Surgery, Gastroenterology and Oncology

|
|
Introduction: Globally, gliomas account for > 70% of all brain tumors. In Iraq, there is a tendency towards a higher rate of gliomas. The Iraqi Cancer Registry in 2019 stated that central nervous system tumors were among the top three cancers in Iraq and the top ten cancers in Sulaymaniyah.
Method: In this hospital-based frequency analysis study, information from 104 glioma patients was reviewed. Data were then subjected to critical analysis, taking into consideration all relevant factors related to demographic data (age, sex, and ethnicity). The histopathological type, tumor grade, and site of disease were collected.
Results: Patients with grade I gliomas had a mean age of 24.71±19.08 years, while those with grade II, grade III, and grade IV tumors had mean ages of 38.5±17.02, 45.67±17.03, and 50.74 ± 17.83 years, respectively (p = 0.001). Grade IV gliomas had a higher percentage (45, 47.4%). HGG (64.2%) were more common than LGG (35.8%). GBM was common in 49 patients (80.3%). The left parietotemporal region was the most common site of gliomas (32.2%). The results indicated that older age at diagnosis was significantly associated with higher risk (OR=1.04, p=0.002). Additionally, tumors located in the parietotemporal region were more likely to have HGG (OR=2.3, p=0.03).
Conclusion: HGG, particularly glioblastoma, is more prevalent than low-grade gliomas. The mean age at diagnosis increased with the tumor grade, indicating that older patients were more likely to develop high-grade tumors. Tumor location is also an important factor, with high-grade gliomas more frequently observed in the parietotemporal and frontal lobes. Multivariate analysis identified age and parietotemporal location as the independent predictors of HGG.
Introduction
Neoplastic transformation in NS is a multistep process in which the normal controls of cell proliferation and cell-cell interactions are suppressed or disabled. This process involves the alteration of several types of genes, including oncogenes, tumor suppressor genes, DNA repair genes, and cell death genes, among others (1,2). Results from comprehensive analyses of tumor genomes (DNA) and transcriptomes (mRNA) are especially intriguing regarding the potential of molecular profiling approaches toward the differential diagnosis of adult malignant gliomas (3,4).
The SEER registry reports that the primary CNS tumor rate is between 2.2 and 8.3 per 100,000 people per year, based on race and sex (5). The WHO grading of CNS tumors establishes a malignancy scale based on the histological features of the tumor (6). The presenting signs and symptoms in patients with primary brain tumors can be generalized or focal. In the initial stages of the disease (low-grade tumors), most symptoms are focal. Generalized symptoms occur with an increase in tumor size. Common generalized symptoms include headache, nausea, vomiting, seizures, and altered mental functions (7).
Diagnosis begins with appropriate brain imaging followed by histopathology to confirm the diagnosis. Several imaging modalities can be helpful when performing the initial work-up and follow-up and in the evaluation and treatment of brain tumors. With recent advances in structural and functional brain imaging techniques, physicians can determine the tumor location and biological activity. These techniques are also used to assess the effects of treatment, differentiate tumor recurrence from radiation necrosis, and determine tumor progression (8,9).
Comprehensive neurosurgical evaluation is necessary to obtain tissue for diagnosis or possible resection of the tumor. Surgery is the treatment of choice for primary brain tumors if the patient is a candidate for complete resection. Surgery is often curative for intracranial tumors that occur outside the brain, such as pituitary adenomas, schwannomas, and meningiomas (10). The decision to perform complete resection is based on the location and extent of the tumor, histopathology, and comorbid conditions. Patients with asymptomatic meningiomas can be followed-up clinically with serial CT or MRI scans every two–three years. A significant increase in meningioma size on a scan or presence of neurological symptoms requires neurosurgical evaluation. In patients with high-grade gliomas, near-total resection is important to decrease tumor burden, relieve intracranial pressure, and improve survival (10). Postsurgical radiation is the standard treatment for high-grade gliomas, although its role in the treatment of low-grade gliomas remains controversial. Radiation can be delivered either externally or internally (11).
