the patients handled with antibiotics according to culture and sensitivity presented a reduces hospital stay and morbidity (21). Lohith et al. in a study on 50 patients with different sites of perforation on the gastrointestinal tract found that E. coli was the most common organism isolated in all sites of perforation and that there was an increasing resistance against third generation cephalosporins (22). Tayal et al. in a study on 43 intra-operative specimens from cases of perforated peptic ulceration, found that the bacterial culture in PPU revealed also gram-negative bacilli morphologically resembling H. pylori 41.86% specimens with an H. pylori culture positivity of 18.60% (23).
Antibiotic treatment
Initial empirical antibiotic therapy has to be adapted to the bacterial culture results and susceptibility, and it has been suggested that the most potent antibiotics should be used in cases of peritonitis, instead of the most commonly used antibiotics (24). Empiric broadspectrum antibiotic regimen against a mixture of Gramnegative, Gram positive and anaerobic bacteria should be administered, if possible, after the peritoneal fluid has been collected. According to WSES guidelines for perforated and bleeding peptic ulcers, in patients with PUP, a short-course (3–5 days) of antibiotic therapy is recommended (2C) (25).
There is a controversy on the empirical administration of the anti-fungal agents (AF) in PPU. In most cases, fungal infections are present for surgical patients, especially intra-abdominal abscesses. The routine use of empiric AF is not sustained by the literature (26). Barmparas et al., in a study of 554 patients with PPU, found that empiric use of AF presented no clinical advantage in preventing infections, even those due to Candida spp., thus its administration was unnecessary (27). Nevertheless, Lee et al., in a study on 62 patients with PPU, found that 37.1% of patients presented positive culture to Candida spp. This study recommended that AF agents should be administered in all cases of fungal contamination to lower the mortality rate and shorten hospital stay (28). Shan et al., in a taiwanese study on 145 patients with PPU, observed that 43 % patients presented a positive fungal infection and Candida spp. was the most ordinary pathogen isolated from peritoneal fluid and wound cultures. This study recommended low-dose amphotericin B for critically ill surgical patients with intraperitoneal infection (29).
Strenghs and limitations
One of the strengths of this study represents a well exhaustive description of the bacteria isolated in the peritonitis secondary to PPU. However, one of its limitations was a modest number of patients suitable for analysis, mainly due to the reduced prevalence of this clinical scenario compared to other entities such as colonic or appendicular peritonitis. Otherwise, there was a limitation during follow-up because of a reduced number of postoperative upper endoscopies, which Surgery, Gastroenterology and Oncology, 27 (2), 2022 119 Bacteriology in Perforated Peptic Ulcer prevented to analyse the true incidence of H. pylori as the aetiology of the PUD.
CONCLUSION
Candida spp., Klebsiella spp. and Enterobacter were the most common organisms isolated in the microbiological samples of perforated peptic-ulcer peritonitis, with an overall resistance to antibiotics rate of a 17.5 %. The Charlson Comorbidity Index was an independent predictor for bacterial infection, while no factors could be identified as predictors of fungal infection.
Ethical approval and Consent to participate
The study was approved by the Ethical Committee of Saint Pierre University Hospital. The authors confirm that all research was performed in accordance with relevant guidelines/regulations, and the consent was obtained from all participants and/or their legal guardians.
Availability of data and materials
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
Competing interest
No competing interests exist for any of the authors.
Authors contributions
Sorin Cimpean: Conceptualization, Methodology, Software. Alberto Gonzalez Barranquero: Data curation, Writing - Original draft preparation. Benjamin Cadiere: Writing - Reviewing and Editing. Guy Bernard Cadiere: Supervision, Validation.
Funding statement
The authors received no financial support for the research, authorship, and/or publication of this article.
Acknowledgments
I thank to my colleagues for their expertise and assistance throughout all aspects of our study and for their help in writing the manuscript, in particular to the Professor G.B. Cadiere for guidance and support.
REFERENCES
1. Søreide K. Current insight into pathophysiology of gastroduodenal ulcers: Why do only some ulcers perforate? J Trauma Acute Care Surg. 2016 ;80(6):1045-8.
2. Søreide K, Thorsen K, Harrison EM, Bingener J, Møller MH, Ohene- Yeboah M, et al. Perforated peptic ulcer. Lancet. 2015;386(10000): 1288-98.
3. Dadfar A, Edna T-H. Epidemiology of perforating peptic ulcer: A population-based retrospective study over 40 years. World J Gastroenterol. 2020;26(35):5302-5313.
4. Thorsen K. Epidemiology of perforated peptic ulcer: Age - and gender-adjusted analysis of incidence and mortality. World J Gastroenterol. 2013;19(3):347-54.
