Surgery, Gastroenterology and Oncology

|
|
Introduction: Different types of soft tissue tumors affect the gastrointestinal tract. Among them are leiomyomas, which are relatively uncommon. They are commonly asymptomatic masses and are discovered incidentally on radiological scans. Pathological examination of the tumors is the mainstay of the diagnosis. Surgery remains the cornerstone of management.
Case presentation: We present a 41-year-old female patient who was referred to our surgical department with a radiologically discovered pelviabdominal swelling, with vague symptoms mimicking uterine fibroid that was proved to be colonic leiomyoma after surgery and histopathological examination.
Conclusion: Colonic leiomyomas are extremely rare and fairly benign. Surgical resection is the appropriate and only treatment option.
Introduction
Leiomyomas are benign mesenchymal tumors commonly originating from the female genital tract and are rarely reported in extrauterine locations (1). True gastrointestinal leiomyomas commonly occur in the esophagus or stomach, while colorectal leiomyomas are extremely rare and account for only 3% of all gastrointestinal tumors (2). The most commonly affected parts are the transverse and sigmoid colon (3). They can present either as a sessile mass or a pedunculated polyp and as intraluminal, extraluminal, or intramural (4,5). Colonic leiomyomas are usually asymptomatic and discovered incidentally. Furthermore, radiologic findings are unspecific, but preoperative biopsy could help determine the diagnosis. Interestingly, leiomyomas could be large, and surgery would be warranted to confirm the diagnosis and rule out malignancy (6). We hereby report a rare case of large extraluminal ascending colon leiomyoma that mimicked a uterine fibroid radiologically.
Case presentation
A 41-year-old female patient was referred to our surgical oncology clinic with a sonographically detected pelviabdominal swelling. She had a history of vague abdominal symptoms and dysfunctional uterine bleeding. She had no significant history apart from 2 Caesarian sections. Clinical examination revealed a palpable mass in the right iliac region. The mass was about 10 x 10 cm, firm in consistency, mobile and non-tender. The lower border of the swelling could not be palpated. The overlying skin was normal except for a Pfannenstiel incision.
Abdominal ultrasonography revealed a well-defined rounded soft tissue mass iso to hypoechoic, measuring 9.5 x 9 cm with internal vascularity in the right lumbar and iliac region, related to the pelvic colon. Post-contrast Computerized tomography scan of the abdomen and pelvis showed a right adnexal isodense well-defined soft tissue mass 9 x 9.5 cm, which is seen closely related to the surrounding bowel loops and inseparable from the right broad ligament region and the right uterine side raising the possibility of subserous versus broad ligament fibroid.
A post-contrast MRI of the pelvis showed a normal-sized uterus with normal zonal anatomy, in addition to a large mass with heterogenous intermediate to high signal changes. Coronal sections showed the mass separable from the right adnexal region and the right ovary. The sagittal view raised the suspicion that the mass could be originating from an intestinal loop. Moreover, T1 MRI images revealed homogenous enhancement of the mass equivalent to that of the bowel loop fig. 1.
Figure 1 - (a) Coronal CT image showing right adnexal isodense well-defined soft tissue mass 9 x 9.5 cm (arrowed), which is seen closely related to the related bowel loops and inseparable from the right broad ligament region and the right uterine side. (b) Axial T2 MRI WI shows the mass with heterogenous intermediate to high signal changes (arrowed). (c) Coronal T2 WI shows the mass (arrowed) separable from the right adnexal region and the right ovary (curved arrow). (d) Sagittal T2 WI shows the mass (arrowed) with the origin arising from the loop (Dash-arrowed). (e&f) Coronal and sagittal FAT-SAT post-contrast T1 MRI showing the mass with homogenous enhancement equivalent to the bowel loop (arrowed) with regional LN (head arrow).

Our patient was prepared for surgery after a multidisciplinary team discussion. Laparoscopic assessment revealed a bulky uterus with a huge mass of about 12 x 10 cm in the right iliac fossa. The mass was adherent to the psoas muscle and the lateral abdominal wall, with infiltrated ileocecal junction and lower cecum. The trial for mass dissection from the lateral abdominal wall failed, thus conversion to laparotomy was decided.
The mass was dissected from the lateral abdominal wall. The right round ligament was opened to facilitate dissection, and then mass excision en bloc with segmental colectomy just below the hepatic flexure, finishing with an end-to-end hand-sewn ileocolic anastomosis. Total abdominal hysterectomy with right salpingo-oophrectomy was done and preservation of the left ovary fig. 2. The patient received one unit of packed RBCs. The patient experienced an uneventful smooth postoperative course and was discharged on the 5th postoperative day.
