Surgery, Gastroenterology and Oncology
Official journal of the International Association of Surgeons, Gastroenterologists and Oncologists

|
|
Literature Review of Oropahyngeal Cancer Patients Assessed with EORTC QLQ-30 EORTC H&N-35#
Background: Oropharyngeal cancer is the most common type of head and neck cancers,
with a 5-years survival of 64.7%. In the last 40 years risk factors and etiology changed, from
the incidence associated mostly with tobacco and alcohol to HPV infection in 70% of the
cases. Treatments that are standard of care for OPSCC include chemotherapy, radiotherapy
or combination of surgery and radiotherapy have a high chronic treatment-related toxicity
and functional loss. These therapies have significant impact on the quality of life (QOL) of
survivors of oropharyngeal squamous cell carcinoma (OPSCC).
Objectives: The objective of this study is to review literature on 1-year evolution of quality of life of patients treated for OPSCC with standard of care.
Methods: A comprehensive search of the literature of treated OPSCC patients assessed with EORTC QLQ-30 EORTC H&N-35 at pre-treatment 12-month post treatment.
Results: The first study showed that standard of care treatment produces chronic side effects, such as xerostomia, poor oral and dental health, dysphagia, feeding tube dependency in, and other fibrotic changes likely caused by radiotherapy or combination of surgery and radiotherapy.
Conclusions: Standard of care treatment produces chronic side effects, such as xerostomia, poor oral and dental health, dysphagia, feeding tube dependency in, and other fibrotic changes.
INTRODUCTION
In 2017 the American Cancer Society reported 49670 news cases of and 9700 deaths from head and neck cancers, respectively, with a 5-year overall survival of 64.7% (1-3). While the proportion of oropharyngeal squamous cell carcinoma was approximately 20% of Head and Neck cancers in 1980s in the US, it currently represents 70% (4,5). The current standard of care for advanced stages (III and IV) OPSCC- including HPV related OPSCC- includes high doses of chemotherapy (usually cisplatin based) and radiotherapy. The current standard of care treatment for patients with locally advanced OPSCC is known to be highly toxic, and these approaches leave survivors with significant and lifelong morbidity (6-11).
The short-term and long-term sequelae of OPSCC treatment are known to impact survivors quality of life. These include loss of salivary function, dry mouth, sticky saliva, dental loss, poor oral health, dysphagia, feeding tube dependency, neck muscle dystonia, fibrotic loss of lower cranial nerve function, pharyngeal and laryngeal stenosis, soft tissue necrosis, chronic mucosal ulcerations, chronic feeding tube dependency, muscle atrophy, and osteoradionecrosis. Social, economic, and emotional factors are permanently changed after cancer is successfully cured. Even minimal damage to swallowing, talking, eating and respiration diminish quality of life of the patients.
Quality of life questionnaires evaluate multiple dimensions of life that are of importance to patients. One of the most used questionnaires in more than 3000 studies since 1993, European Organisation for Research and Treatment of Cancer (EORTC) Quality of Life Questionnaire (QLQ-C30) is also available in 130 languages.
It has 30 questions in 15 subscales relevant to people with cancer: five distinct aspects of functioning (physical, role, emotional, cognitive, social), eight symptoms (fatigue, nausea/vomiting, pain, dyspnoea, insomnia, appetite loss, constipation, diarrhoea), financial difficulties, and global health/quality of life. Therefore, the current review was undertaken to provide a summary of the 1-year evolution of quality of life of patients treated for oropharyngeal cancer. The review focuses on the studies that used the EORTC QLQ30 and H&N35 to assess quality of life at diagnosis and at 12-month post-treatment, to understand how the current standard of care treatment for OPSCC impacts patients in the year following treatment.