5. Chung KT, Shelat VG. Perforated peptic ulcer - an update. World J Gastrointest Surg. 2017;9(1):1-12.
6. Malik S, Singh A, Sidhu DS, Nagpal N, Sharma D. A prospective study to assess clinical profile and golden period for operative intervention in patients with perforation peritonitis. Int Surg J. 2018;5(4):1492.
7. Kishore R. VR, Ashwin K, Vasanth Kumar SB. An observational study on peritoneal fluid bacteriology in cases of gastrointestinal perforations, antibiotic management and outcome in tertiary care center. Int Surg J. 2020;7(2):385.
8. Punamiya AR, Chougule PG, Ahuja BR, Singh V, Mohite ST. Commonest Organisms and Antibiotic Sensitivity in Peritonitis Due To Duodenal Ulcer Perforation in Krishna Hospital, Karad. Int J. Health Sciences. 2014;(8):5.
9. Yawar B, Marzouk AM, Ali H, Ghorab TM, Asim A, Bahli Z, et al. Seasonal Variation of Presentation of Perforated Peptic Ulcer Disease: An Overview of Patient Demographics, Management and Outcomes. Cureus. 2021;13(11):e19618.
10. Bekele A, Zemenfes D, Kassa S, Deneke A, Taye M, Wondimu S. Patterns and Seasonal Variations of Perforated Peptic Ulcer Disease: Experience from Ethiopia. Ann Afr Surg. 2018 DOI: 10.4314/aas.v14i2.7
11. Kavitt RT, Lipowska AM, Anyane-Yeboa A, Gralnek IM. Diagnosis and Treatment of Peptic Ulcer Disease. Am J Med. 2019;132(4): 447-456.
12. Ahmed M, Mansoor T, Rab AZ, Rizvi SAA. Risk factors influencing postoperative outcome in patients with perforated peptic ulcer: a prospective cohort study. Eur J Trauma Emerg Surg. 2022;48(1): 81-86.
13. Rohit DK, Verma RS, Pandey G. Clinical study and management of peritonitis secondary to perforated peptic ulcer. Int Surg J. 2017; 4(8):2721.
14. Tan S, Wu G, Zhuang Q, Xi Q, Meng Q, Jiang Y, et al. Laparoscopic versus open repair for perforated peptic ulcer: A meta-analysis of randomized controlled trials. Int J Surg. 2016;33 Pt A:124-32.
15. Asanasak P. The case series of peritonitis due to perforated peptic ulcer: How does conservative management play role? Int J Surg Case Rep. 2019;58:74-76.
16. Grotelüschen R, Heidelmann LM, Lütgehetmann M, Melling N, Reeh M, Ghadban T, et al. Antibiotic sensitivity in correlation to the origin of secondary peritonitis: a single center analysis. Sci Rep. 2020; 10(1):18588.
17. Gowda DB, Kadambari D, Vijayakumar C, Elamurugan T P, Jagdish S. A clinico-microbiological profile in patients with perforated peptic ulcer with special reference to anaerobic organisms: a descriptive study. Int Surg J. 2017;4(1):125-130.
18. ALwahed IA. Bacteroides spp. isolation from pus or peritoneal fluid in patients with perforated peptic ulcer. 2008;4(5):190-198.
19. Brook I. Microbiology and Management of Abdominal Infections. Dig Dis Sci. 2008;53(10):2585-91.
20. Ermetov A. Microbiologic structure of diffuse peritonitis. Medical and Health Science Journal, MHSJ. 2011;7:111-8.
21. Bhosale S, Gujar N, Chougule P. Long-term results of simple closure of perforated duodenal ulcer in the era of proton pump inhibitor and anti-H. Pylori. Therapy. 14 janv 2022
22. Lohith P, Jindal RK, Ghuliani D, Rajshekar P. The anatomical site of perforation peritonitis and their microbiological profile: a crosssectional study. Int Surg J. 26 mars 2020;7(4):1251.
23. Tayal R, De A, Sundaravadanan S, Kumar M, Baveja S. Role of Culture in Cases of Perforated Peptic Ulcers Due To Helicobacter Pylori. 2015;3(3):4.
24. Sudhaharan S, Kanne P, Vemu L, Chavali P, Desmukha SR. Bacteriological profile of intra-abdominal infections in a tertiary care hospital. Iran J Microbiol 2018;10(4):208-214.
25. Tarasconi A, Coccolini F, Biffl WL, Tomasoni M, Ansaloni L, Picetti E, et al. Perforated and bleeding peptic ulcer: WSES guidelines. World J Emerg Surg. 2020;15(1):64.
