Surgery, Gastroenterology and Oncology

|
|
Background: Gallstone disease (GSD) is a prevalent illness worldwide. Study aimed to investigate the association of Xbal (rs 693) G/A and TLR4 – rs 4986791 C/T genes with gallbladder disease.
Material and Methods: Over the course of three months, from October 2020 to December 2021, samples were taken from fifty healthy persons and one hundred patients with a diagnosis of gall bladder infection from Al. Hilla Teaching Hospital. The patients' ages ranged from fifteen to seventy-one years. TLR4 –rs 4986791 C/T and Xbal (rs 693) G/A genes were found using the tetra arms approach in both sick and healthy groups.
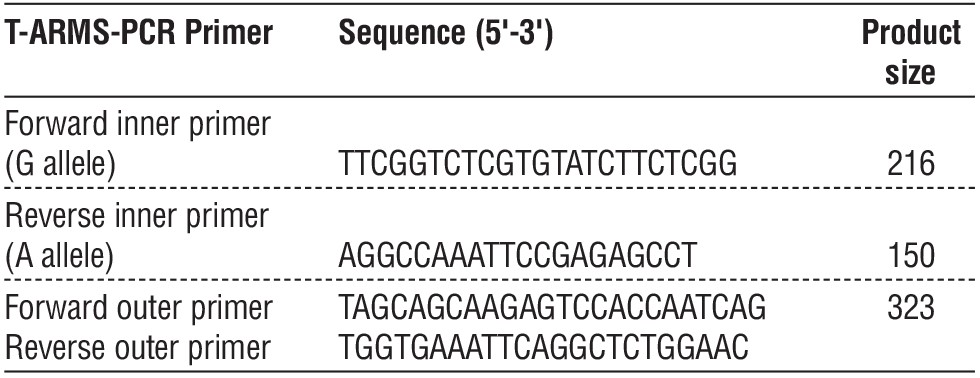
Results: In the event that polymorphisms exist in TLR4 rs4986791 C/T, T-ARMS-PCR was employed to genotype the variant. Based on whether the polymorphism is present or not, the TLR4 rs4986791 C/T distribution is as follows: In GG wild homozygous, the T-ARMS-PCR product only contains the C allele at 295 bp. For the lane (TT) mutant type homozygote, the 219 bp T-ARMS-PCR product showed only the T allele; for the heterozygote (CT), the C and T alleles were detectable in the 295 bp and 219 bp T-ARMS-PCR product. Using T-ARMS-PCR, the outer internal control at 468 bp was identified in order to genotype the Xbl rs693 G/A in the presence of Xbl rs693 G/A polymorphisms. Distribution of the Xbl rs693 G/A in the following categories according to the existence or absence of polymorphism: the (GG) wild type homozygote only showed the G allele, as evidenced by a 216 bp T-ARMS-PCR result. The
heterozygote (GA) at 216 bp and 150 bp displayed both the G and A allele, however the lane (AA) mutant type homozygote only displayed the A allele at 150 bp, according to T-ARMS-PCR. The external internal control was found in the 323 bp T-ARMS-PCR product.
Conclusion: Patients with GD may benefit from early detection and alertness due to the potential power of the APOB rs693 polymorphism as a predictor of GD risk. This makes it useful for the goal of detection in clinics. TLR4 and gallbladder disease, however, do not connect.
INTRODUCTION
Up to 15% of Americans suffer from gallbladder disease (GD), which is a relatively common ailment that has a substantial health care cost in the country (1,2). Over two times as many women as men will experience GD during their lives (3). and GD affects them more frequently than males (4). According to recent data from ultrasonography surveys, ethnicity is a recognized risk factor for GD; especially, Hispanic individuals with a background in central and south America have the greatest rate of the disease (2,5). A high incidence of GD is also seen in the north Indian population, impacting 29.5% of men and 64.1% of women (5). Conversely, those of East-South Asian (China, Japan, India, and Thailand) and African American (African American) heritage exhibit a decreased prevalence of GD development (6).