The main types of external beam radiation are conventional and stereotactic radiosurgery. Stereo-tactic radiosurgery often delivers a single high dose of radiation in a one-day session, but it can be administered in two or three large doses. Currently, stereotactic radiosurgery can be used to palliate small, well-demarcated volumes of recurrent glioblastoma multiforme, and as a boost after conventional external beam radiotherapy. However, its use in the treatment of newly diagnosed malignant gliomas is controversial. Interstitial radiotherapy (brachytherapy) involves surgical implantation of radioactive material directly inside the tumor (11,12). Combining temozolomide (Temodar) therapy with radiation improves the survival of patients with HGG (13).
Data from the Glioma Outcomes Project revealed that tumor grade, patient age, functional status, and complete resection were prognostic factors for survival in patients with recently diagnosed malignant gliomas (14).
Due to such a discrepancy between Western literature and the Iraqi data, we believe that there is a need to study the pattern of brain tumors in the Iraqi Kurdish population. We started with Sulaymaniyah province because of the noticeable availability of local dates and the progress that has been witnessed in the management of these patients. Piecemeal in-depth studies of this nature could act as a building block for future national data collection involving the entire country.
Methods
Study Design and Setting
Hospital-based frequency analysis study information was reviewed from 104 glioma patients Kurdish patients from Sulaymaniyah with primary brain tumors between January 2019 and July 2023, via revision of the patient’s database provided by the hospitals’ registration systems.
Data Collection
The data included age and sex of the patients. The histopathological type, tumor grade, and site of disease were also recorded.
Inclusion criteria
1. Primary brain tumors.
2. Kurdish ethnicity.
Exclusion criteria
1. Spinal cord tumors.
2. Brain lymphoma.
3. Secondary metastasis to the brain.
4. Non-Glioma brain tumors.
5. Non-Kurdish ethnicity.
Then, all available data were subjected to descriptive and analytic studies as much as possible.
Statistical analysis
Statistical analyses were performed using the appropriate statistical software (IBM SPSS version 24). Continuous variables were expressed as mean ± SD, while categorical variables were presented as frequencies and percentages. ANOVA was used to compare the mean age at diagnosis among the different grades (I–IV). The chi-squared (c2) test was used to evaluate the association between categorical variables. Statistical significance was set at p ? 0.05.
Results
Of the 150 patients, 104 cases were included, of which 95 cases were histologically identified and reported as brain glioma and 9 patients were without histopathology reports. Based on the 2007 WHO classification system, the following types of brain tumors were identified: astrocytomas (n=19), glio-blastoma (n=49), oligodendroglioma (n=2), oligo-astrocytoma (n=1), ependymoma (n=1), brainstem tumor (n=1), medulloblastoma (n=1), pituitary adenoma (n=1), and meningioma (n=21). 46 cases with spinal cord tumors (n=6), lymphoma (n=2), germ cell tumors (n=1), and secondary brain metastasis (n=37) were excluded (fig. 1).
Figure 1 - Flow-chart of the study

Figure 2 - Geographical distribution of brain and CNS tumors is concerned in the Province of Sulaymaniyah.