26. Huston JM, Kreiner L, Ho VP, Sanders JM, Duane TM. Role of Empiric Anti-Fungal Therapy in the Treatment of Perforated Peptic Ulcer Disease: Review of the Evidence and Future Directions. Surg Infect (Larchmt). 2019;20(8):593-600.
27. Barmparas G, Alhaj Saleh A, Huang R, Eaton BC, Bruns BR, Raines A, et al. Empiric antifungals do not decrease the risk for organ space infection in patients with perforated peptic ulcer. Trauma Surg Acute Care Open. mai 2021;6(1):e000662.
28. Lee S-C, Fung C-P, Chen H-Y, Li C-T, Jwo S-C, Hung Y-B, et al. Candida peritonitis due to peptic ulcer perforation: incidence rate, risk factors, prognosis and susceptibility to fluconazole and amphotericin B. Diagn Microbiol Infect Dis. 2002;44(1):23-7.
29. Shan Y-S, Hsu H-P, Hsieh Y-H, Sy ED, Lee J-C, Lin P-W. Significance of intraoperative peritoneal culture of fungus in perforated peptic ulcer. Br J Surg. 2003;90(10):1215-9.
Full Text Sources:
Abstract:
Views: 2584

Material and methods: We performed a single-centre, retrospective observational study of all consecutive patients who presented with PPU peritonitis and underwent emergent surgery in Saint Pierre University Hospital, Brussels, Belgium, between January 2013 and December 2020. The medical history, parameters at admission, bacterial culture, antibiotic resistance and postoperative outcomes were analysed.
Results: A total of 43 patients were included in the study. The microbiological culture rate was positive in 31% (13/43) patients. The bacterial culture revealed that the most frequently isolated bacteria were Klebsiella spp. and Enterobacter in 7 % (3/43) of the patients, while the most prevalent fungus isolation was Candida spp. in 16.7 % (7/43) patients. The most prevalent resistances were against ampicillin (17.1 % [7/43]). The Charlson Comorbidity Index was an independent predictor for bacterial infection.
Conclusion: Candida spp., Klebsiella spp. and Enterobacter were the most common organisms isolated in the setting of PPU peritonitis.
BACKGROUND
Peptic-ulcer disease (PUD) affects 4 million people around the world. One of its leading factors is Helicobacter pylori infection (1). About 50% of the global population presents H. pylori in the gastric mucosa but PUD is present in only 10–20% of people, a prevalence that increases in up to 90% in perforated peptic ulcers (PPU). Other factors include non-steroidal anti-inflammatory drug use, smoking or alcohol (2). Perforations are present in 5% and 20% of complicated ulcers (3). The occurrence of PPU is rare, but remains a life threatening disease. The lifetime prevalence of perforation in patients with PUD is about 5%. In the last decade, more than half of the patients with PPU were older than 70 years and predominantly female (3). The mortality rates vary from 10 40 % (4).
Thirty day mortality rates were reported to be around 20 %, and up to 30% in 90 days (5). The PPU morbidity and mortality can be related to complications like leaks, wound infection, fistula, shock and multiorgan failure (6). Empiric antimicrobial therapy is initiated perioperatively and adapted, if necessarily, accordingly with peritoneal fluid culture sensitivity. This may reduce the incidence of postoperative complications, morbidity, and mortality (7). In the literature, Escherichia coli was the commonest organism isolated from peritonitis due to PPU, and the best bacterial sensibility reported was to piperacillin/tazobactam and cefotaxime (8). The aim of the study is to describe the microbiota found in the peritonitis due to a PPU. The secondary outcome was to evaluate the predictors for bacterial or fungal infection.
MATERIAL AND METHODS
Study design
We performed a single-centre, retrospective observational study of all consecutive patients who presented with peptic-ulcer perforation peritonitis and underwent emergent surgery in Saint Pierre University Hospital, Brussels, Belgium, between January 2013 and December 2020.
The inclusion criteria for the 43 patients analysed were: patients over 18 years old, who underwent surgery related to PPU in an emergency setting with the presence of peritonitis, localisation of the perforation on the stomach or duodenum, and enough quantity of intraabdominal free fluid to allow a bacteriological analysis.
The exclusion criteria were: the presence of peritonitis from a different origin, conservative treatment of PPU, the absence of free fluid, the absence of a bacteriological sample or the absence of bacteriological analysis. The study was approved by the Ethics Committee of our institution.