In addition to race, aging, being in a particular sex group, eating a hypercaloric diet heavy in carbo-hydrates and low in fiber, and other characteristics are risk factors for the development of GD. Obesity is a significant factor in the development of GD. Hepatic cholesterol buildup, intestinal bile acid absorption, bile acid production, inflammation, and stasis are other factors that affect the start of GD. There is mounting evidence that genetic factors play a major role in the development of GD (3,7). The low risk of complications suggests that laparoscopic correction (LC) is a safe procedure that can be performed on a day case basis with high patient satisfaction and a low readmission rate. This is as a result of the fact that most problems were minor and easily handled. Additionally, LC will decrease both the rate of bed occupancy and the overall burden on hospitals (8). Ultimately, the same-day diagnostic and patient care can be provided more quickly thanks to the microwave approach's faster turnaround time than the previous method. For small biopsies, a microwave might be a better option than a larger one. The two main drawbacks of using the micro-wave approach in high throughput histopathology facilities continue to be its lack of automation and the need for additional personnel (9).
Because it is believed to affect lipid metabolism and composition, the apolipoprotein B (APOB) gene is essential for the development of GD (10). The APOB gene's exon 26 has the synonymous variation XbaI polymorphism site (rs693) (11). Synonymous single-nucleotide polymorphisms (SNPs) are known to be considered spurious occurrences under no to little selection. SNPs are defined as nucleotide changes at synonymous codons that preserve the encoded aminoacid (12). Rather of being distributed haphazardly across genes, synonymous SNPs are more likely to target conserved areas (13). Furthermore, synonymous mutations are thought to be the primary cause of several diseases' mutations and have a significant role in disease penetrance, accounting for a higher percentage of somatic mutations seen in human pathologies (14), for instance, GD. Numerous investigations have looked at the connection between this polymorphism and GD, but the results haven't been entirely obvious (15). The TLR4 gene is found on chromosome 9 and has three exons. At exon 3, two non-synonymous SNPs (+1196C/T rs4986791) and Asp299Gly (Thr399Ile), respectively, replace these positions. A decrease in ligand recognition, protein interaction, and lipopoly-saccharide response may arise from Asp299Gly alterations, which modify the extracellular region of TLR4's normal structure (16).
TLR4 is a well-known member of the TLR family, which includes both immunological and non-immune cells. Immune cell TLR4 signaling influences a variety of immune response mechanisms, including CD8+ T-cell cytotoxicity, antigen presentation, and dendritic cell (DC) maturation, all of which are essential components of anti-tumor immunity (17). Since the Tlr4 gene's coding sequence contains 15 polymorphisms, it is highly polymorphic (18). Asp299Gly and Thr399Ile are two co-segregating SNPs in this gene among its numerous SNPs. Many studies have looked into the relationship between these SNPs and the chance of developing various cancers, including leukemia (23), breast cancer (19), gastric cancer (20), prostate cancer (21), hepatocellular carcinoma (22), colorectal cancer (26), gall bladder cancer (24), and leukemia (23). This study aimed to investigate the association of Xbal (rs 693) G/A and TLR4 – rs 4986791 C/T genes with gallbladder disease.
Table 1 - The Tetra-ARMS-PCR Primers for Xbl rs693 G/A polymorphisms with their sequence and amplicon size
MATERIALS AND METHODS
Sample Collection
Between October 2023 and June 2024, a case-control study was conducted in Babylon, Iraq. The 100 gallbladder patients from Al. Hila Teaching Hospital, whose ages ranged from 15 to 71 years, were divided into 50 controls for the study. The researcher used a questionnaire form she had created during an interview with these patients. For Xbal and TLR4 genotyping polymorphisms, the final three milliliters were placed in an EDTA tube.