Figure 2 - Postoperative specimen showing the resected colonic leiomyoma.
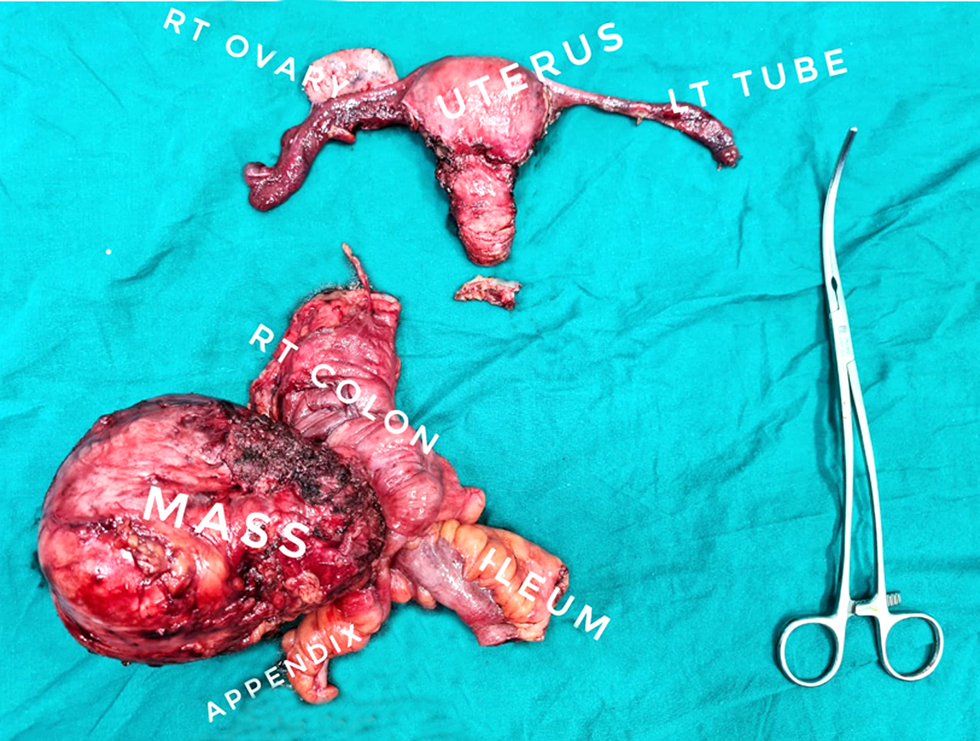
Post-operative gross pathologic examination found the mass arising from the colonic wall with intact mucosa. The mass measured 10 x 10 x 9 cm, and on dissection, it showed well-circumscribed greyish-white with firm and whirly construction. Microscopic examination revealed benign tumoral proliferation of spindle cells arranged in interlacing fascicles. The cells showed bland cigar nuclei with tapering ends. Hemanigiopericytomatous blood vessels could also be seen. The appendix and both safety margins were free from any tumor tissue. Free dissected 10 lymph nodes from tumor tissue exhibited reactive follicular hyperplasia. Immunohistochemistry was crucial for identifying and therefore diagnosing such a case. Smooth Muscle Actin, Desmin, Caldesmon, and S100 all showed focal positive reactions. However, both CD34 and CD117 were negative fig. 3. On the other hand, the Ki67 reaction showed a low proliferation index (2%). Histopathological evaluation of the uterus, Fallopian tubes, and the right ovary revealed simple endometrial hyperplasia and chronic non-specific cervicitis with a compatible to-age ovary.