Intervention
All patients with the diagnosis of PPU peritonitis underwent either an open or laparoscopic emergent surgery for the suture of the perforation. Omentoplasty and drainage placement were performed at surgeons discretion. The intraabdominal free fluid was collected in sterile conditions and sent for bacteriological analysis. Postoperative broad-spectrum antibiotics were prescript postoperatively. The antibiotic treatment was adapted accordingly to the results and sensitivity of the bacterial culture, usually available after 48 hours of treatment. Patients were discharged in the absence of relevant postoperative complications.
Variables
Baseline characteristics collected were: age, sex, nationality, patients personal history of hypertension, dyslipidaemia, gastritis or PUD, the Charlson Comorbidity Index (which predicts the ten-year mortality for a patient according to their comorbid conditions), and personal history of abdominal surgery or other surgeries. At admission, the physical examination signs analysed were: heart rate (HR), mean arterial pressure (MAP), and temperature; and the C-reactive protein and the white cell blood count from the blood tests. The intraoperative variables collected were the localization of the ulcer, the surgical intervention and the drainage placed.
The bacteriological sample was analysed was to identify the presence of bacteria or fungus, and its resistance to antibiotics. We also evaluated the antibiotic treatment prescribed, in the first, second and third line.
Postoperative variables were the hospital stay, blood tests at 48 and 96 hours after surgery, overall morbidity, its severity according to the Clavien Dindo classification, and the need of re intervention and the specific complications. We also evaluated the gastroscopy findings after discharge.
Statistical analysis
Categorical variables were described with numbers and percentages. Quantitative variables were described with mean and standard deviation if they followed a normal distribution and with median and interquartile range (IQR) if they followed a non-normal distribution. The Shapiro–Wilk test was used as normality test. To explore the predictors for bacterial or fungal infection, all variables were tested in univariate logistic regression. Statistically significant variables in univariate analysis were then included in a multivariable logistic regression model, erasing nonsignificant outcomes until all variables were adjusted to each other in the final model. A p-value < 0.05 in a two tailed statistical analysis was considered statistically significant. Statistical analysis was performed with the statistical software SPSS 23.0 for Windows (IBM SPSS Inc., Chicago, IL).
RESULTS
A total of 43 patients were operated for PPU perforation associated with peritonitis by laparotomy or laparoscopy in the marked period and fulfilled the inclusion criteria.
The median age of the study sample was 46 years (IQR: 32 60). There was a male predominance, with 83.7 % (36/43) male and 16,3% (7/43) female patients. The patient’s personal medical history revealed that 9.3 % (4/43) patients had a documented history of gastritis or previous peptic ulcer, and 18.6 % (8/43) demonstrated history of previous abdominal surgeries. The median score on Charlson Comorbidity Index was 1 (IQR: 0 2). (table 1)

Preoperative assessment
The physical examination at admission revealed that in the median heart rate was 80 bpm (IQR: 69 100) with tachycardia (>100 bpm) in 25.6% (11/43) of the patients. The mean arterial pressure was 95.2 mmHg (SD: 17.6) with hypotension (< 65 mmHg) in 5.4% (2/43) of the patients. The blood test at the admission was performed, and the inflammatory syndrome was evaluated. The medium value of the C-reactive protein was 14.8 g/dL (IQR: 3 – 68.6). The median white cell blood count at admission was 12 600/mm3 (IQR: 10 500 15 730/mm3).
Surgical treatment
The localisation of the perforation was recorded intraoperatively and was predominantly gastric localisation in 54.8 % (23/43) patients, while 45.2 % (19/43) patients presented a duodenal perforation. The surgeries performed consisted on a suture of the perforation for all the patients in the study with omentoplasty in only 16.3 % (7/43) patients. An abdominal drainage was placed in subhepatic area and next to the suture in 60.5 % (26/43) patients, in the pelvis and peri-splenic area for 14 % (6/43) patients, and in other localisations in 36.2 % (14/43) patients.
Bacterial culture
The microbiological culture rate was positive in 31% (13/43) patients. The isolation revealed the presence of bacteria in 20.9 % (9/43) patients and fungal infection in 16.7 % (7/43). The bacterial culture revealed that the most frequently isolated bacteria was Klebsiella spp. and Enterobacter in 7 % (3/43) of the patients, followed by Haemophilus spp. and Streptococcus spp. in 4.7% (2/43) of the patients. The most prevalent fungus isolation was Candida spp., which was positive in 16.7 % (7/43) patients (table 2).
The Enterobacter infection was linked to an older age and more comorbid patients, whereas the Candida spp. cultures were associated with younger patients without significant comorbidities (table 3).

Antibiotic/Antifungal treatment
There was a 17.5 % (7/43) rate of resistance to antibiotics. The most prevalent resistances were against ampicillin (17.1 % [7/43]), amoxicillin/clavulanic acid (9.5 % [4/43]), cefuroxime and trimethoprim/ sulfamethoxazole (4.8% each [2/43]). Other resistances were to cefotaxime, ciprofloxacin, levofloxacin and tigecycline.