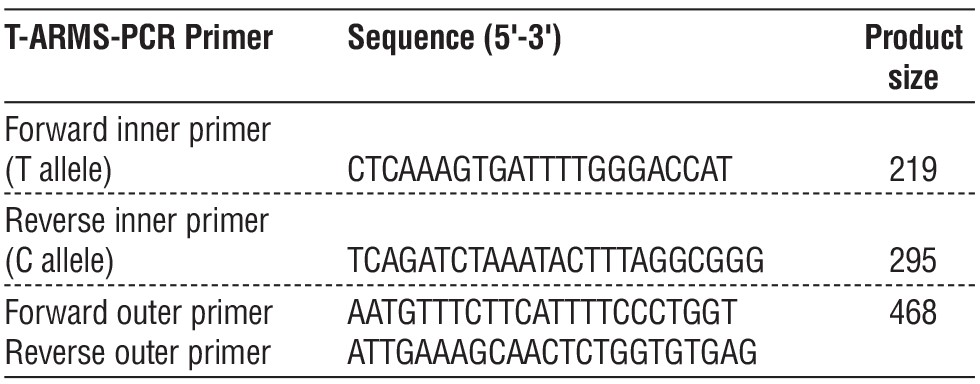
Table 2 - The Tetra-ARMS-PCR Primers for TLR4 rs4986791 C/T gene polymorphisms gene polymorphism with their sequence and amplicon size
Genotyping of Xbal and TLR4 Polymorphisms
In compliance with company guidelines, genomic DNA was extracted from blood samples using a gSYAN DNA kit extraction kit (frozen blood) from Geneaid, USA.ARMS-PCR, or amplification-refractory mutation system PCR, is a rapid and simple method for geno-typing Xbal and TLR4. Primers for Xbal and TLR4 genotyping are included in tables 1 and 2.
Table 3 - All PCR thermocycler conditions were done for genes (TLR4 rs4986791 C/T and Xbl rs693 G/A
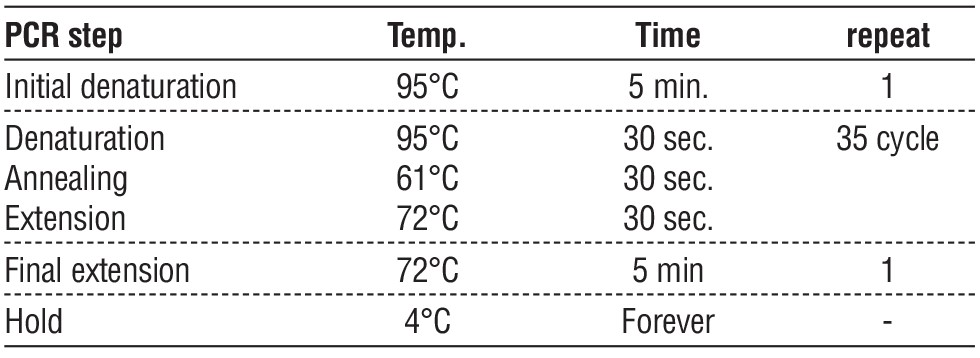
Thermo cycling for TLR4 rs4986791 C/T and Xbl rs693 G/A was performed under settings that PCR conditions in table 3.
Statistical Analysis
The patients' and the control group's genotype distributions were compared using the chi-square test. Less than 0.05 was the threshold for a statistically significant p-value.
RESULTS
Genotype of TLR4 rs4986791 C/T by Arms Methods T-ARMS-PCR was used to genotype TLR4 rs4986791 C/T in the presence of polymorphisms in TLR4 rs4986791 C/T. The distribution of TLR4 rs4986791 C/T in groups according to the presence or absence of the polymorphism is as follows: only the C allele at 295 bp in the T-ARMS-PCR product is present in GG wild homozygous. Only the T allele was visible in the 219 bp T-ARMS-PCR product for the lane (TT) mutant type homozygote, while the
C and T alleles were seen in the 295 bp and 219 bp T-ARMS-PCR product for the heterozygote (CT).
Fig. 1 shows that the outer internal control was detected at 468 bps.
