Surgery, Gastroenterology and Oncology

|
|
Introduction: Meckel’s diverticulum (MD) is a congenital anomaly resulting from incomplete regression of the vitelline duct, typically occurring in 2% of the population. Though usually asymptomatic, complications such as obstruction, bleeding, diverticulitis, and perforation may arise, especially in children. Diagnosis is often delayed due to its nonspecific presentation and similarity to other abdominal conditions.
Case Presentation: A 14-year-old boy presented with generalized abdominal pain, vomiting, fever, and signs of intestinal obstruction. The patient underwent exploratory laparotomy, which revealed a 7 cm Meckel’s diverticulum with proximal ileal twisting. A wedge excision with primary closure was performed. A second laparotomy revealed an ileal stricture at the excision site with dense adhesions. Adhesiolysis and segmental ileal resection with anastomosis were performed.
Literature Search Method: We conducted a literature search on PubMed, Google Scholar, and Cochrane using the keywords “Meckel’s diverticulum” and “children,” focusing on articles published between 2012 and 2022. Studies were included if they addressed incidental findings, symptomatic cases, complications, surgical techniques, and resection options relevant to the case presented.
Discussion: A total of eight studies met the inclusion criteria. The findings revealed that the
clinical presentation of Meckel’s diverticulum commonly mimics acute appendicitis, with patients frequently reporting right lower quadrant abdominal pain. Symptomatic MD is more common in males and often presents with obstruction or gastrointestinal bleeding. Surgical
management varies; wedge resection is suitable for narrow-based diverticula, while segmental resection is preferred for broad-based or complicated cases. Inadequate initial resection may lead to postoperative complications such as stenosis or adhesive small bowel obstruction.
Conclusion: Proper selection between wedge excision and segmental resection is essential to prevent postoperative complications, particularly in pediatric patients.
INTRODUCTION
Meckel’s diverticulum is a remnant of the embryonic vitelline (omphalomesenteric) duct, which connects the fetal intestine to the yolk sac and typically involutes between the 5th and 7th weeks of gestation. Failure of ductal regression may lead to several anomalies, the most common (90%) being classic Meckel’s diverticulum. It is a true diverticulum, composed of all normal layers of the intestinal wall (1,2).
Meckel’s diverticulum can be challenging to diagnose due to its asymptomatic nature or its presentation mimicking other abdominal conditions such as appendicitis, Crohn’s disease, and peptic ulcer disease. Clinical symptoms often arise from complications (4-6%) and are more frequently observed in children (males) than in adults, with reported male-to-female ratios ranging from 1.8:1 to 3:1 across different studies (1-4).
Individuals with Meckel’s diverticulum have a 4-6% lifetime risk of developing complications. The most common are intestinal obstruction (36.5%), intussusception (13.7%), diverticulitis and perforation (12.7% and 7.3%, respectively), gastrointestinal bleeding (11.8%), and tumors (3.2%). Intestinal obstruction is the most common manifestation in adults, although some studies report otherwise. An inflamed stricture at the base of Meckel’s diverticulum is among the rarest causes of small bowel obstruction, with only two cases reported in the literature to date (5-7).
Meckel’s diverticulum is rarely diagnosed in adolescents. Due to its uncommon presentation, symptomatic Meckel’s diverticulum is often misdiagnosed preoperatively. Definitive management includes surgical excision, such as wedge excision or segmental resection, depending on the integrity of the diverticular base, adjacent ileum, and the presence and location of ectopic tissue. There is no consensus regarding the preferred type of resection for symptomatic cases or whether incidentally discovered Meckel’s diverticulum should be resected. This remains a subject of debate, and it is reasonable that resection decisions be guided by identified risk factors (7,8).
CASE PRESENTATION
A 14-year-old boy with adequate nutritional status presented with generalized abdominal pain, described as continuous and accompanied by nausea and vomiting twice daily, with greenish fluid content.