MATERIALS AND METHODS
Search strategy
With the help of a librarian, we performed an extensive literature search through Medline and Cochrane database for studies from 1999 to December 2019. We used the following terms "oropharynx", "oropharyngeal"," cancer"," neoplasm", "tumour"," quality of life" with databased- specific coding and combinations. Mesh headings were used in different combinations. Inclusion criteria were analytical studies (case-control, cross-sectional, cohort, randomized control trials, qualitative studies, systematic reviews, and meta-analyses) that used the EORTC assessment tool at diagnosis and 12 months after treatment in OPSCC patients. Articles published in English were included with no intention of searching unpublished literature. Additional limits were set for rejecting results that involved animals or children. HPV status could not be used as a search criterion because there was no clear separation in any study.
RESULTS
In total, 206 papers were identified. After reviewing initially the titles followed by the abstracts of these papers, only four met the inclusion criteria. We extracted the following data from the four studies: author and year of publication, country, sample size, age, sex, cancer stage, treatment, and quality of life at diagnosis and 12-months post-treatment.
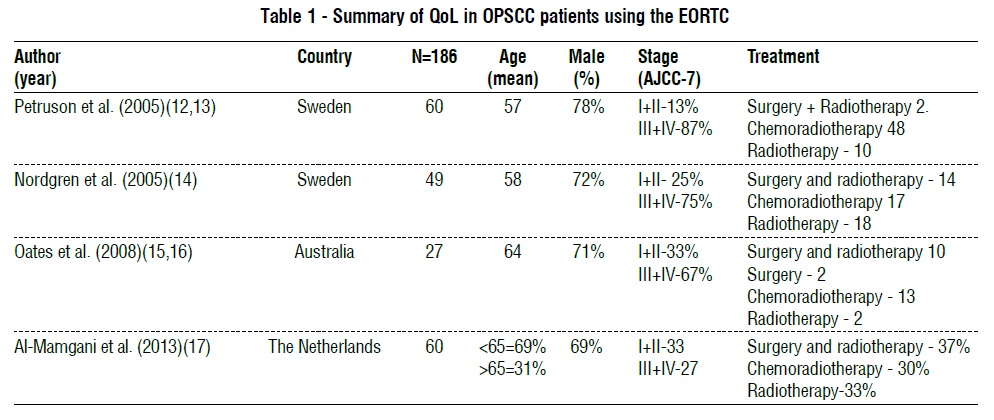
As described in table 1, 186 patients from three countries were included in this review. Mean age ranged between 57 and 64 in 3 studies; in the fourth study, 69% of patients were under 65 years of age. All studies had more male than female patients. Treatment included surgery alone, surgery followed by radiotherapy, surgery and chemoradiotherapy or radiotherapy alone. Only one study described the presence of a PEG at 12-month time-point. There was no information on the HPV status of the tumour.
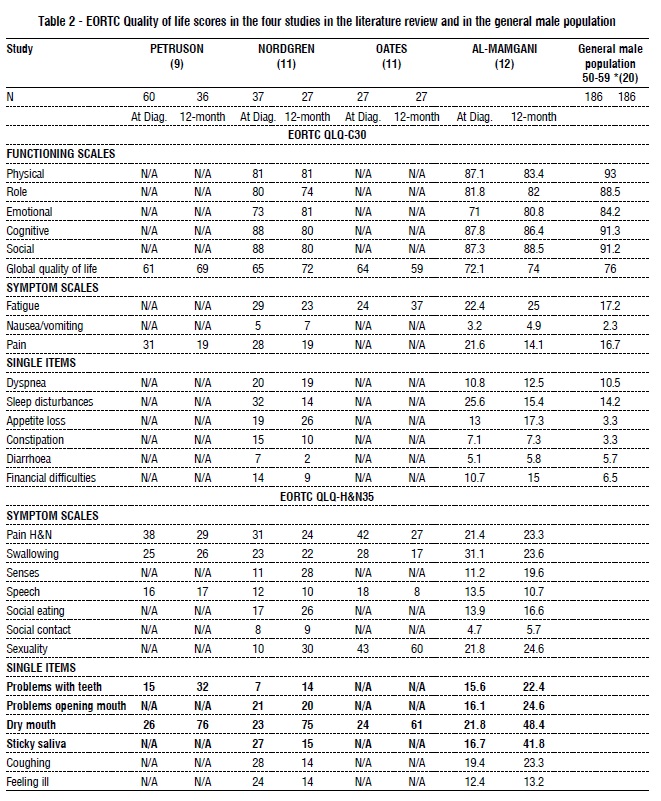
Table 2 shows the EORTC OLQ-C 30 values at diagnosis, and 12-months post-treatment of the four included studies included as well as those of the general male population aged 50–59.