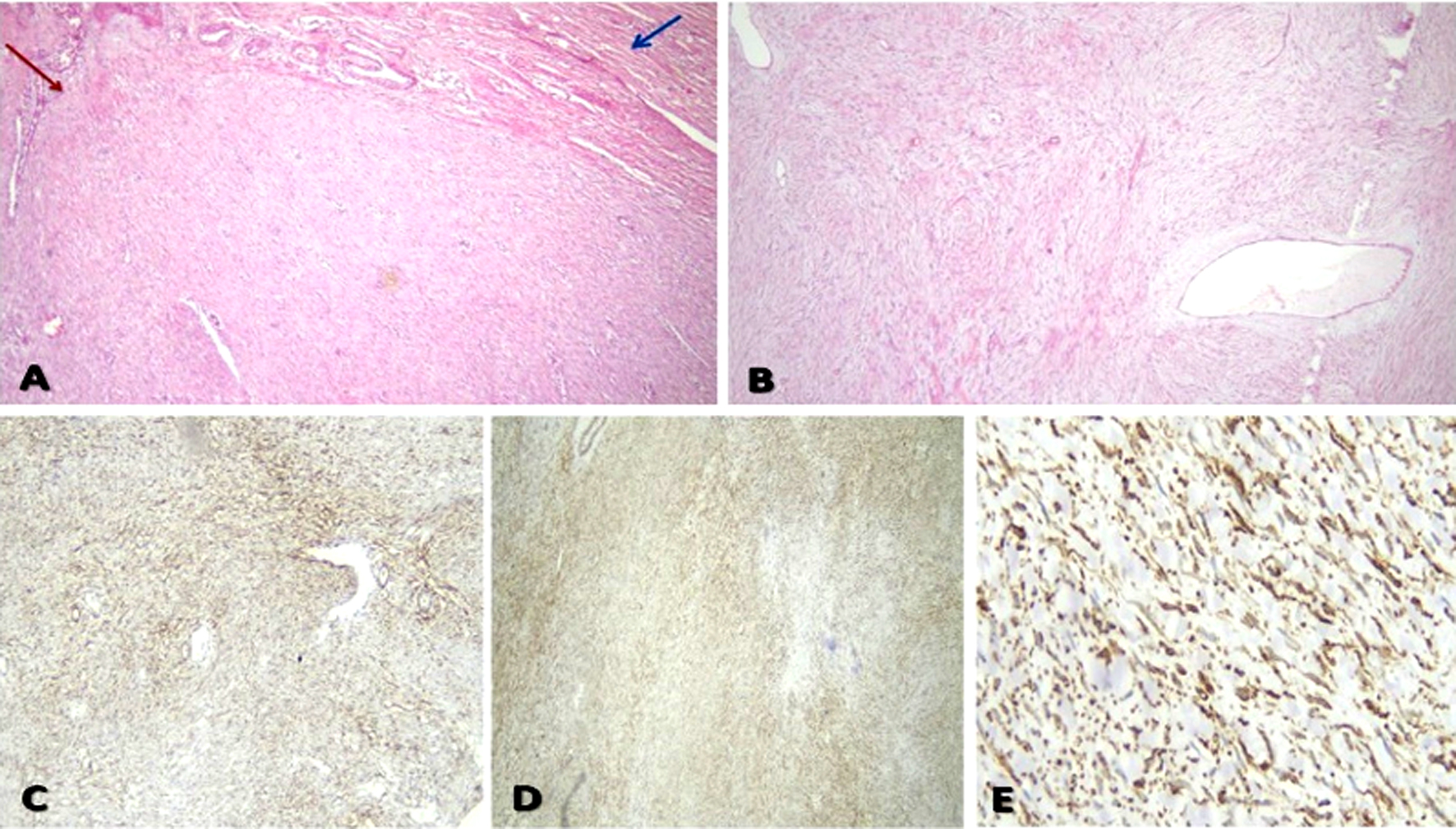
Figure 3 - Postoperative histopathological examination of the specimen (a) Spindle cell proliferation (red arrow) arising from the muscularis propria of the colonic wall (blue arrow) (H&E 200x) (b) Tumor was formed of bland-looking spindle cells arranged in whorls and long fascicles. No detected pleomorphism, abnormal mitosis, or necrosis (H&E 200x) (c) Immunohistochemical staining for alpha-smooth muscle actin (a-SMA) showed strong diffuse cytoplasmic positivity (200x) (d) Strong diffuse cytoplasmic positivity of tumor cells for Desmin (200x) (e) Tumor cells showed strong diffuse positive cytoplasmic staining for H-Caldesmon (400x).
Discussion
Smooth intestinal tumors are the second most common mesenchymal tumors affecting the gastro-intestinal tract after Gastrointestinal stromal tumors (GIST) (7). Leiomyomas of the colon are extremely uncommon, accounting for 3% of GIT leiomyomas and 1% of all GIT tumors. They arise from either the muscularis mucosae or the muscularis propria. Colonic leiomyomas were commonly reported to involve the left side than the right side of the colon where most cases were reported in the sigmoid or the transverse colon. Colonic leiomyomas have a male predominance with a peak age of more than 60 (8). Since colonic leiomyomas can present as a pedunculated polyp and their proximity to pelvic organs, they can be mistaken as an adnexal fibroid early in the diagnostic journey (2). In this article, we report a 41-year-old female patient diagnosed with a right-sided large colonic leiomyoma that mimicked a uterine leiomyoma.