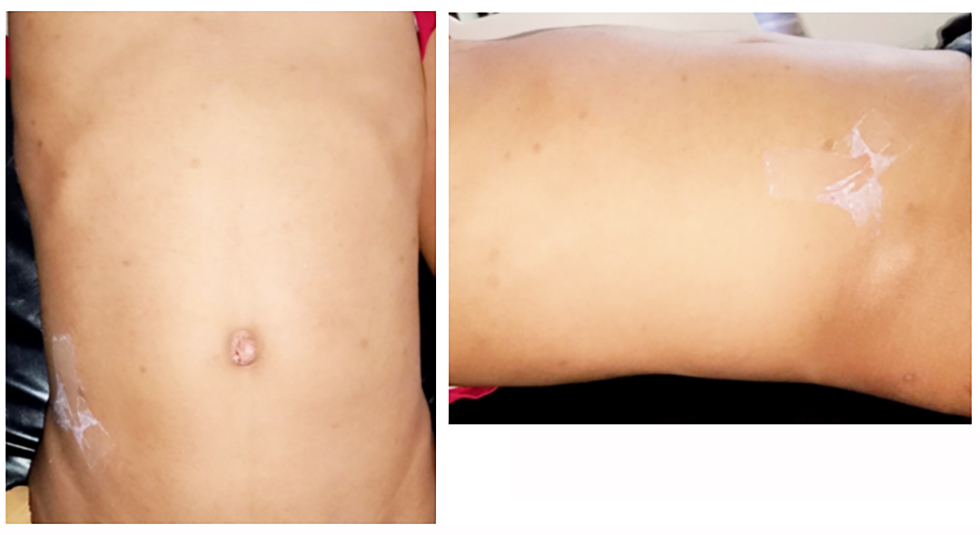
Figure 1 - Clinical Abdominal Photograph
 His last bowel movement occurred three hours prior to hospital admission, with a small amount of yellow, watery stool. Two days earlier, he had experienced
His last bowel movement occurred three hours prior to hospital admission, with a small amount of yellow, watery stool. Two days earlier, he had experienced frequent loose stools (>3 times/day) with particulate matter. The last passage of flatus was also two days ago. The symptoms were accompanied by fever, which began three days prior and persisted. The patient had undergone traditional abdominal massage therapy three times without improvement before being brought to the Emergency Department of Dr. Hasan Sadikin General Hospital, Bandung.
Physical Examination Findings
The patient was alert (compos mentis) with a body temperature of 38°C. Abdominal examination revealed a distended abdomen without visible bowel contours or peristaltic movements (as can be seen in fig. 1).
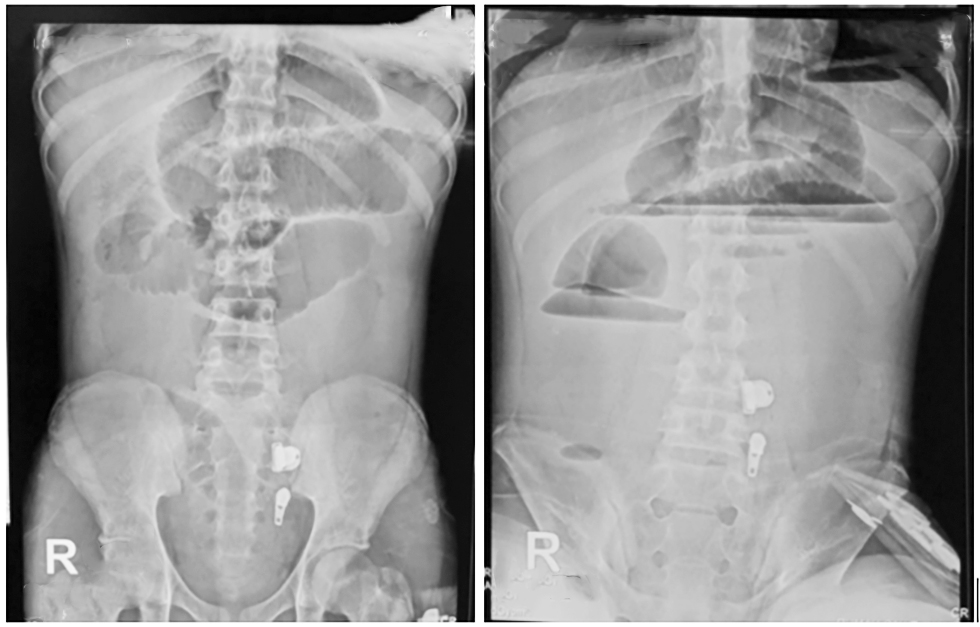
Figure 2 - Abdominal X-ray
 Bowel sounds were diminished. The abdomen was tense with generalized tenderness and muscular guarding. Digital rectal examination showed a collapsed ampulla. Nasogastric tube insertion yielded 250 cc of greenish fluid. Laboratory tests showed a leukocyte count of 4,910/µL and a C-reactive protein (CRP) level of 13.28 mg/L.
Bowel sounds were diminished. The abdomen was tense with generalized tenderness and muscular guarding. Digital rectal examination showed a collapsed ampulla. Nasogastric tube insertion yielded 250 cc of greenish fluid. Laboratory tests showed a leukocyte count of 4,910/µL and a C-reactive protein (CRP) level of 13.28 mg/L.
(a) front view; (b) side view
Abdominal X-ray revealed dilated small bowel loops with a "hearing bone" appearance. No colonic gas was observed. Multiple air-fluid levels were present, suggesting a high-level intestinal obstruction (fig. 2).
(a) supine; (b) erect
The patient was clinically diagnosed with diffuse peritonitis, suspected to be due to a perforated appendicitis, and was scheduled for exploratory laparotomy.