The Global quality of life improved in all the studies from diagnosis to 12-month post-treatment General population scores were comparable to scores at diagnosis of patients in all four studies. Some studies reported degradation of quality of life in different scales. At 12 months, post-treatment Nordgren et al (14) reported deterioration in social functioning, an increase in appetite loss, senses problems, social eating problems, sexuality problems, as well as remarkable worsening of problems with teeth and dry mouth compared to diagnosis. Petruson et al (12) reported an increase in problems with teeth and dry mouth at 12-month post-treatment compared to diagnosis. Al-Mamgani et al (17) reported worse scores in appetite, pain related to head and neck, senses problems, social eating, sexuality, and remarkable worsening in problems with teeth, problems opening the mouth, dry mouth and sticky saliva. Oates (13) reported increases in fatigue and dry mouth at 12-month post-treatment compared with values at diagnosis.
Oates et al was the only study that determined the presence of a PEG at 12 months, which was reported in 25% of patients.
DISCUSSION AND CONCLUSIONS
In summary, this brief literature review showed that patients with OPSCC treated with standard of care- that includes surgery followed by RT/CRT, or chemotherapy and radiotherapy in different combinations- have a QoL at 12-month post-treatment that is lower compared with pre-treatment one. Most problems were related to long term side effects of radiotherapy, such as xerostomia, sticky saliva, trismus and problems with teeth.
Patients also reported lower role, functioning scores, and a possible cause for that is the presence of a PEG that impedes oral diet in all daily life situations. To date, and likely deterioration in other scales. For now, there are no data in the literature to assess the impact of HPV positivity on the quality of life of OPSCC patients following treatment as these studies included all OPC irrespective of HPV status, However, the treatment is the major determinant of the QOL and that being the same irrespective of HPV status, these results likely apply to HPV positive OPC. Given the prevalence of HPV positive oropharyngeal cancer, likely most patients in these studies were HPV positive OPC.
Acknowledgements
I thank Dr Maida Sewitch, Associate Professor, Department of Medicine, McGill University and Dr Nader Sadeghi for their input as supervisors of my masters degree. I also thank Ibtisam Mahmoud, Librarian McGill University Health Centre for helping me with the search of literature.
Conflict of interests
The authors declare no conflicts of interests.
Ethical approval
Ethical approval was not needed for this retrospective study.
REFERENCES
1. Timbang MR, Sim MW, Bewley AF, Farwell DG, Mantravadi A, Moore MG. HPV-related oropharyngeal cancer: a review on burden of the disease and opportunities for prevention and early detection. Hum Vaccin Immunother. 2019;15(7-8):1920-1928.
2. Bjordal K, de Graeff A, Fayers PM, Hammerlid E, van Pottelsberghe C, Curran D, et al. A 12 country field study of the EORTC QLQ-C30 (version 3.0) and the head and neck cancer specific module (EORTC QLQ-H&N35) in head and neck patients. EORTC Quality of Life Group. Eur J Cancer. 2000;36(14):1796-807.
3. McLaughlin-Drubin ME, K. Munger, Oncogenic activities of human papillomaviruses. Virus Res, 2009. 143(2):195-208.
4. Marur S, Burtness B. Oropharyngeal squamous cell carcinoma treatment: current standards and future directions. Curr Opin Oncol. 2014;26(3):252-8.
5. Ducatman BS. The Role of Human Papillomavirus in Oropharyngeal Squamous Cell Carcinoma. Arch Pathol Lab Med. 2018;142(6): 715-718.