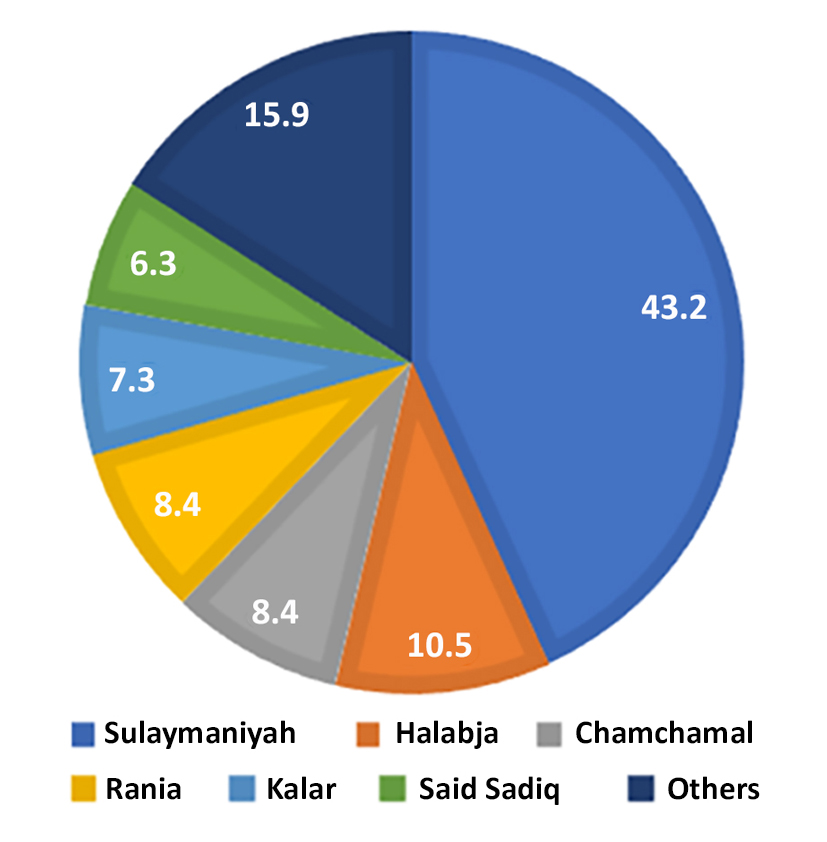
Regarding the geographical distribution of brain and CNS tumors in the Province of Sulaymaniyah during the study period, 41(43.2%) were from the city of Sulaymaniyah, and the rest were from the outskirts of the city. Ten cases (10.5%) were from Halabja, eight (8.4%) were from Chamchamal and Rania, seven (7.3%) from Kalar, six (6.3%) from Said Sadiq, and 15(15.9%) from other villages (fig. 2).
According to age at diagnosis, the range was 1-85 years. The median age of our patients was 42 years, and the mean was 42.25±20.49 years. Table 1 shows the mean age at diagnosis according to tumor grade. The mean age at diagnosis increased progressively with higher tumor grade. Patients with grade I gliomas had a mean age of 24.71 ± 19.08 years, while those with grade II, grade III, and grade IV tumors had mean ages of 38.50 ± 17.02, 45.67 ± 17.03, and 50.74 ± 17.83 years, respectively. Statistical analysis using a one-way ANOVA test (F= 9.65) revealed a significant difference in the mean age among the different tumor grades (p = 0.001), indicating that higher-grade gliomas tend to occur in older patients.
Table 1 - The mean age at diagnosis in relation to grade of the tumor (n=95).

Figure 3 - Grades of Glioma
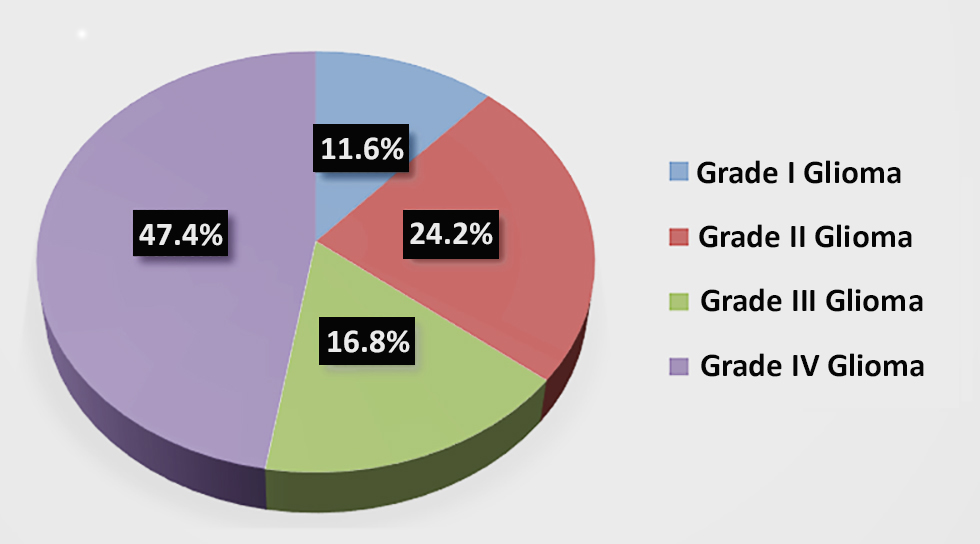
Glioblastoma has a higher number of cases each year. Grade IV gliomas had the highest percentage (45, 47.4%), followed by Grade II (23, 24.2%), Grade III (15, 16.8%), and Grade I (11, 11.6%) gliomas. The percentage of HGG (64.2%) was higher than that of LGG (35.8%) (1.79:1). In addition, out of the 61 cases of HGG, 49(80.3%) had HGG, which was more than two-thirds of the HGG cases.