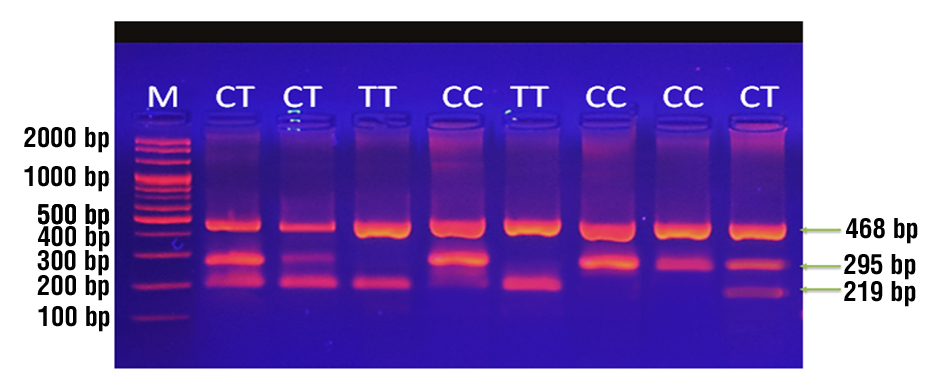
Figure 1 - Analysis of the TLR4-rs4986791 product using T-ARMS-PCR An picture of agarose gel electrophoresis showed the polymorphism of the C/T gene. There the marker, M (100-2000 bp), is found. The lane (CC) wild type homozygote's 295 kb T-ARMS-PCR result only showed the C allele. For the lane (TT) mutant type homozygote, the 219 bp T-ARMS-PCR product showed only the T allele; for the heterozygote (CT), the C and T alleles were detectable in the 295 bp and 219 bp T-ARMS-PCR product. The outer internal control was found in the T-ARMS-PCR product at 468 bps.
Depending on whether the polymorphism is present or not, the genotyping result for Xbl rs693 G/A in the presence of Xbl rs693 G/A polymorphisms by T-ARMS-PCR determines the distribution of Xbl rs693 G/A in groups: the 216 kb T-ARMS-PCR result for the (GG) wild type homozygote only revealed the G allele. The lane (GA) heterozygote had both the G and A alleles at 216 bp and 150 bp, but the lane (AA) mutant type homozygote only had a single allele at 150 bp.
T-ARMS-PCR product, 323 bp, showed the outer internal control (fig. 2).
Association of TLR4 rs4986791 C/T gene SNPs
In this case-control study, we looked at the distribution of alleles and genotype frequency of TLR4 SNPs genotypes (rs4986791 C/T) among Iraqi patients with gall bladder disease and healthy controls. The genotype and allele frequency patterns for the (rs4986791 C/T) SNPs are shown in table 4. When comparing genotypes TT and CC in gall bladder patients to control groups, we discovered no significant changes (p > 0.4 and p > 0.1, respectively) in the case of (rs4986791 C/T) SNPs. No significant differences were seen with heterozygote CT, p≤0.06. While the frequency of CT genotype in the control group was 30%, the frequency in the gall bladder was 48.0%, with an odd ratio of 2.154 (0.948 - 4.894). The gall bladder's estimated TT genotype frequency was (20%), whereas the control group's frequency was (14%). The odd ratio was 1.535 (0.533 - 4.422). The frequency of the TLR4 SNP alleles and genotypes (rs4986791 C/T) is displayed in table 4. The geno-typing results for the heterozygote mutant type TT were as follows: the genotype for CT was 2.154 (0.948 - 4.894) P = 0.06), and the genotype for TT was 1.535 (0.533 - 4.422) and p = 0.4.
Table 5 shows that when comparing female gall bladder patients to male groups, there were no significant differences in genotypes TT and CC (p > 0.5 and p > 0.9, respectively). With heterozygote CT, no significant differences were seen (p ≤ 0.3). In the female gall bladder group, the frequency of the CT genotype was 84.0%, with an odd ratio of 1.467 (0.464 - 4.638). On the other hand, 42% of the male group had the CT genotype frequency.In the female group with gall bladders, the estimated TT genotype frequencies were 26%, whereas the similar numbers in the male group were 26%. 0.539 (0.133 - 2.183) was the odd ratio.