6. Dörr W, Hendry JH. Consequential late effects in normal tissues. Radiother Oncol. 2001;61(3):223-31.
7. Rieger JM, Zalmanowitz JG, Wolfaardt JF. Functional outcomes after organ preservation treatment in head and neck cancer: a critical review of the literature. Int J Oral Maxillofac Surg. 2006;35(7): 581-7.
8. Fung K, Lyden TH, Lee J, Urba SG, Worden F, Eisbruch A, et al. Voice and swallowing outcomes of an organ-preservation trial for advanced laryngeal cancer. Int J Radiat Oncol Biol Phys. 2005; 63(5):1395-9.
9. Eisbruch A, Schwartz M, Rasch C, Vineberg K, Damen E, Van As CJ, et al. Dysphagia and aspiration after chemoradiotherapy for headand- neck cancer: which anatomic structures are affected and can they be spared by IMRT? Int J Radiat Oncol Biol Phys. 2004; 60(5):1425-39.
10. Nguyen NP, Smith HJ, Sallah S. Evaluation and management of swallowing dysfunction following chemoradiation for head and neck cancer. Curr Opin Otolaryngol Head Neck Surg. 2007;15(2):130-3.
11. Jensen K, Lambertsen K, Grau C. Late swallowing dysfunction and dysphagia after radiotherapy for pharynx cancer: frequency, intensity and correlation with dose and volume parameters. Radiother Oncol. 2007;85(1):74-82.
12. Petruson K, Mercke C, Lundberg LM, Silander E, Hammerlid E. Longitudinal evaluation of patients with cancer in the oral tongue, tonsils, or base of tongue--does interstitial radiation dose affect quality of life? Brachytherapy. 2005;4(4):271-7.
13. Oates J, Clark JR, Read J, Reeves N, Gao K, O'Brien CJ. Integration of prospective quality of life and nutritional assessment as routine components of multidisciplinary care of patients with head and neck cancer. ANZ J Surg. 2008;78(1-2):34-41.
14. Nordgren M, Jannert M, Boysen M, Ahlner-Elmqvist M, Silander E, Bjordal K, et al. Health-related quality of life in patients with pharyngeal carcinoma: a five-year follow-up. Head Neck. 2006; 28(4):339-49.
15. Oates J, Clark JR, Read J, Reeves N, Gao K, O'Brien CJ. Integration of prospective quality of life and nutritional assessment as routine components of multidisciplinary care of patients with head and neck cancer. ANZ J Surg. 2008;78(1-2):34-41.
16. Adnan A, Högmo A, Sjödin H, Gebre-Medhin M, Laurell G, Reizenstein J, et al. Health-related quality of life among tonsillar carcinoma patients in Sweden in relation to treatment and comparison with quality of life among the population. Head Neck. 2020;42(5):860-872.
Full Text Sources:
Abstract:
Views: 2285

Objectives: The objective of this study is to review literature on 1-year evolution of quality of life of patients treated for OPSCC with standard of care.
Methods: A comprehensive search of the literature of treated OPSCC patients assessed with EORTC QLQ-30 EORTC H&N-35 at pre-treatment 12-month post treatment.
Results: The first study showed that standard of care treatment produces chronic side effects, such as xerostomia, poor oral and dental health, dysphagia, feeding tube dependency in, and other fibrotic changes likely caused by radiotherapy or combination of surgery and radiotherapy.
Conclusions: Standard of care treatment produces chronic side effects, such as xerostomia, poor oral and dental health, dysphagia, feeding tube dependency in, and other fibrotic changes.
INTRODUCTION
In 2017 the American Cancer Society reported 49670 news cases of and 9700 deaths from head and neck cancers, respectively, with a 5-year overall survival of 64.7% (1-3). While the proportion of oropharyngeal squamous cell carcinoma was approximately 20% of Head and Neck cancers in 1980s in the US, it currently represents 70% (4,5). The current standard of care for advanced stages (III and IV) OPSCC- including HPV related OPSCC- includes high doses of chemotherapy (usually cisplatin based) and radiotherapy. The current standard of care treatment for patients with locally advanced OPSCC is known to be highly toxic, and these approaches leave survivors with significant and lifelong morbidity (6-11).