Table 2 - Distribution of sex according to Glioma grades.
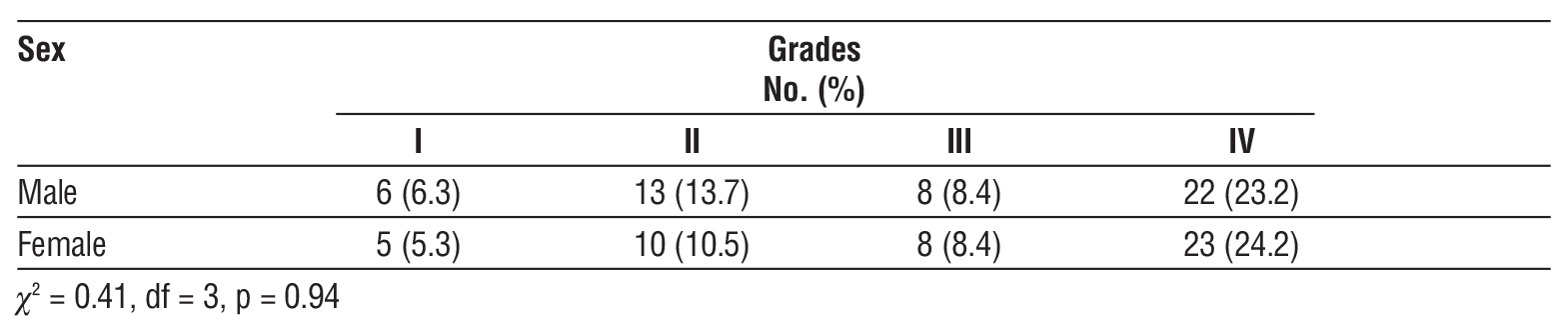
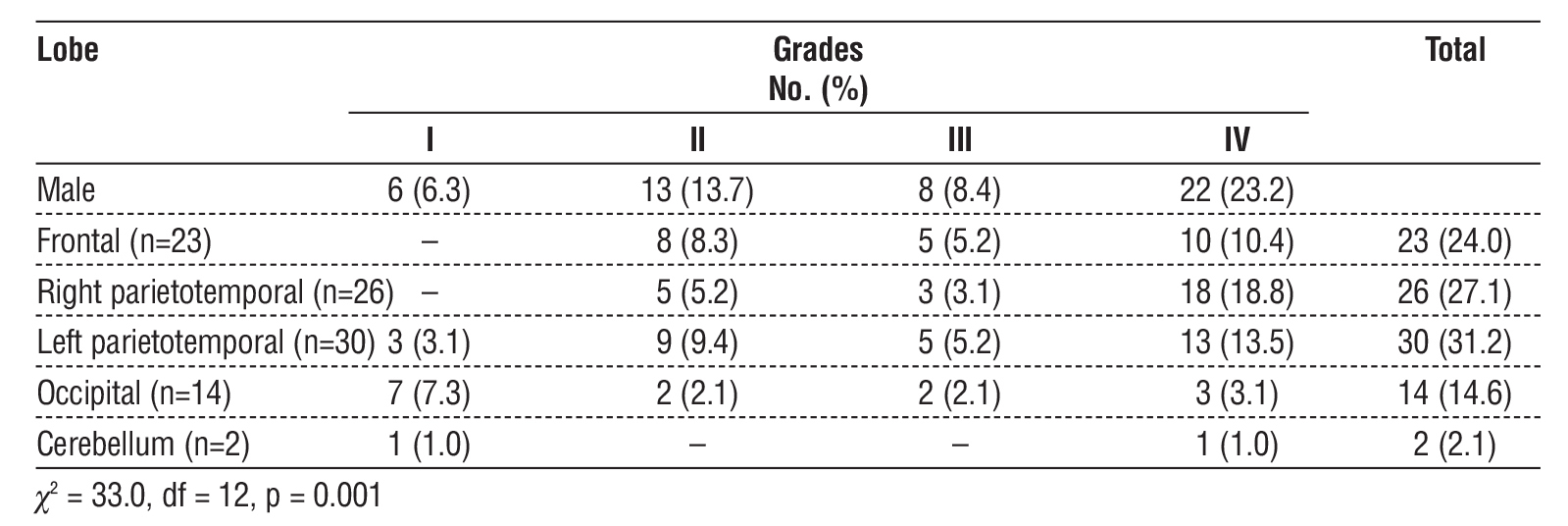
Table 3 - The distribution of Glioma site in regard to grades.