Most patients received amoxicillin/clavulanic acid as a first line of treatment (92.9% [39/43]). There was a 7 % (3/43) rate of resistance against the first line of antibiotic treatment. The second line of antibiotics administered were piperacillin/tazobactam and ciprofloxacin ornidazole (2.4% each [1/43]).
Postoperative outcomes
The median hospital stay was 5 days (IQR: 5 7 days). Overall morbidity was 11.6 % (5/43), while 30-day complications according to Clavien Dindo classification revealed a 4.7 % (2/43) grade II complications, 4.7 % (2/43) grade III complications, and one (2.3%) grade V complication, due to irreversible septic shock. Two patients (4.7 %) needed reoperation due to surgical site infection.

A systematic blood test was performed at the second and fourth postoperative days to follow the dynamics of the inflammatory syndrome.

The C-reactive protein at 48 h postoperative presented a median value of 179 mg/dl (IQR: 94.3 - 273.3) and the white cell blood count recorded a median value of 10 200/mm3 (IQR: 7 690 - 11 920). At 96h the tendency to normalisation was more relevant for the white cell blood with a median value of 7 940/mm3 (IQR: 5 245 - 14 470). The C-reactive protein presented also a decreased value of 99.5 mg/dl (IQR: 39.5 - 175.2). A postoperative control gastroscopy was prescribed in 6-8 weeks but only 23 (53.5%) patients underwent the exam. The findings were chronic gastritis (46.5% [20/43]), H. pylori infection after the biopsy analysis (9.3% [4/43]) one case of cancer (4.3%) (table 4).
Predictors of bacterial and fungal infection
All variables were evaluated in univariate analysis as predictors for bacterial or fungal infection, and variables with statistical significance were evaluated in a multivariate analysis. In univariate analysis, the predictors for bacterial infection were: age, personal history of hypertension, the Charlson Comorbidity Index, and the mean arterial pressure at admission. In multivariate analysis only Charlson Comorbidity Index was an independent predictor for bacterial infection. These results are shown in table 5. No factors could be identified as predictors of fungal infection.
DISCUSSION
The incidence of PUD is about 0.1%-0.3% per year with a prevalence that was about 5.7% in 1998, which has been progressively declining (9). PPU in some studies account for more than 70% of deaths associated with PUD. The incidence of duodenal perforation (DP) is 7–10 cases/100,000 adults per year (10). Factors associated with an adverse outcome in patients with complicated PPU include comorbid disease, poor general and medical status, sign of shock and sepsis (hypotensive shock, metabolic acidosis, acute renal failure, hypoalbuminemia), and delayed treatment (11). Independent risk factors accounted for postoperative morbidity and mortality are elevated levels of serum creatinine and advanced age. Adequate resuscitation, sepsis control, addressing comorbidities and early access to hospital can minimize the risk of morbidity and mortality in patients with PPU (12).
Surgical intervention is the most widespread attitude practiced in cases of PPU. The postoperative outcome after surgical repair is influenced by delayed presentation, presence of pus in the peritoneal cavity, sepsis and shock (13). The choice of the surgical approach (laparoscopy versus laparotomy) has an impact on the immune response in PPU. Schietroma et al., in a study on 119 patients with PPU, showed that 1 hour after the intervention, bacteraemia and the levels of endotoxin was significantly higher for the patients who undergo surgery by laparotomy. More, the laparotomy caused a significant increase in immune neutrophil concentration, neutrophil-elastase, IL-1 and IL-6, CRP and decrease of HLA-DR (14). A conservative, non-operative attitude has equally been described. Asanasak et al., in a 9-year retrospective study on 38 patients with PPU who received non-operative treatment, concluded that for proper selected patients this attitude can be successful, with shorter hospital stay and decrease the number of patients that require an operation (15). Whatever the attitude, the broad-spectrum antibiotics administration is essential for the treatment.

Bacteriology in PPU
The mortality rate in patients in case of intraabdominal sepsis can be as high as 18 55 % when grampositive cocci are present. Along the years, due to the improved care of peritonitis, the mortality decreased from 90 % in 1900 to 15–25 % nowadays. The actual challenge is the increased microbial resistance and the administration of an appropriate empiric antibiotic treatment (16). In case of anaerobic intra-abdominal contamination, there is a significant increase in septicaemia without a significant increase in mortality. Gowda et al., in a study on 275 consecutive patients with PPU, found that the factors that increase the risk of anaerobic infection were: age over 50 years, patients’ comorbidity, peritonitis of more than 48 hours, perforation diameter > 5 mm, peritoneal fluid > 1000 ml and purulent contamination (17).