The short-term and long-term sequelae of OPSCC treatment are known to impact survivors quality of life. These include loss of salivary function, dry mouth, sticky saliva, dental loss, poor oral health, dysphagia, feeding tube dependency, neck muscle dystonia, fibrotic loss of lower cranial nerve function, pharyngeal and laryngeal stenosis, soft tissue necrosis, chronic mucosal ulcerations, chronic feeding tube dependency, muscle atrophy, and osteoradionecrosis. Social, economic, and emotional factors are permanently changed after cancer is successfully cured. Even minimal damage to swallowing, talking, eating and respiration diminish quality of life of the patients.
Quality of life questionnaires evaluate multiple dimensions of life that are of importance to patients. One of the most used questionnaires in more than 3000 studies since 1993, European Organisation for Research and Treatment of Cancer (EORTC) Quality of Life Questionnaire (QLQ-C30) is also available in 130 languages.
It has 30 questions in 15 subscales relevant to people with cancer: five distinct aspects of functioning (physical, role, emotional, cognitive, social), eight symptoms (fatigue, nausea/vomiting, pain, dyspnoea, insomnia, appetite loss, constipation, diarrhoea), financial difficulties, and global health/quality of life. Therefore, the current review was undertaken to provide a summary of the 1-year evolution of quality of life of patients treated for oropharyngeal cancer. The review focuses on the studies that used the EORTC QLQ30 and H&N35 to assess quality of life at diagnosis and at 12-month post-treatment, to understand how the current standard of care treatment for OPSCC impacts patients in the year following treatment.
MATERIALS AND METHODS
Search strategy
With the help of a librarian, we performed an extensive literature search through Medline and Cochrane database for studies from 1999 to December 2019. We used the following terms "oropharynx", "oropharyngeal"," cancer"," neoplasm", "tumour"," quality of life" with databased- specific coding and combinations. Mesh headings were used in different combinations. Inclusion criteria were analytical studies (case-control, cross-sectional, cohort, randomized control trials, qualitative studies, systematic reviews, and meta-analyses) that used the EORTC assessment tool at diagnosis and 12 months after treatment in OPSCC patients. Articles published in English were included with no intention of searching unpublished literature. Additional limits were set for rejecting results that involved animals or children. HPV status could not be used as a search criterion because there was no clear separation in any study.
RESULTS
In total, 206 papers were identified. After reviewing initially the titles followed by the abstracts of these papers, only four met the inclusion criteria. We extracted the following data from the four studies: author and year of publication, country, sample size, age, sex, cancer stage, treatment, and quality of life at diagnosis and 12-months post-treatment.

As described in table 1, 186 patients from three countries were included in this review. Mean age ranged between 57 and 64 in 3 studies; in the fourth study, 69% of patients were under 65 years of age. All studies had more male than female patients. Treatment included surgery alone, surgery followed by radiotherapy, surgery and chemoradiotherapy or radiotherapy alone. Only one study described the presence of a PEG at 12-month time-point. There was no information on the HPV status of the tumour.
Table 2 shows the EORTC OLQ-C 30 values at diagnosis, and 12-months post-treatment of the four included studies included as well as those of the general male population aged 50–59.
The Global quality of life improved in all the studies from diagnosis to 12-month post-treatment General population scores were comparable to scores at diagnosis of patients in all four studies. Some studies reported degradation of quality of life in different scales. At 12 months, post-treatment Nordgren et al (14) reported deterioration in social functioning, an increase in appetite loss, senses problems, social eating problems, sexuality problems, as well as remarkable worsening of problems with teeth and dry mouth compared to diagnosis. Petruson et al (12) reported an increase in problems with teeth and dry mouth at 12-month post-treatment compared to diagnosis. Al-Mamgani et al (17) reported worse scores in appetite, pain related to head and neck, senses problems, social eating, sexuality, and remarkable worsening in problems with teeth, problems opening the mouth, dry mouth and sticky saliva. Oates (13) reported increases in fatigue and dry mouth at 12-month post-treatment compared with values at diagnosis.