The clinical presentation of colonic leiomyomas varies according to their site, size, and direction of growth (9). They are most commonly asymptomatic, although manifestations such as abdominal pain, perforation, and rarely hemorrhage were documented. Other manifestations might include microcytic hypo-chromic anemia with a positive fecal occult blood test, a change in bowel habits, or a palpable pelviabdominal mass (10). In this report, the patient presented with vague abdominal symptoms, and the mass was initially discovered by ultrasonography.
Imaging findings are not specific enough to diagnose colonic leiomyomas. Ultrasonography can be used initially but is usually inconclusive. Computerized Tomography could reveal heterogeneity, calcifications, and necrosis. It is worth mentioning that accurate diagnosis necessitates a combination of multiple imaging modalities including CT, MRI, and endoscopic ultrasonography(8). Radiologic evaluation of the mass in our patient was a matter of conflict, where CT suggested uterine leiomyoma while MRI raised the possibility of intestinal origin.
Interestingly, previous case reports and studies evaluated the prevalence of large extraluminal leiomyomas. In their study, Hatch et al. found 5 cases out of 25 descending colon leiomyoma cases were larger than 15 cm (11). Moreover, Sayer et al., and Sagnotta et al., reported 18 cm rectal and descending colon leiomyomas respectively (3,12). Khandhasamy et al. described complicated large leiomyoma where they reported a right flank subcutaneous abscess due to complicated 9cm cecal leiomyoma (8). To the best of our knowledge, the present case is one of the few large extraluminal right-sided colonic leiomyomas that were previously reported.
Taking into account the fact that gastrointestinal stromal tumors (GIST) are by far the commonest gastrointestinal tumors and that they might mimic leiomyomas, especially under light microscopy,
differentiating between the two is a vital step in both diagnosis and management. Microscopically, leiomyomas are well-differentiated, well-circumscribed smooth muscle mass with interlacing spindle-shaped myofibers with abundant eosinophilic cytoplasm and oval pale nuclei. Absence of atypia, mitotic activity, and any other anaplastic features. As per immunohistochemistry, all leiomyomas show diffuse positivity for αSMA, Caldesmon, and Desmin and strong negativity to CD117. Some of them had CD34 weakly positive cells. On the other hand, GISTs are strongly positive to CD117 and DOG-1, in addition, they might manifest positivity to CD34(13). In our patient, Immunohistochemistry confirmed the diagnosis of leiomyoma where Smooth Muscle Actin, Desmin, Caldesmon, and S100 all showed focal positive reaction while both CD34 and CD117 were negative. On the other hand, the Ki67 reaction showed a low proliferation index (2%).
There are different techniques for the management of colonic leiomyomas either submucosal excision in case of small intramural lesions to subtotal colectomy in larger ones. Interestingly, surgical resection is the valid option for most leiomyomas. The extent of resection is based on the nature, size, and the site of the tumor. Excisional biopsy could be an acceptable option for small benign lesions while colectomy might be necessary to ensure complete mass resection (3). Post-operative care remains only supportive, as the risk of recurrence is minimal unless immunohistochemistry shows features of anaplasia, the latter would demand an oncology referral (4).
Conclusion
Colonic leiomyoma is an extremely rare benign tumor that can mimic various smooth muscle tumors, especially adnexal masses posing a diagnostic challenge. Histoimmunopathology is the key step of diagnosis. Surgical resection remains the cornerstone of management followed by an excellent prognosis with a rare chance of recurrence.
Conflict of interests
The authors disclose no conflict of interests.
Human rights
This report was conducted in accordance with the 1964 Declaration of Helsinki.
Informed consent
Written informed consent was obtained from the patient.
Funding
No funding was received.
Authors' contribution
MZ: Manuscript writing and formatting, KB: case presentation and manuscript revision, MS: Manuscript writing, SA: Manuscript revision and revision, NM: pathology interpretation, ME: radiology interpretation, OB: manuscript revision and formatting, AH: Manuscript writing and revision. All authors revised and approved the manuscript.
References
1. Wang X, Ren J, Chen G, Xu S. Giant abdominal leiomyoma of male: a case report and literature reviews. Int J Clin Exp Pathol. 2015; 8(7):8532-5.