Alwahed et al., in a study on PPU on 888 patients, found that 48.6% of the studied patients had positive cultures for Bacteroides spp., and that patients with increase age (> 50 years) had a higher prevalence of contamination (83.7 %) (18). The review of Brook et al. sustained that anaerobic contamination in PPU was explainable by the fact that in the gastrointestinal tract flora, the ratio of anaerobe bacteria to aerobe bacteria was of 1,000 - 10,000 to 1 (19). Quantitative analyses of intestinal microbiota of the digestive tube revealed a non-homogeneous distribution. The number of bacterial cells in 1 g chime is lesser in the stomach and duodenum (101 – 103) and increase progressively until reaching their maximum in large intestine (1011-1012) (20). In terms of isolated species in PPU, Bhavin et al. in a study on 200 patients with PPU found that E. coli was the commonest organism isolated, and that the patients handled with antibiotics according to culture and sensitivity presented a reduces hospital stay and morbidity (21). Lohith et al. in a study on 50 patients with different sites of perforation on the gastrointestinal tract found that E. coli was the most common organism isolated in all sites of perforation and that there was an increasing resistance against third generation cephalosporins (22). Tayal et al. in a study on 43 intra-operative specimens from cases of perforated peptic ulceration, found that the bacterial culture in PPU revealed also gram-negative bacilli morphologically resembling H. pylori 41.86% specimens with an H. pylori culture positivity of 18.60% (23).
Antibiotic treatment
Initial empirical antibiotic therapy has to be adapted to the bacterial culture results and susceptibility, and it has been suggested that the most potent antibiotics should be used in cases of peritonitis, instead of the most commonly used antibiotics (24). Empiric broadspectrum antibiotic regimen against a mixture of Gramnegative, Gram positive and anaerobic bacteria should be administered, if possible, after the peritoneal fluid has been collected. According to WSES guidelines for perforated and bleeding peptic ulcers, in patients with PUP, a short-course (3–5 days) of antibiotic therapy is recommended (2C) (25).
There is a controversy on the empirical administration of the anti-fungal agents (AF) in PPU. In most cases, fungal infections are present for surgical patients, especially intra-abdominal abscesses. The routine use of empiric AF is not sustained by the literature (26). Barmparas et al., in a study of 554 patients with PPU, found that empiric use of AF presented no clinical advantage in preventing infections, even those due to Candida spp., thus its administration was unnecessary (27). Nevertheless, Lee et al., in a study on 62 patients with PPU, found that 37.1% of patients presented positive culture to Candida spp. This study recommended that AF agents should be administered in all cases of fungal contamination to lower the mortality rate and shorten hospital stay (28). Shan et al., in a taiwanese study on 145 patients with PPU, observed that 43 % patients presented a positive fungal infection and Candida spp. was the most ordinary pathogen isolated from peritoneal fluid and wound cultures. This study recommended low-dose amphotericin B for critically ill surgical patients with intraperitoneal infection (29).
Strenghs and limitations
One of the strengths of this study represents a well exhaustive description of the bacteria isolated in the peritonitis secondary to PPU. However, one of its limitations was a modest number of patients suitable for analysis, mainly due to the reduced prevalence of this clinical scenario compared to other entities such as colonic or appendicular peritonitis. Otherwise, there was a limitation during follow-up because of a reduced number of postoperative upper endoscopies, which Surgery, Gastroenterology and Oncology, 27 (2), 2022 119 Bacteriology in Perforated Peptic Ulcer prevented to analyse the true incidence of H. pylori as the aetiology of the PUD.
CONCLUSION
Candida spp., Klebsiella spp. and Enterobacter were the most common organisms isolated in the microbiological samples of perforated peptic-ulcer peritonitis, with an overall resistance to antibiotics rate of a 17.5 %. The Charlson Comorbidity Index was an independent predictor for bacterial infection, while no factors could be identified as predictors of fungal infection.
Ethical approval and Consent to participate
The study was approved by the Ethical Committee of Saint Pierre University Hospital. The authors confirm that all research was performed in accordance with relevant guidelines/regulations, and the consent was obtained from all participants and/or their legal guardians.
Availability of data and materials
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
Competing interest
No competing interests exist for any of the authors.
Authors contributions
Sorin Cimpean: Conceptualization, Methodology, Software. Alberto Gonzalez Barranquero: Data curation, Writing - Original draft preparation. Benjamin Cadiere: Writing - Reviewing and Editing. Guy Bernard Cadiere: Supervision, Validation.