Oates et al was the only study that determined the presence of a PEG at 12 months, which was reported in 25% of patients.
DISCUSSION AND CONCLUSIONS
In summary, this brief literature review showed that patients with OPSCC treated with standard of care- that includes surgery followed by RT/CRT, or chemotherapy and radiotherapy in different combinations- have a QoL at 12-month post-treatment that is lower compared with pre-treatment one. Most problems were related to long term side effects of radiotherapy, such as xerostomia, sticky saliva, trismus and problems with teeth.
Patients also reported lower role, functioning scores, and a possible cause for that is the presence of a PEG that impedes oral diet in all daily life situations. To date, and likely deterioration in other scales. For now, there are no data in the literature to assess the impact of HPV positivity on the quality of life of OPSCC patients following treatment as these studies included all OPC irrespective of HPV status, However, the treatment is the major determinant of the QOL and that being the same irrespective of HPV status, these results likely apply to HPV positive OPC. Given the prevalence of HPV positive oropharyngeal cancer, likely most patients in these studies were HPV positive OPC.
Acknowledgements
I thank Dr Maida Sewitch, Associate Professor, Department of Medicine, McGill University and Dr Nader Sadeghi for their input as supervisors of my masters degree. I also thank Ibtisam Mahmoud, Librarian McGill University Health Centre for helping me with the search of literature.
Conflict of interests
The authors declare no conflicts of interests.
Ethical approval
Ethical approval was not needed for this retrospective study.
REFERENCES
1. Timbang MR, Sim MW, Bewley AF, Farwell DG, Mantravadi A, Moore MG. HPV-related oropharyngeal cancer: a review on burden of the disease and opportunities for prevention and early detection. Hum Vaccin Immunother. 2019;15(7-8):1920-1928.
2. Bjordal K, de Graeff A, Fayers PM, Hammerlid E, van Pottelsberghe C, Curran D, et al. A 12 country field study of the EORTC QLQ-C30 (version 3.0) and the head and neck cancer specific module (EORTC QLQ-H&N35) in head and neck patients. EORTC Quality of Life Group. Eur J Cancer. 2000;36(14):1796-807.
3. McLaughlin-Drubin ME, K. Munger, Oncogenic activities of human papillomaviruses. Virus Res, 2009. 143(2):195-208.
4. Marur S, Burtness B. Oropharyngeal squamous cell carcinoma treatment: current standards and future directions. Curr Opin Oncol. 2014;26(3):252-8.
5. Ducatman BS. The Role of Human Papillomavirus in Oropharyngeal Squamous Cell Carcinoma. Arch Pathol Lab Med. 2018;142(6): 715-718.
6. Dörr W, Hendry JH. Consequential late effects in normal tissues. Radiother Oncol. 2001;61(3):223-31.
7. Rieger JM, Zalmanowitz JG, Wolfaardt JF. Functional outcomes after organ preservation treatment in head and neck cancer: a critical review of the literature. Int J Oral Maxillofac Surg. 2006;35(7): 581-7.
8. Fung K, Lyden TH, Lee J, Urba SG, Worden F, Eisbruch A, et al. Voice and swallowing outcomes of an organ-preservation trial for advanced laryngeal cancer. Int J Radiat Oncol Biol Phys. 2005; 63(5):1395-9.
9. Eisbruch A, Schwartz M, Rasch C, Vineberg K, Damen E, Van As CJ, et al. Dysphagia and aspiration after chemoradiotherapy for headand- neck cancer: which anatomic structures are affected and can they be spared by IMRT? Int J Radiat Oncol Biol Phys. 2004; 60(5):1425-39.
10. Nguyen NP, Smith HJ, Sallah S. Evaluation and management of swallowing dysfunction following chemoradiation for head and neck cancer. Curr Opin Otolaryngol Head Neck Surg. 2007;15(2):130-3.