Surgery
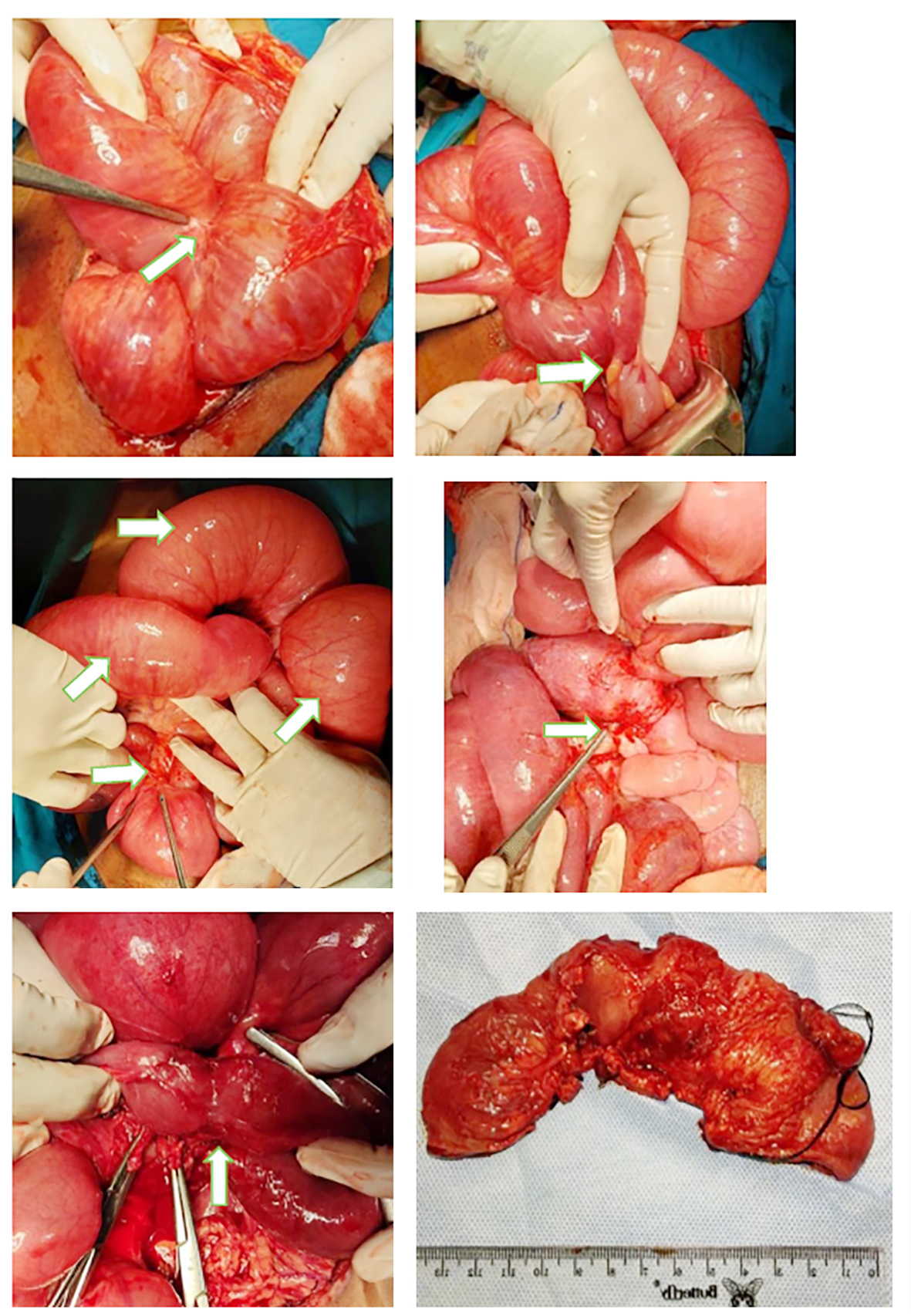
Intraoperative findings revealed a Meckel’s diverticulum 270 cm distal to the ligament of Treitz, accompanied by Grade 2 adhesions and evidence of proximal ileal twisting at its base. A wedge excision of the diverticulum was performed, followed by primary closure and over-sewing of the involved bowel segment. The Meckel’s diverticulum measured 7 cm in length, 4 cm in width, with a 1.5 cm base diameter (fig. 3).
Figure 3 - Intraoperative photographs from exploratory laparotomy.