Multivariate logistic regression analysis was performed to identify independent predictors of HGG (grades III-IV), with tumor grades dichotomized into low-grade (grades I-II) and high-grade (grades III-IV). The analysis included age at diagnosis, sex, and tumor location as potential predictors (table 4). The results indicated that older age at diagnosis was significantly associated with a higher odds of having a high-grade tumor (OR=1.04, p=0.002). Additionally, tumors located in the parietotemporal region were more likely to have HGG than other locations (OR=2.3, p=0.03).
Table 4 - Multivariate logistic regression analysis for predictors of HGG
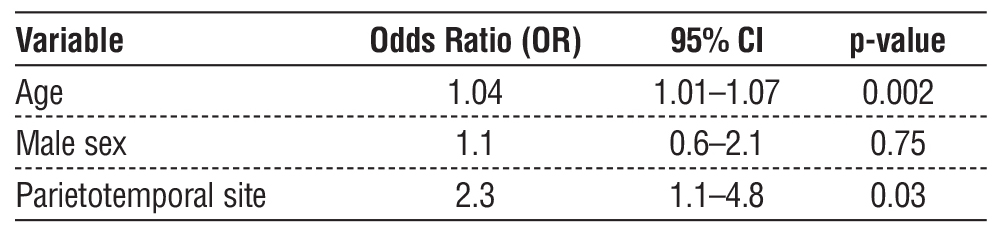
Sex was not a significant predictor in this model.
These findings suggest that age and tumor site are independent predictors of glioma grade, highlighting their potential importance in risk stratification and clinical management.
Discussion
Brain tumors in the last four years from patients from all of Iraq in Hiwa Hospital were ranked 7th, 5th, 4th, 5th, 6th sequentially. In comparison with the worldwide reported data brain tumor not included in the top ten commonest cancer (15). CNS tumors are among the top 10 most common cancers all over Iraq, according to the Iraqi Cancer Board registry 2009 and 2023 (16-18).
According to the Pathology Group of the International Agency for Research on Cancer, Lyon, France. Gliomas account for more than (70%) of all brain tumors. Glioblastoma is the most frequent and malignant histological type of glioblastoma (19).
According to the CBTRUS Statistical Report 2012, Primary Brain and Central Nervous System Tumors diagnosed in the US in 2005–2009, astrocytomas, and glioblastomas accounted for (76%) of all gliomas (20).
Our study showed that grade IV gliomas had a higher percentage (45, 47.4%). The percentage of HGG (64.2%) was higher than that of LGG (35.8%). In addition, out of the 61 cases of HGG, GBM was
common in 49 patients (80.3%). In Comparison to the CBTRUS Statistical Report 2012 (21), the percentage of glioblastoma is near, while the total number of astrocytomas with glioblastoma is lower.