Association of Xbal (rs 693) G/A Gene SNPs
Table 6 reports the genotype and allele frequency distributions for the (rs 693 )G/A) SNPs. Regarding the (rs 693)G/A) SNPs, we discovered that, when compared to the control groups, patients with gall bladders had genotypes AA and GG that differed significantly (p=0.01* and p=0.005**, respectively). Heterozygote GA showed no significant changes (p=0.3). In the gall bladder, the prevalence of the GA genotype was 22.0%, with an odd ratio of 1.732 (0.611 - 4.912), compared to 14% in the control group. The estimated frequencies of AA genotype in the gall bladder were (32%), whereas the control group had a frequency of AA genotype of only 12%. The odd ratio was 3.451 (1.220 – 9.759).
Table 6 - Numbers and percentage frequencies of Xbal (rs 693) G/A genotypes and their Hardy-Weinberg equilibrium (HWE) in gall bladder patients compared with control groups

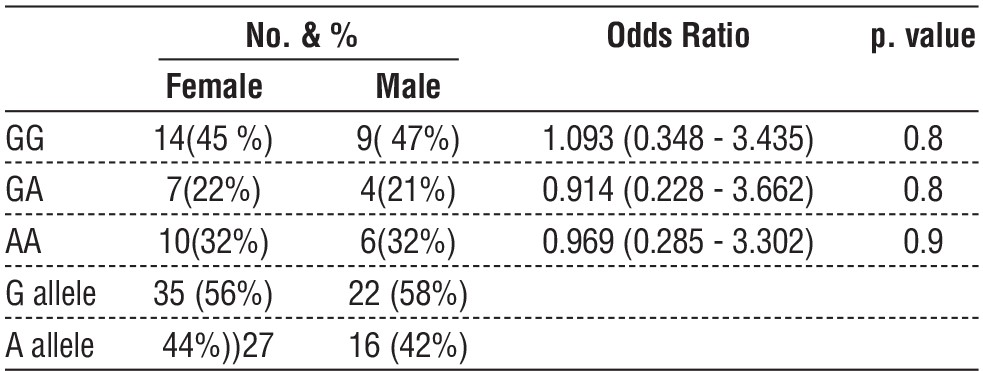
In the instance of (rs 693) G/A) SNPs, table 7 demonstrated that no significant differences were observed between genotypes AA and GG in female gall bladder patients when compared to male groups (p=0.9 and p=0.8, respectively). Heterozygote GA did not demonstrate any significant differences either (p= 0.8). When compared to the male group, the frequency of the GA genotype was (21%), but the frequency in the gall bladder female group was 22.0% and the odd ratio was 0.914 (0.228 - 3.662). An odd ratio of 0.969 (0.285 - 3.302) was found for the gall bladder female group, and the predicted frequencies of the AA genotype in that group were (32%).
Table 7 - Numbers and percentage frequencies of Xbal (rs 693) G/A genotypes and their Hardy-Weinberg equilibrium (HWE) in gall bladder patient's male and female
DISCUSSION
TLRs are members of the family of pattern-recognition receptors (PRRs), which are expressed by antigen-presenting cells including T and dendritic cells, among others. Following their activation by pathogen-associated chemicals, TLRs transduce signals through several intracellular pathways, which in turn activate transcription factors such as NF-κB, AP-1, and interferon regulatory factors (IRFs). Furthermore, inflammatory reactions are brought on by these transcription factors' production of type I interferon and inflammatory cytokines (27,28). The phagocytosis process and other host defense systems, like pattern recognition in microbial infections, depend on TLRs. Receptors that identify conserved molecular patterns that are expressed by infectious diseases are known as pattern recognition receptors, or PRRs for short. They participate in the mediation of transcription factor activation, including NF-κB and pro-inflammatory cytokines (25). This causes inflammation. According to a study (29), polymorphisms in these TLR genes are not expected to be associated with an increased risk of bladder cancer. Additionally, there was no correlation found between the TLR3, 4, and 9 genes and bladder cancer risk. The investigation's conclusions support that assessment. Ethnically diverse populations' functional studies may provide light on changes connected to the onset of bladder cancer and changes brought on by the disease.