Funding statement
The authors received no financial support for the research, authorship, and/or publication of this article.
Acknowledgments
I thank to my colleagues for their expertise and assistance throughout all aspects of our study and for their help in writing the manuscript, in particular to the Professor G.B. Cadiere for guidance and support.
REFERENCES
1. Søreide K. Current insight into pathophysiology of gastroduodenal ulcers: Why do only some ulcers perforate? J Trauma Acute Care Surg. 2016 ;80(6):1045-8.
2. Søreide K, Thorsen K, Harrison EM, Bingener J, Møller MH, Ohene- Yeboah M, et al. Perforated peptic ulcer. Lancet. 2015;386(10000): 1288-98.
3. Dadfar A, Edna T-H. Epidemiology of perforating peptic ulcer: A population-based retrospective study over 40 years. World J Gastroenterol. 2020;26(35):5302-5313.
4. Thorsen K. Epidemiology of perforated peptic ulcer: Age - and gender-adjusted analysis of incidence and mortality. World J Gastroenterol. 2013;19(3):347-54.
5. Chung KT, Shelat VG. Perforated peptic ulcer - an update. World J Gastrointest Surg. 2017;9(1):1-12.
6. Malik S, Singh A, Sidhu DS, Nagpal N, Sharma D. A prospective study to assess clinical profile and golden period for operative intervention in patients with perforation peritonitis. Int Surg J. 2018;5(4):1492.
7. Kishore R. VR, Ashwin K, Vasanth Kumar SB. An observational study on peritoneal fluid bacteriology in cases of gastrointestinal perforations, antibiotic management and outcome in tertiary care center. Int Surg J. 2020;7(2):385.
8. Punamiya AR, Chougule PG, Ahuja BR, Singh V, Mohite ST. Commonest Organisms and Antibiotic Sensitivity in Peritonitis Due To Duodenal Ulcer Perforation in Krishna Hospital, Karad. Int J. Health Sciences. 2014;(8):5.
9. Yawar B, Marzouk AM, Ali H, Ghorab TM, Asim A, Bahli Z, et al. Seasonal Variation of Presentation of Perforated Peptic Ulcer Disease: An Overview of Patient Demographics, Management and Outcomes. Cureus. 2021;13(11):e19618.
10. Bekele A, Zemenfes D, Kassa S, Deneke A, Taye M, Wondimu S. Patterns and Seasonal Variations of Perforated Peptic Ulcer Disease: Experience from Ethiopia. Ann Afr Surg. 2018 DOI: 10.4314/aas.v14i2.7
11. Kavitt RT, Lipowska AM, Anyane-Yeboa A, Gralnek IM. Diagnosis and Treatment of Peptic Ulcer Disease. Am J Med. 2019;132(4): 447-456.
12. Ahmed M, Mansoor T, Rab AZ, Rizvi SAA. Risk factors influencing postoperative outcome in patients with perforated peptic ulcer: a prospective cohort study. Eur J Trauma Emerg Surg. 2022;48(1): 81-86.
13. Rohit DK, Verma RS, Pandey G. Clinical study and management of peritonitis secondary to perforated peptic ulcer. Int Surg J. 2017; 4(8):2721.
14. Tan S, Wu G, Zhuang Q, Xi Q, Meng Q, Jiang Y, et al. Laparoscopic versus open repair for perforated peptic ulcer: A meta-analysis of randomized controlled trials. Int J Surg. 2016;33 Pt A:124-32.
15. Asanasak P. The case series of peritonitis due to perforated peptic ulcer: How does conservative management play role? Int J Surg Case Rep. 2019;58:74-76.
16. Grotelüschen R, Heidelmann LM, Lütgehetmann M, Melling N, Reeh M, Ghadban T, et al. Antibiotic sensitivity in correlation to the origin of secondary peritonitis: a single center analysis. Sci Rep. 2020; 10(1):18588.
17. Gowda DB, Kadambari D, Vijayakumar C, Elamurugan T P, Jagdish S. A clinico-microbiological profile in patients with perforated peptic ulcer with special reference to anaerobic organisms: a descriptive study. Int Surg J. 2017;4(1):125-130.
18. ALwahed IA. Bacteroides spp. isolation from pus or peritoneal fluid in patients with perforated peptic ulcer. 2008;4(5):190-198.