For the histological subgroups of total gliomas, diffuse, pilocytic, and anaplastic astrocytomas, glioblastoma, oligodendrogliomas, ependymomas, and brainstem gliomas were distributed within all age intervals, with a sudden peak appearing in older age groups. This is similar to the rate of malignant gliomas in people aged > 70 years in the US, and the CBTRUS Statistical Report 2012 showed that the median age at diagnosis for all primary brain and CNS tumors was 59 years (19).
According to the CBTRUS Statistical Report, primary brain and CNS tumors diagnosed in the US in 2004-2008, showed that the rate of brain gliomas is higher in males than in females by about 1.42:1 (19). In addition, the frequency of brain and CNS tumors according to histology and sex is higher in male than females (21). The data of this study showed a relatively similar percentage in both sexes.
According to the Cancer Research UK and UK Association of Cancer Registries (UKACR), 2004 registry, it shown that males are more likely to be diagnosed with brain tumors than females, with a male: female ratio of 1.5:1. However, this disguises the fact that women are more likely to develop meningiomas than men (22).
In this study, the left parietotemporal region was the most common site of glioma, representing 30 cases (32.2%), followed by the right parietotemporal region (26 cases, 27.1%), and the frontal lobe (23 cases, 24.0%). Occipital tumors accounted for 14 cases (14.6%), whereas the cerebellum was the least frequent site in two cases (2.1%). According to an international study that showed the anatomic distribution of gliomas, the number of tumors is substantially higher in the frontal and temporal lobes than in other lobes (23).
According to a study of three major cancer registries, the Los Angeles County Cancer Surveillance Program (LAC), the California Cancer Registry (CCR), and the National Cancer Institute's Surveillance, Epidemiology, and End Results (SEER) program, data demonstrated increased GBM numbers in the frontal lobe, temporal lobe, and cerebellum, despite a decrease (24).
These results show a significant difference in GBM distribution among anatomical brain sites in our study group compared with that in the United States. These differences require further research and investigation to determine the etiology of this difference in the anatomical site distribution of glioblastoma multiforme.
Limitations
This study had several limitations. First, it was a single-center, retrospective analysis, which may limit the generalizability of the findings to other regions or populations. Second, nine cases lacked histo-pathological confirmation, potentially affecting the accuracy of the tumor classification. Third, the sample size for certain tumor subtypes, such as oligodendroglioma, ependymoma, and medulloblastoma, was very small, limiting the statistical power of subgroup analyses. Additionally, molecular and genetic profiling of tumors is not available, which could provide further insights into prognosis and tumor behavior. Finally, geographical and referral biases may have influenced the distribution of cases, as patients from rural areas or neighboring provinces might have been underrepresented.
Recommendation
The molecular diagnostics were unavailable and this one of the limitations, which is significantly limits comparability with modern literature as the 2016 WHO classification (incorporated molecular para-meters) and the 2021 WHO classification (redefined adult-type diffuse gliomas). Second, the interpretation of anatomical distribution cautiously acknowledges the small sample size and old potential classification bias.
Conclusion
In this study of 95 histologically confirmed brain glioma cases in Sulaymaniyah Province, high-grade gliomas, particularly glioblastoma, were more prevalent than low-grade gliomas, accounting for 64.2% of cases. The mean age at diagnosis increased with the tumor grade, indicating that older patients were more likely to develop high-grade tumors. Tumor location is also an important factor, with high-grade gliomas more frequently observed in the parietotemporal and frontal lobes, whereas low-grade tumors are predominantly located in the occipital lobe. Sex did not significantly influence tumor grade. Multivariate analysis identified age and parietotemporal location as independent predictors of high-grade gliomas, highlighting their potential roles in risk stratification and guiding clinical management strategies.
Author’s Contributions
Shaiban Almukhtar; Conceptualization; Data Curation; Project administration; Resources; Writing – review & editing. Zahraa Hameedi; Conceptualization; Project administration; Writing – review & editing. Mustafa Al-tamer; Conceptualization; Data Curation; Investigation; Methodology; Project administration; Resources; Software; Writing – original draft and writing – review and editing. Anwar Sheikha; Conceptualization; Data Curation; Investigation; Supervisor; Resources; Writing – original draft; Writing – review and editing. Hazha Ameen; Conceptualization; Data Curation; Supervisor; Investigation; Methodology. Ahmed Alshewered; Conceptualization; Data Curation; Project administration, Resources; Software; Writing, review, and editing.