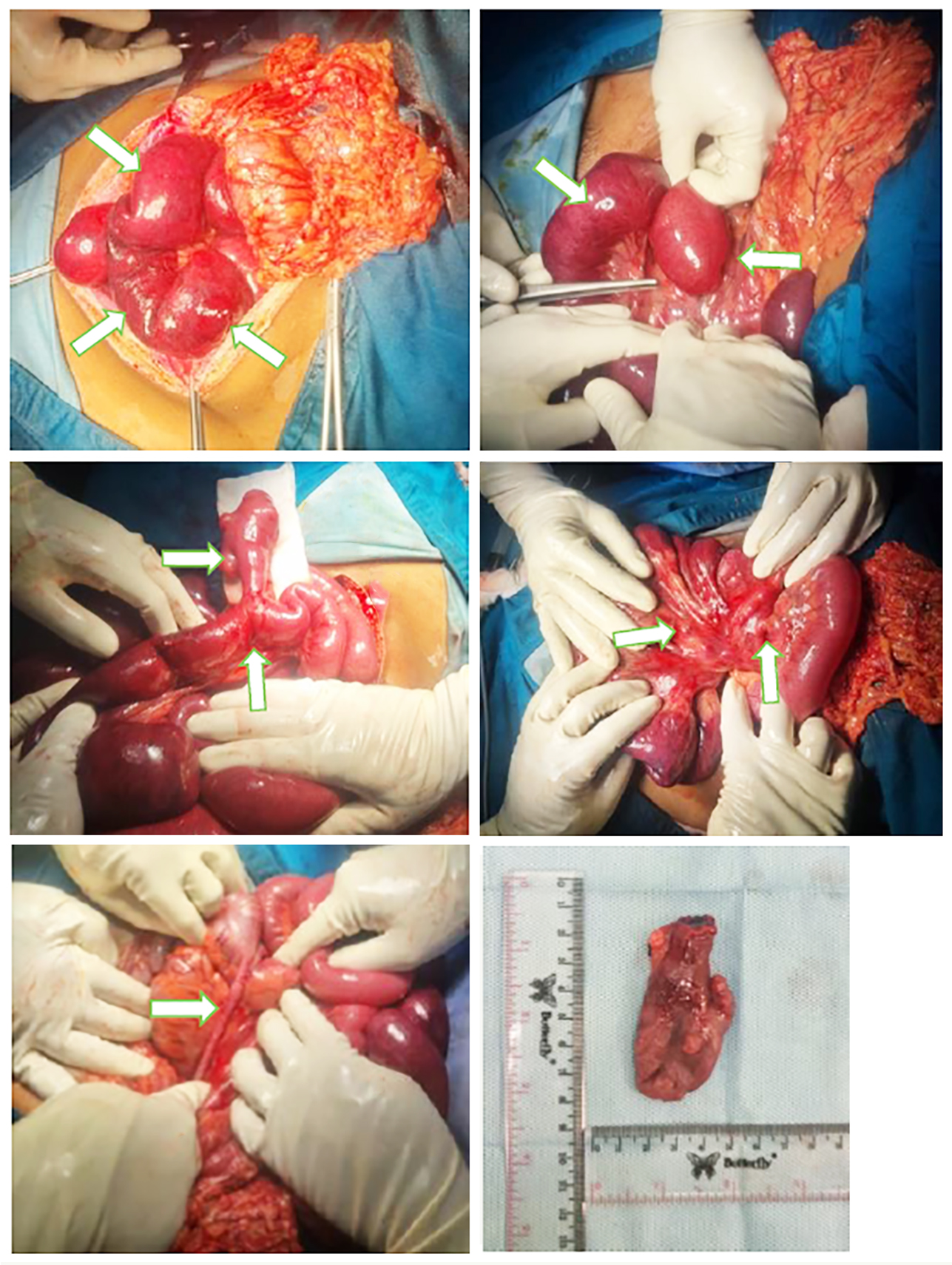
(a) hematoma along the entire jejunoileal region;
(b) dilatation of the jejunum extending to the ileum; (c) Meckel’s diverticulum located 270 cm distal to the ligament of Treitz, with Grade 2 adhesions in the surrounding area and evidence of proximal ileal twisting at the diverticular base; wedge excision was performed followed by
primary closure and over-sewing of the affected intestinal segment; (d) enlarged lymph nodes in the mesentery near the diverticulum; (e) no signs of inflammation in the appendix; (f) Meckel’s diverticulum measuring 7 cm in length, 4 cm
in width, and 1.5 cm at the base diameter
The histopathological examination revealed non-specific chronic inflammation with mild dysplasia in the Meckel’s diverticulum. On postoperative day 10, signs of partial small bowel obstruction were observed (fig. 4), suspected to be due to adhesions or ileal stenosis at the site of previous wedge excision with primary closure and over-sewing. Initially, the condition was managed with conservative measures, including bowel rest (NPO), intravenous fluid administration, and nasogastric tube decompression to relieve pressure. However, as these measures did not resolve the obstruction, a decision was made to proceed with a re-exploratory laparotomy to lyse the adhesions and restore normal bowel function.