19. Brook I. Microbiology and Management of Abdominal Infections. Dig Dis Sci. 2008;53(10):2585-91.
20. Ermetov A. Microbiologic structure of diffuse peritonitis. Medical and Health Science Journal, MHSJ. 2011;7:111-8.
21. Bhosale S, Gujar N, Chougule P. Long-term results of simple closure of perforated duodenal ulcer in the era of proton pump inhibitor and anti-H. Pylori. Therapy. 14 janv 2022
22. Lohith P, Jindal RK, Ghuliani D, Rajshekar P. The anatomical site of perforation peritonitis and their microbiological profile: a crosssectional study. Int Surg J. 26 mars 2020;7(4):1251.
23. Tayal R, De A, Sundaravadanan S, Kumar M, Baveja S. Role of Culture in Cases of Perforated Peptic Ulcers Due To Helicobacter Pylori. 2015;3(3):4.
24. Sudhaharan S, Kanne P, Vemu L, Chavali P, Desmukha SR. Bacteriological profile of intra-abdominal infections in a tertiary care hospital. Iran J Microbiol 2018;10(4):208-214.
25. Tarasconi A, Coccolini F, Biffl WL, Tomasoni M, Ansaloni L, Picetti E, et al. Perforated and bleeding peptic ulcer: WSES guidelines. World J Emerg Surg. 2020;15(1):64.
26. Huston JM, Kreiner L, Ho VP, Sanders JM, Duane TM. Role of Empiric Anti-Fungal Therapy in the Treatment of Perforated Peptic Ulcer Disease: Review of the Evidence and Future Directions. Surg Infect (Larchmt). 2019;20(8):593-600.
27. Barmparas G, Alhaj Saleh A, Huang R, Eaton BC, Bruns BR, Raines A, et al. Empiric antifungals do not decrease the risk for organ space infection in patients with perforated peptic ulcer. Trauma Surg Acute Care Open. mai 2021;6(1):e000662.
28. Lee S-C, Fung C-P, Chen H-Y, Li C-T, Jwo S-C, Hung Y-B, et al. Candida peritonitis due to peptic ulcer perforation: incidence rate, risk factors, prognosis and susceptibility to fluconazole and amphotericin B. Diagn Microbiol Infect Dis. 2002;44(1):23-7.
29. Shan Y-S, Hsu H-P, Hsieh Y-H, Sy ED, Lee J-C, Lin P-W. Significance of intraoperative peritoneal culture of fungus in perforated peptic ulcer. Br J Surg. 2003;90(10):1215-9.
Full Text Sources:
Abstract:
Views: 2584

Watch Video Articles
For Authors
Journal Subscriptions

Current Issue
Dec 2025
Supplements
Instructions for authors
Online submission
Contact
ISSN: 2559 - 723X (print)
e-ISSN: 2601 - 1700 (online)
ISSN-L: 2559 - 723X
Journal Abbreviation: Surg. Gastroenterol. Oncol.
Surgery, Gastroenterology and Oncology (SGO) is indexed in:
e-ISSN: 2601 - 1700 (online)
ISSN-L: 2559 - 723X
Journal Abbreviation: Surg. Gastroenterol. Oncol.
Surgery, Gastroenterology and Oncology (SGO) is indexed in:
- SCOPUS
- EBSCO
- DOI/Crossref
- Google Scholar
- SCImago
- Harvard Library
- Open Academic Journals Index (OAJI)
Open Access Statement
Surgery, Gastroenterology and Oncology (SGO) is an open-access, peer-reviewed online journal published by Celsius Publishing House. The journal allows readers to read, download, copy, distribute, print, search, or link to the full text of its articles.
Surgery, Gastroenterology and Oncology (SGO) is an open-access, peer-reviewed online journal published by Celsius Publishing House. The journal allows readers to read, download, copy, distribute, print, search, or link to the full text of its articles.
Journal Metrics
Time to first editorial decision: 25 days
Rejection rate: 61%
CiteScore: 0.2
Time to first editorial decision: 25 days
Rejection rate: 61%
CiteScore: 0.2

Meetings and Courses in 2025
Meetings and Courses in 2024
Meetings and Courses in 2023
Meetings and Courses in 2022
Meetings and Courses in 2021
Meetings and Courses in 2020
Meetings and Courses in 2019
Verona expert meeting 2019

Surgery, Gastroenterology and Oncology applies the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits readers to copy and redistribute the material in any medium or format, remix, adapt, build upon the published works non-commercially, and license the derivative works on different terms, provided the original material is properly cited and the use is non-commercial. Please see: https://creativecommons.org/licenses/by-nc/4.0/
Publisher’s Note:
The opinions, statements, and data contained in article are solely those of the authors and not of Surgery, Gastroenterology and Oncology journal or the editors. Publisher and the editors disclaim responsibility for any damage resulting from any ideas, instructions, methods, or products referred to in the content.
 IASGO Society News
IASGO Society News