Several genes have been associated with an increased risk of GD thus far, including apolipoprotein E, mucin-like protocadherin, and ATP binding cassette subfamily G member 8 (30,31). Moreover, a growing body of research is associating the APOB rs693 polymorphism with a higher risk of GD. Because so few samples were utilized in each study, the findings from each one could not be entirely trustworthy. Dixit et al.'s study comprised 322 healthy controls and 214 GD patients; the rs693 polymorphism may not be connected to a higher incidence of GD (31,32). imply that people who have the A-allele or AA genotype of the rs693 polymorphism may be more susceptible to GD. The Asian population (OR: 1.58, 95 % CI: 1.48–2.84 in the heterozygote model), the control group's hospital-based source (OR: 1.79, 95 % CI: 1.13–2.84 in the dominant model), and the group as a whole (OR: 1.40, 95 % CI: 1.05–1.87) all showed a significant correlation with the rs693 polymorphism and increased risk of GD. shows the possible connection between the GD risk and the APOB rs693 polymorphism, offering a helpful marker for identification in future large-scale treatment trials.
CONCLUSION
According to our current meta-analysis, the APOB rs693 polymorphism may be a strong predictor of GD risk. This means that it can be used as a diagnostic tool in clinics to identify and warn patients with GD early on. However, TLR4 is not linked to gallbladder illness.
Acknowledgements
We thank Dr. Majid Moosa Alkhafaji (Consultant surgeon CABS ) for his help to collect gall bladder tissue.
Conflict of interest
All authors declared there is no any conflict of interesting.
Funding
Self support.
Ethical Approval
This study approved by Ethical committee of Department of Biology (No.33 in 2023).
REFERENCES
-
Di Ciaula A, Wang DQ, Portincasa P. Cholesterol cholelithiasis: part of a systemic metabolic disease, prone to primary prevention. Expert Rev Gastroenterol Hepatol. 2019;13(2):157–71.
-
Stinton LM, Shaffer EA. Epidemiology of gallbladder disease: cholelithiasis and cancer. Gut Liver. 2012;6(2):172–87.
-
Littlefield A, Lenahan C. Cholelithiasis: presentation and management. J Midwifery Women Health. 2019;64(3):289–97.
-
Shaffer EA. Gallstone disease: epidemiology of gallbladder stone disease. Best Pract Res Clin Gastroenterol. 2006;20(6):981-96.
-
Figueiredo JC, Haiman C, Porcel J, Buxbaum J, Stram D, Tambe N, et al. Sex and ethnic/racial-specific risk factors for gallbladder disease. BMC Gastroenterol. 2017;17(1):153.
-
Gurusamy KS, Davidson BR. Gallstones. BMJ. 2014;348:g2669.
-
Gutt C, Schläfer S, Lammert F. The treatment of gallstone disease. Deutsches Arzteblatt Int. 2020;117(9):148–58.
-
Khalid Arqam MM, Musa DH, Mohammed SA. Laparoscopic cholecystectomy as a day-case surgery in Azadi Teaching Hospital, Duhok. Med J Babylon 2023;20:531-5.
-
Shirbhate PN, Deshmukh AV, Shivkumar VB. Rapid microwave tissue processing and staining method using a kitchen microwave oven in histopathology laboratory: A comparative study with routine histoprocessing method. Med J Babylon 2022;19:383-90.
-
Feingold KR, ed. Introduction to Lipids and Lipoproteins. In: Feingold KR, Anawalt B, Boyce A, Chrousos G, de Herder WW, Dungan K, Grossman A, Hershman JM, Hofland HJ, Kaltsas G, et al., editors. In: Endotext [Internet]. South Dartmouth (MA): MDText.com, Inc.; 2000.
-
Benn M, Nordestgaard BG, Jensen JS, Grande P, Sillesen H, Tybjaerg-Hansen A. Polymorphism in APOB associated with increased low-density lipoprotein levels in both genders in the general population. J Clin Endocrinol Metab. 2005;90(10):5797–803.
-
Rauscher R, Ignatova Z. Timing during translation matters: synonymous mutations in human pathologies influence protein folding and function. Biochem Soc Transact. 2018;46(4):937–44.
-
Zheng S, Kim H, Verhaak RGW. Silent mutations make some noise. Cell. 2014;156(6):1129–31.
-
Supek F, Miñana B, Valcárcel J, Gabaldón T, Lehner B. Synonymous mutations frequently act as driver mutations in human cancers. Cell. 2014;156(6):1324–35.