11. Jensen K, Lambertsen K, Grau C. Late swallowing dysfunction and dysphagia after radiotherapy for pharynx cancer: frequency, intensity and correlation with dose and volume parameters. Radiother Oncol. 2007;85(1):74-82.
12. Petruson K, Mercke C, Lundberg LM, Silander E, Hammerlid E. Longitudinal evaluation of patients with cancer in the oral tongue, tonsils, or base of tongue--does interstitial radiation dose affect quality of life? Brachytherapy. 2005;4(4):271-7.
13. Oates J, Clark JR, Read J, Reeves N, Gao K, O'Brien CJ. Integration of prospective quality of life and nutritional assessment as routine components of multidisciplinary care of patients with head and neck cancer. ANZ J Surg. 2008;78(1-2):34-41.
14. Nordgren M, Jannert M, Boysen M, Ahlner-Elmqvist M, Silander E, Bjordal K, et al. Health-related quality of life in patients with pharyngeal carcinoma: a five-year follow-up. Head Neck. 2006; 28(4):339-49.
15. Oates J, Clark JR, Read J, Reeves N, Gao K, O'Brien CJ. Integration of prospective quality of life and nutritional assessment as routine components of multidisciplinary care of patients with head and neck cancer. ANZ J Surg. 2008;78(1-2):34-41.
16. Adnan A, Högmo A, Sjödin H, Gebre-Medhin M, Laurell G, Reizenstein J, et al. Health-related quality of life among tonsillar carcinoma patients in Sweden in relation to treatment and comparison with quality of life among the population. Head Neck. 2020;42(5):860-872.
Full Text Sources:
Abstract:
Views: 2285

Watch Video Articles
For Authors
Journal Subscriptions

Current Issue
Dec 2025
Supplements
Instructions for authors
Online submission
Contact
ISSN: 2559 - 723X (print)
e-ISSN: 2601 - 1700 (online)
ISSN-L: 2559 - 723X
Journal Abbreviation: Surg. Gastroenterol. Oncol.
Surgery, Gastroenterology and Oncology (SGO) is indexed in:
e-ISSN: 2601 - 1700 (online)
ISSN-L: 2559 - 723X
Journal Abbreviation: Surg. Gastroenterol. Oncol.
Surgery, Gastroenterology and Oncology (SGO) is indexed in:
- SCOPUS
- EBSCO
- DOI/Crossref
- Google Scholar
- SCImago
- Harvard Library
- Open Academic Journals Index (OAJI)
Open Access Statement
Surgery, Gastroenterology and Oncology (SGO) is an open-access, peer-reviewed online journal published by Celsius Publishing House. The journal allows readers to read, download, copy, distribute, print, search, or link to the full text of its articles.
Surgery, Gastroenterology and Oncology (SGO) is an open-access, peer-reviewed online journal published by Celsius Publishing House. The journal allows readers to read, download, copy, distribute, print, search, or link to the full text of its articles.
Journal Metrics
Time to first editorial decision: 25 days
Rejection rate: 61%
CiteScore: 0.2
Time to first editorial decision: 25 days
Rejection rate: 61%
CiteScore: 0.2

Meetings and Courses in 2025
Meetings and Courses in 2024
Meetings and Courses in 2023
Meetings and Courses in 2022
Meetings and Courses in 2021
Meetings and Courses in 2020
Meetings and Courses in 2019
Verona expert meeting 2019

Surgery, Gastroenterology and Oncology applies the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits readers to copy and redistribute the material in any medium or format, remix, adapt, build upon the published works non-commercially, and license the derivative works on different terms, provided the original material is properly cited and the use is non-commercial. Please see: https://creativecommons.org/licenses/by-nc/4.0/
Publisher’s Note:
The opinions, statements, and data contained in article are solely those of the authors and not of Surgery, Gastroenterology and Oncology journal or the editors. Publisher and the editors disclaim responsibility for any damage resulting from any ideas, instructions, methods, or products referred to in the content.
 IASGO Society News
IASGO Society News