Figure 4 - Follow-up Abdominal X-ray
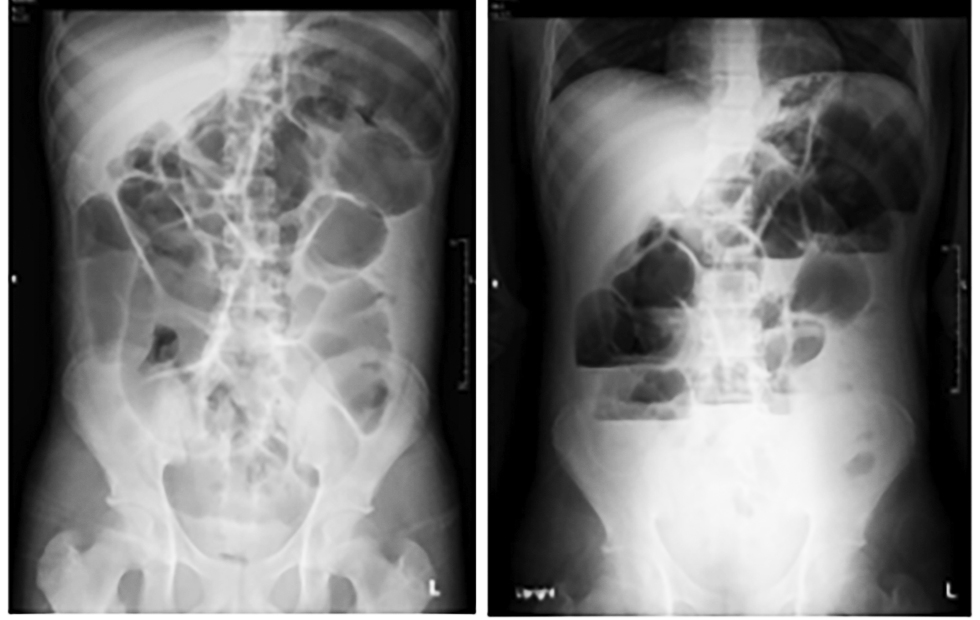
(a) supine; (b) erect
Re-exploratory laparotomy revealed a partial small bowel obstruction caused by an ileal stricture at the site of the previous Meckel’s diverticulum, following wedge excision, primary closure, and over-sewing. This was accompanied by Grade 3-4 adhesions. The patient subsequently underwent adhesiolysis and end-to-end ileal resection with anastomosis (fig. 5).
Figure 5 - Intraoperative photographs from re-exploratory laparotomy. (a) grade 3-4 adhesions between ileal loops, with evidence of healing from a previous ileal hematoma; (b) grade 3-4 adhesions between the ileum and sigmoid colon; (c) dilated ileum at adhesion sites; (d, e) fibrotic tissue at the previous wedge excision site forming an 8 cm ileal stricture; (f) a 14 cm ileal segment was resected, including the stricture portion
LITERATURE SEARCH
Search Strategy
We conducted a literature search using PubMed, Google Scholar, and Cochrane Database with the keywords "Meckel’s diverticulum," "children," and "management." The search ocused on the management approaches used in patients. Articles included were those published between 2012 and 2022. Studies selected for analysis addressed incidental findings, symptomatic cases, complications, surgical methods, and resection options relevant to the case presented above - a 14-year-old boy who underwent exploratory laparotomy with wedge excision of Meckel’s diverticulum. We conducted a critical appraisal using the CEBM (University of Oxford) and the JBI critical appraisal checklist. The level of evidence (LoE) was determined according to the 2011 classification by the Centre for Evidence-Based Medicine, University of Oxford.
Searching Result
The search yielded 347 articles, of which only 8 met the inclusion criteria and were therefore included in the review (table 1). The flow diagram of the study selection process is presented at fig. 6. A summary of the included articles is presented in table 2. Due to the time required for a comprehensive systematic review, our literature search was conducted up to the year 2022. Consequently, our findings may not reflect publications from 2023 onwards.