Conflict of Interests
The authors declare no conflict of interest regarding this article.
Funding: None.
Ethics Approval
This study was approved by the Internal Review Board of the College of Medicine, University of Sulaimani (no. 88 in 2018) and Oncology and Nuclear Medicine Hospital, Mosul Health Directorate, Ministry of Health (no. # 7/90/10223 in Nov 2018).
References
1. David CJ, Theodore LS. Cancer of the Central Nervous System In: DeVita VT, Lawrence TS, Rosenberg SA. (editors). Principles & Practice of Oncology (8th Edition). London: Lippincott Williams & Wilkins. 2008; pp:1967, 2032.
2. Louis DN, Pomeroy SL, Cairncross JG. Focus on central nervous system neoplasia. Cancer Cell. 2002;1(2):125-8.
3. Czernicki T, Zegarska J, Paczek L, Cukrowska B, Grajkowska W, Zajaczkowska A, et al. Gene expression profile as a prognostic factor in high-grade gliomas. Int J Oncol. 2007;30(1):55-64.
4. Roerig P, Nessling M, Radlwimmer B, Joos S, Wrobel G, Schwaenen C, et al. Molecular classification of human gliomas using matrix-based comparative genomic hybridization. Int J Cancer. 2005; 117(1):95-103.
5. Walker EV, Davis FG; CBTR founding affiliates. Malignant primary brain and other central nervous system tumors diagnosed in Canada from 2009 to 2013. Neuro Oncol. 2019;21(3):360-369.
6. Kleihues P, Burger PC, Scheithauer BW. The new WHO classification of brain tumours. Brain Pathol. 1993;3(3):255-68.
7. Chang SM, Parney IF, Huang W, Anderson FA Jr, Asher AL, Bernstein M, et al. Patterns of care for adults with newly diagnosed malignant glioma. JAMA. 2005;293(5):557-64.
8. Jacobs AH, Kracht LW, Gossmann A, Rüger MA, Thomas AV, Thiel A, et al. Imaging in neurooncology. NeuroRx. 2005 Apr;2(2):333-47.
9. Charnley N, West CM, Barnett CM, Brock C, Bydder GM, Glaser M, et al. Early change in glucose metabolic rate measured using FDG-PET in patients with high-grade glioma predicts response to temozolomide but not temozolomide plus radiotherapy. Int J Radiat Oncol Biol Phys. 2006;66(2):331-8.
10. Hentschel SJ, Sawaya R. Optimizing outcomes with maximal surgical resection of malignant gliomas. Cancer Control. 2003; 10(2):109-14.
11. van den Bent MJ, Afra D, de Witte O, Ben Hassel M, Schraub S, Hoang-Xuan K, et al. Long-term efficacy of early versus delayed radiotherapy for low-grade astrocytoma and oligodendroglioma in adults: the EORTC 22845 randomised trial. Lancet. 2005;366(9490): 985-90. Erratum in: Lancet. 2006;367(9525):1818.
12. Karim AB, Afra D, Cornu P, Bleehan N, Schraub S, De Witte O, et al. Randomized trial on the efficacy of radiotherapy for cerebral low-grade glioma in the adult: European Organization for Research and Treatment of Cancer Study 22845 with the Medical Research Council study BRO4: an interim analysis. Int J Radiat Oncol Biol Phys. 2002; 52(2):316-24.
13. Stupp R, Mason WP, van den Bent MJ, Weller M, Fisher B, Taphoorn MJ, et al; European Organisation for Research and Treatment of Cancer Brain Tumor and Radiotherapy Groups; National Cancer Institute of Canada Clinical Trials Group. Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med. 2005;352(10):987-96.
14. Laws ER, Parney IF, Huang W, Anderson F, Morris AM, Asher A, et al. Survival following surgery and prognostic factors for recently diagnosed malignant glioma: data from the Glioma Outcomes Project. J Neurosurg. 2003;99(3):467-73.