2. Pellegrini JR, Russe JR, Munshi R, Smoller R, Khan N. Leiomyoma in the transverse colon with resection. Cureus. 2021;13(6):e15535.
3. Sagnotta A, Sparagna A, Uccini S, Mercantini P. Giant extraluminal leiomyoma of the colon: rare cause of symptomatic pelvic mass. Int Surg. 2015;100(5):805-8.
4. Badipatla KR, Kamireddy C, Niazi M, Nayudu SK. Cecal leiomyoma: can we attempt endoscopic resection? Gastroenterology Res. 2016; 9(6):105-107.
5. Choi HH, Cho Y-S, Choi SK, Kim H-K, Kim SS, Chae H-S, et al. Clinical outcomes of endoscopic removal in patients with colorectal polypoid leiomyomas. Korean J Gastroenterol. 2016;68(4):179-85.
6. Dreifuss NH, Mangano A, Aguiluz G, Mehta V, Koppe S, Berkelhammer C, et al. Colonic leiomyoma mimicking a liver tumor: an unusual diagnosis after en-block robotic resection. J Surg case reports. 2021;2021(9):rjab418.
7. Alpert L, Al-Sabti R, Graham RP, Pai RK, Gonzalez RS, Zhang X, et al. Smooth muscle tumors of the gastrointestinal tract: an analysis of prognostic features in 407 cases. Mod Pathol. 2020; 33(7):1410-9.
8. Kandhasamy SC, Sangwan A, Sahoo AK, Gunasekaran G, Sahani N, Raju TR, et al. An uncommon presentation of leiomyoma cecum as a subcutaneous abscess of the right flank. Cureus. 2018; 10(10):e3432.
9. Abruzzo A, Spinell G, Martorana A. Management of leiomyoma of the transverse colon: case report. Eur J Oncol. 2013;18(18):211-4.
10. Forlemu AN, Chuang K-Y. Sigmoid leiomyoma: an uncommon occurrence. Cureus. 2020;12(4):e7567.
11. Hatch KF, Blanchard DK, Hatch III GF, Wertheimer-Hatch L, Davis GB, Foster Jr RS, et al. Tumors of the appendix and colon. World J Surg. 2000;24(4):430–6.
12. Sayer RA, Amundsen CL. Giant pelvic retroperitoneal leiomyoma arising from the rectal wall. Obstet Gynecol. 2003;101(5):1132–4.
13. Janevska V, Qerimi A, Basheska N, Stojkova E, Janevski V, Jovanovic R, et al. Superficial leiomyomas of the gastrointestinal tract with interstitial cells of Cajal. Int J Clin Exp Pathol. 2015;8(12):15977-84.
Full Text Sources:
Abstract:
Views: 5043

For Authors
Journal Subscriptions

Dec 2025
Supplements
Instructions for authors
Online submission
Contact
e-ISSN: 2601 - 1700 (online)
ISSN-L: 2559 - 723X
Journal Abbreviation: Surg. Gastroenterol. Oncol.
Surgery, Gastroenterology and Oncology (SGO) is indexed in:
- SCOPUS
- EBSCO
- DOI/Crossref
- Google Scholar
- SCImago
- Harvard Library
- Open Academic Journals Index (OAJI)
Surgery, Gastroenterology and Oncology (SGO) is an open-access, peer-reviewed online journal published by Celsius Publishing House. The journal allows readers to read, download, copy, distribute, print, search, or link to the full text of its articles.
Time to first editorial decision: 25 days
Rejection rate: 61%
CiteScore: 0.2

Meetings and Courses in 2025
Meetings and Courses in 2024
Meetings and Courses in 2023
Meetings and Courses in 2022
Meetings and Courses in 2021
Meetings and Courses in 2020
Meetings and Courses in 2019
Verona expert meeting 2019

Surgery, Gastroenterology and Oncology applies the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits readers to copy and redistribute the material in any medium or format, remix, adapt, build upon the published works non-commercially, and license the derivative works on different terms, provided the original material is properly cited and the use is non-commercial. Please see: https://creativecommons.org/licenses/by-nc/4.0/
Publisher’s Note:
The opinions, statements, and data contained in article are solely those of the authors and not of Surgery, Gastroenterology and Oncology journal or the editors. Publisher and the editors disclaim responsibility for any damage resulting from any ideas, instructions, methods, or products referred to in the content.
 IASGO Society News
IASGO Society News