Table 1 - Literature search strategy and results by database

Table 2 - Literature search results
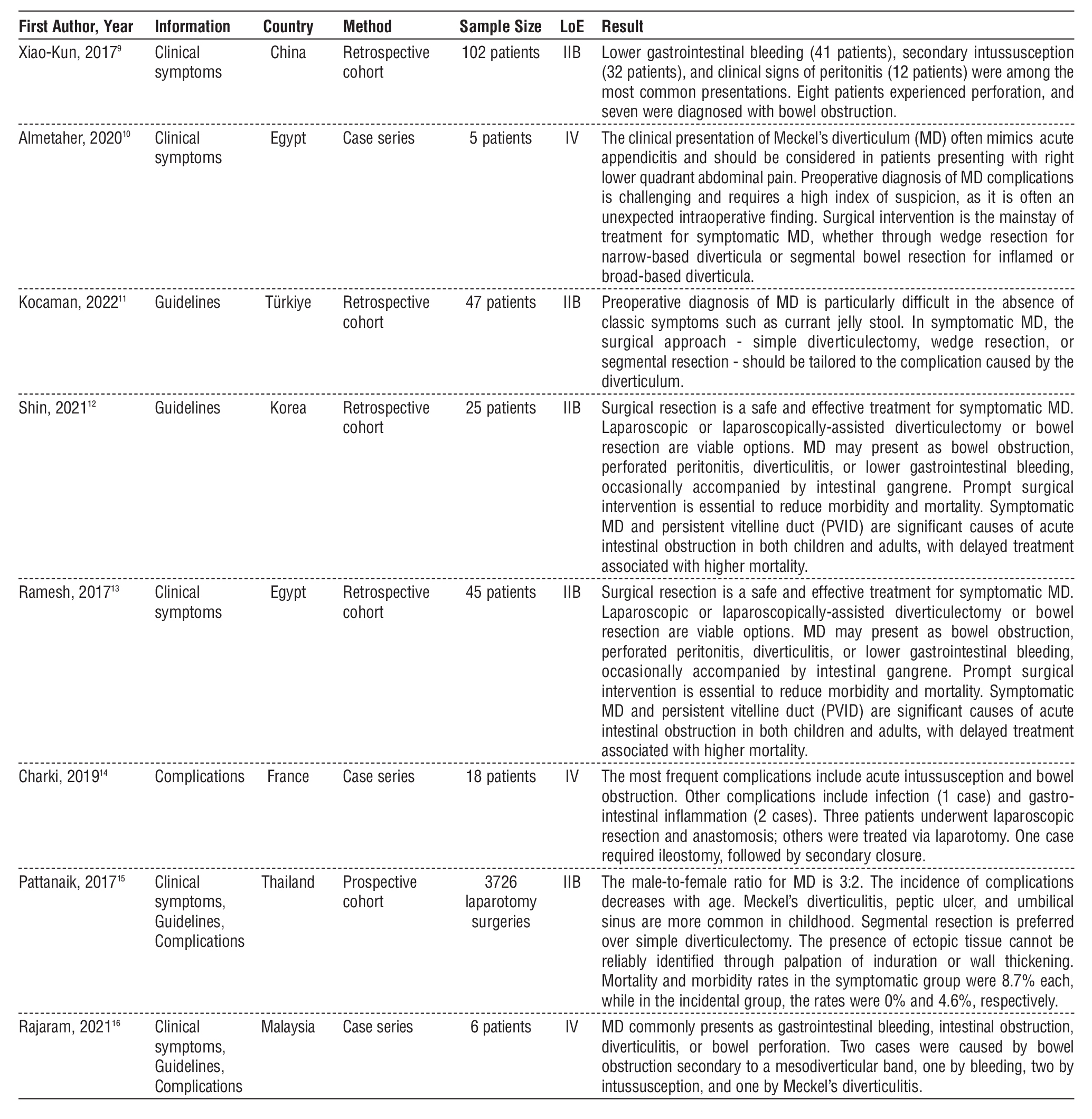
Figure 6 - Literature search flow diagram
DISCUSSION
A 14-year-old boy with adequate nutrition presented with generalized abdominal pain. On
physical examination, no visible bowel contour or peristalsis was observed on the abdominal wall, bowel sounds were diminished, and muscular guarding was noted. Digital rectal examination revealed a collapsed ampulla. The working diagnosis was diffuse peritonitis, suspected to be due to perforated appendicitis, and the patient was scheduled for exploratory laparotomy.
A study by Shin et al. (12) reported a male-to-female ratio of 3.1:1 in patients with Meckel’s diverticulum (MD). Males were more likely to present with melena, bowel obstruction, and diverticulitis, while no gender difference was found in intussusception or perforation cases. The mean age at diagnosis was 92.4 months (range: 6 months to 17 years), with 18.2% of patients under 1 year old, 18.2% between 1 and 5 years, and 63.6% over 5 years. Average age by presentation was 7.2 years (melena), 9.2 years (obstruction), 6.8 years (intussusception), 8.4 years (perforation), and 6.8 years (diverticulitis).
These findings indicate that MD often presents with nonspecific symptoms that vary by age. MD may manifest as bowel obstruction, perforated peritonitis, diverticulitis, or lower gastrointestinal bleeding, sometimes with associated bowel gangrene. Prompt surgical intervention is essential to reduce morbidity and mortality. Symptomatic MD is a significant cause of acute abdominal or intestinal obstruction in both children and adults, and delays in treatment are linked to increased mortality (13).
Meckel’s diverticulum results from incomplete obliteration of the omphalomesenteric duct, which connects the yolk sac to the midgut during embryonic development. This duct normally regresses by the seventh gestational week (17). Failure of regression can result in omphalomesenteric cysts, fistulas, or fibrous bands that may lead to obstruction. If no attachments are present, a true diverticulum may form - Meckel’s diverticulum (18).
The "rule of twos" summarizes MD: present in 2% of infants, 2 inches long, located within 2 feet of the ileocecal valve, twice as common in males, and often containing two types of tissue (gastric or pancreatic). It is a true diverticulum, containing all layers of the small intestinal wall. Ectopic mucosa - typically gastric, but also pancreatic, duodenal, colonic, or hepatobiliary -may be present in 15% of cases (19,20).