15. International Agency for Research on Cancer and Cancer Research UK (2012). World cancer factsheet. Cancer Research UK: London. Global-factsheet-2012-1.pdf
16. Iraqi Cancer Registry. (2012) Iraqi Cancer Board - Ministry of Health, Baghdad. 16 edition.2012; 23. https://moh.gov.iq/upload/881.pdf
17. Iraqi Cancer Registry. (2022) Iraqi Cancer Board - Ministry of Health, Baghdad. 23 edition.2023; 22. https://storage.moh.gov.iq/2024/03 /31/2024_03_31_11983087032_3940351786864953.pdf
18. Ferlay J, Shin HR, Bray F, Forman D, Mathers C, Parkin DM. Estimates of worldwide burden of cancer in 2008: GLOBOCAN 2008. Int J Cancer. 2010;127(12):2893-917.
19. Dolecek TA, Propp JM, Stroup NE, Kruchko C. CBTRUS statistical report: primary brain and central nervous system tumors diagnosed in the United States in 2005-2009. Neuro Oncol. 2012;14 Suppl 5(Suppl 5):v1-49. Erratum in: Neuro Oncol. 2013;15(5):646-7.
20. Ohgaki H. Epidemiology of brain tumors. Methods Mol Biol. 2009; 472:323-42.
21. Ostrom QT, Gittleman H, Fulop J, Liu M, Blanda R, Kromer C, et al. CBTRUS Statistical Report: Primary Brain and Central Nervous System Tumors Diagnosed in the United States in 2008-2012. Neuro Oncol. 2015;17 Suppl 4(Suppl 4):iv1-iv62.
22. McKinney PA. Brain tumours: incidence, survival, and aetiology. J Neurol Neurosurg Psychiatry. 2004;75 Suppl 2(Suppl 2):ii12-7.
23. Larjavaara S, Mäntylä R, Salminen T, Haapasalo H, Raitanen J, Jääskeläinen J, et al. Incidence of gliomas by anatomic location. Neuro Oncol. 2007;9(3):319-25.
24. Zada G, Bond AE, Wang YP, Giannotta SL, Deapen D. Incidence trends in the anatomic location of primary malignant brain tumors in the United States: 1992-2006. World Neurosurg. 2012;77(3-4):518-24.
Full Text Sources:
Abstract:
Views: 54

For Authors
Journal Subscriptions

Dec 2025
Supplements
Instructions for authors
Online submission
Contact
e-ISSN: 2601 - 1700 (online)
ISSN-L: 2559 - 723X
Journal Abbreviation: Surg. Gastroenterol. Oncol.
Surgery, Gastroenterology and Oncology (SGO) is indexed in:
- SCOPUS
- EBSCO
- DOI/Crossref
- Google Scholar
- SCImago
- Harvard Library
- Open Academic Journals Index (OAJI)
Surgery, Gastroenterology and Oncology (SGO) is an open-access, peer-reviewed online journal published by Celsius Publishing House. The journal allows readers to read, download, copy, distribute, print, search, or link to the full text of its articles.
Time to first editorial decision: 25 days
Rejection rate: 61%
CiteScore: 0.2

Meetings and Courses in 2025
Meetings and Courses in 2024
Meetings and Courses in 2023
Meetings and Courses in 2022
Meetings and Courses in 2021
Meetings and Courses in 2020
Meetings and Courses in 2019
Verona expert meeting 2019

Surgery, Gastroenterology and Oncology applies the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits readers to copy and redistribute the material in any medium or format, remix, adapt, build upon the published works non-commercially, and license the derivative works on different terms, provided the original material is properly cited and the use is non-commercial. Please see: https://creativecommons.org/licenses/by-nc/4.0/
Publisher’s Note:
The opinions, statements, and data contained in article are solely those of the authors and not of Surgery, Gastroenterology and Oncology journal or the editors. Publisher and the editors disclaim responsibility for any damage resulting from any ideas, instructions, methods, or products referred to in the content.
 IASGO Society News
IASGO Society News