Many patients with MD remain asymptomatic. Risk factors for symptomatic MD include age <50, male sex, diverticula >2 cm, presence of ectopic mucosa, wide base, and fibrous bands. Gastric mucosa secretes acid that is not neutralized, leading to adjacent ulceration and painless rectal bleeding (21-23).
Ectopic mucosa may originate from the pancreas, jejunum, or both. Bleeding usually occurs distal to the diverticulum. Fibrous bands can cause small bowel obstruction, and MD may serve as a lead point for intussusception. Retained MD can also obstruct the small bowel. Diverticulitis may result in perforation (23,24).
Histopathology in this case revealed nonspecific chronic inflammation with mild dysplasia in the Meckel’s diverticulum. While MD typically shows ileal mucosa, ectopic tissues like gastric (57%), duodenal, colonic, pancreatic, Brunner's glands, hepatobiliary, or endometrial mucosa may be found, usually near the tip. Reported complication rates range from 4-16% (19).
Shin et al. (12) also found that some MD cases were incidentally discovered during unrelated surgeries: 10% during appendectomy, one during resection of a mature ovarian teratoma, and another during laparotomy for congenital megacolon. Symptomatic cases made up 70%, with presentations including melena (39.4%), obstruction (21.2%), intussusception (15.2%), perforation (15.2%), and diverticulitis (9%).
On postoperative day 10, the patient exhibited signs of partial small bowel obstruction, suspected to be due to adhesive small bowel obstruction (ASBO) or ileal stenosis following wedge excision and primary closure at the previous MD site. The patient underwent re-exploratory laparotomy. Pattanaik et al. reported an 8.7% mortality rate following relaparotomy and recommended segmental resection over simple diverticulectomy (LoE: IV, Recommendation: B).
A case series by Rajaram et al. (16) showed that surgical resection is the preferred treatment for symptomatic MD. Options include wedge resection for narrow-based diverticula or segmental ileal resection for broad-based lesions. Resection can be performed via laparotomy, laparoscopically assisted intracorporeal or extracorporeal methods (LoE: IV, Recommendation: C).
Kocaman et al. (11) recommended wedge resection to include adjacent ileum in patients with gastro-intestinal bleeding, while segmental resection is preferred for larger or advanced lesions. Simple diverticu-lectomy may suffice for early intussusception, but in advanced cases, segmental resection is preferred due to associated edema (LoE: IIB, Recommendation: B).
Another series involved 15 patients undergoing segmental bowel resection to remove MD, with primary anastomosis in 12 cases. Morbidities included two fistulas and two cases of postoperative peritonitis. One patient died from septic shock. Heterotopic mucosa was found in three patients, including gastric, colonic, and combined types (25).
CONCLUSION
It is essential to determine whether to perform wedge excision or segmental resection of Meckel’s diverticulum based on intraoperative findings - particularly in pediatric patients - considering the length and width of the diverticulum, the presence of fibrotic changes at its base, and the surrounding area’s potential for recurrent obstruction due to ileal stricture.
Conflict of Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Funding
This work received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
REFERENCES
1. Keese D, Rolle U, Gfroerer S, Fiegel H. Symptomatic Meckel’s Diverticulum in Pediatric Patients-Case Reports and Systematic Review of the Literature. Front Pediatr. 2019;7:267.
2. Charles L. Meckel Diverticulum. In: Holcomb and Ashcraft’s Pediatric Surgery. Sydney: Lippincot Williams & Wilkins; 2020. hal. 641.
3. Martin JP, Connor PD, Charles K. Meckel’s diverticulum. Am Fam Physician. 2000;61(4):1037-1042,1044.
4. Soltero MJ, Bill AH. The natural history of Meckel’s Diverticulum and its relation to incidental removal. A study of 202 cases of diseased Meckel’s Diverticulum found in King County, Washington, over a fifteen year period. Am J Surg. 1976;132(2):168-73.
5. Cullen JJ, Kelly KA, Moir CR, Hodge DO, Zinsmeister AR, Melton LJ 3rd. Surgical management of Meckel’s diverticulum. An epidemiologic, population-based study. Ann Surg. Oktober 1994;220(4): 564–9.
6. Sharma RK, Jain VK. Emergency surgery for Meckel’s diverticulum. World J Emerg Surg. 2008;3:27.
7. Sallam M, Mena J. Meckels Diverticulum: Wedge Excision vs Segmental Resection Anastomosis. Int J Sci Res. 2021;10(1):617-20.
8. Blouhos K, Boulas KA, Tsalis K, Barettas N, Paraskeva A, Kariotis I, et al. Meckel’s Diverticulum in Adults: Surgical Concerns. Front Surg. 2018;5:55.
9. Lin X-K, Huang X-Z, Bao X-Z, Zheng N, Xia Q-Z, Chen C. Clinical characteristics of Meckel diverticulum in children: A retrospective review of a 15-year single-center experience. Medicine (Baltimore). 2017;96(32):e7760.
10. Almetaher HA, Mansour MA. Acute abdomen in children due to different presentations of complicated Meckel’s diverticulum: a case series. Ann Pediatr Surg. 2020;16(1):45.
11. Kocaman OH, Günendi T. Different clinical symptoms and surgical treatment of Meckel’s diverticulum in children. Dicle T?p Derg. 2022;49:21-8.
12. Shin HW, Kang YN, Park J. Clinicopathologic Characteristics and Treatment Outcomes of Symptomatic Meckel’s Diverticulum in Children. Adv Pediatr Surg. 2021;27(1):8–14.
13. Ramesh A, Rajavelu R, Khan AMI. Management of Meckel’s diverticulum--a retrospective study in a tertiary care Centre. J Evol Med Dent Sci. 2017;6(24):1992-1997.
14. Charki MT, Oukhouya M-A, Benmassaoud Z, Mahmoudi A, Khattala K, Bouabdallah Y. Complications of Meckel’s diverticulum in children: about 18 cases. Pan Afr Med J. 2019;33:113.
15. Pattanaik SK, Dandpat BP, Mishra JM. Incidence, complications and management of Meckel’s diverticulum. Int Surg J. 2017;4(9):3083-3087.
16. Rajaram G, Singh PKBB, Madzlan MF Bin, Fernandez JEG, Badarudin NBM. Meckel’s diverticulum and its plethoric presentation in paediatric surgery: a case series. Int Surg J. 2021;8(12):3668-3672.
17. Grosfeld JL, O’Neill JA, Fonkalsrud EW. Pediatric Surgery. Philadelphia: Elsevier Saunder; 2012.
18. Yahchouchy EK, Marano AF, Etienne JC, Fingerhut AL. Meckel’s diverticulum. J Am Coll Surg. 2001;192(5):658-62.
19. Sagar J, Kumar V, Shah DK. Meckel’s diverticulum: a systematic review. J R Soc Med. 2006;99(10):501–5.
20. Coran AG, Adzick NS, Krummel TM, Laberge JM, Shamberger R, Caldamone A. Pediatric Surgery. 7 ed. New York: Elsevier Saunder; 2012.
21. St-Vil D, Brandt ML, Panic S, Bensoussan AL, Blanchard H. Meckel’s diverticulum in children: a 20-year review. J Pediatr Surg. 1991; 26(11):1289–92.
22. Uppal K, Tubbs RS, Matusz P, Shaffer K, Loukas M. Meckel’s diverticulum: a review. Clin Anat. 2011;24(4):416–22.
23. Horkoff MJ, Smyth NGC, Hunter JM. A large incarcerated Meckel’s diverticulum in an inguinal hernia. Int J Surg Case Rep. 2014; 5(12):899–901.
24. Evola G, Piazzese E, Bonanno S, Di Stefano C, Di Fede GF, Piazza L. Complicated Littre’s umbilical hernia with normal Meckel’s diverticulum: A case report and review of the literature. Int J Surg Case Rep. 2021;84:106126.
25. Diop A, Thiam O, Guèye ML, Seck M, Touré AO, Cissé M, et al. Complicated Meckel diverticula: about 15 cases. Pan Afr Med J. 2018;29:81. French
Full Text Sources:
Abstract:
Views: 1877

For Authors
Journal Subscriptions

Dec 2025
Supplements
Instructions for authors
Online submission
Contact
e-ISSN: 2601 - 1700 (online)
ISSN-L: 2559 - 723X
Journal Abbreviation: Surg. Gastroenterol. Oncol.
Surgery, Gastroenterology and Oncology (SGO) is indexed in:
- SCOPUS
- EBSCO
- DOI/Crossref
- Google Scholar
- SCImago
- Harvard Library
- Open Academic Journals Index (OAJI)
Surgery, Gastroenterology and Oncology (SGO) is an open-access, peer-reviewed online journal published by Celsius Publishing House. The journal allows readers to read, download, copy, distribute, print, search, or link to the full text of its articles.
Time to first editorial decision: 25 days
Rejection rate: 61%
CiteScore: 0.2

Meetings and Courses in 2025
Meetings and Courses in 2024
Meetings and Courses in 2023
Meetings and Courses in 2022
Meetings and Courses in 2021
Meetings and Courses in 2020
Meetings and Courses in 2019
Verona expert meeting 2019

Surgery, Gastroenterology and Oncology applies the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits readers to copy and redistribute the material in any medium or format, remix, adapt, build upon the published works non-commercially, and license the derivative works on different terms, provided the original material is properly cited and the use is non-commercial. Please see: https://creativecommons.org/licenses/by-nc/4.0/
Publisher’s Note:
The opinions, statements, and data contained in article are solely those of the authors and not of Surgery, Gastroenterology and Oncology journal or the editors. Publisher and the editors disclaim responsibility for any damage resulting from any ideas, instructions, methods, or products referred to in the content.
 IASGO Society News
IASGO Society News