-
Báez S, Tsuchiya Y, Calvo A, Pruyas M, Nakamura K, Kiyohara C, Oyama M, Yamamoto M. Genetic variants involved in gallstone formation and capsaicin metabolism, and the risk of gallbladder cancer in Chilean women. World J Gastroenterol. 2010;16(3):372–8.
-
Arbour NC, Lorenz E, Schutte BC, Zabner J, Kline JN, Jones M, et al. (2000) TLR4 mutations are associated with endotoxin hyporesponsiveness in humans. Nat Genet. 2000;25(2):187-91.
-
Schreibelt G, Tel J, Sliepen KH, Benitez-Ribas D, Figdor CG, Adema GJ, et al. Toll-like receptor expression and function in human dendritic cell subsets: implications for dendritic cell-based anti-cancer immunotherapy. Cancer immunology, immunotherapy. 2010;59:1573-82.
-
Schröder NW, Schumann RR. Single nucleotide polymorphisms of Toll-like receptors and susceptibility to infectious disease. The Lancet infectious diseases. 2005;5(3):156-64.
-
Theodoropoulos GE, Saridakis V, Karantanos T, Michalopoulos NV, Zagouri F, Kontogianni P, et al. Toll-like receptors gene poly-morphisms may confer increased susceptibility to breast cancer development. The Breast. 2012 Aug 1;21(4):534-8.
-
https://doi.org/10.1016/j.breast.2012.04.001
-
de Oliveira JG, Silva AE. Polymorphisms of the TLR2 and TLR4 genes are associated with risk of gastric cancer in a Brazilian population. World journal of gastroenterology: WJG. 2012;18(11): 1235.
-
Almayah QS. Study of Association of Single Nucleotide Polymorphism in Toll Like Receptor and CXCL8 Genes, and Trichomonasvaginalis Infection with Prostate Cancer. Ph. D. thesis. University of Babylon/College of Medicine. 2013.
-
Agúndez JA, García-Martín E, Devesa MJ, Carballo M, Martínez C, Lee-Brunner A, et al. Polymorphism of the TLR4 gene reduces the risk of hepatitis C virus-induced hepatocellular carcinoma. Oncology. 2012;82(1):35-40.
-
Miedema KG, Tissing WJ, Te Poele EM, Kamps WA, Alizadeh BZ, Kerkhof M, et al. Polymorphisms in the TLR6 gene associated with the inverse association between childhood acute lymphoblastic leukemia and atopic disease. Leukemia. 2012;26(6):1203-10.
-
Srivastava K, Srivastava A, Kumar A, Mittal B. Significant association between toll-like receptor gene polymorphisms and gallbladder
-
cancer. Liver International. 2010;30(7):1067-72.
-
Pandey S, Mittal RD, Srivastava M, Srivastava K, Singh S, Srivastava S, et al. Impact of Toll-like receptors (TLR) 2 (- 196 to - 174 del) and TLR 4 (Asp299Gly, Thr399Ile) in cervical cancer susceptibility in North Indian women. Gynecol Oncol. 2009;114(3):501-5.
-
Landi S, Gemignani F, Bottari F, Gioia-Patricola L, Guino E, Cambray M, et al. Polymorphisms within inflammatory genes and colorectal cancer. J Negat Results Biomed. 2006;5:15.
-
Jemal A, Siegel R, Xu J, Ward E. Cancer statistics, 2010. CA Cancer J Clin. 2010;60(5):277-300.
-
Shariat SF, Sfakianos JP, Droller MJ, Karakiewicz PI, Meryn S, Bochner BH. The effect of age and gender on bladder cancer: a critical review of the literature. BJU international. 2010;105(3): 300-8.
-
Singh V, Srivastava N, Kapoor R, Mittal RD. Single-nucleotide polymorphisms in genes encoding toll-like receptor-2,-3,-4, and-9 in a case–control study with bladder cancer susceptibility in a North Indian population. Archives of medical research. 2013; 44(1):54-61.
-
Chuang SC, Hsi E, Wang SN, Yu ML, Lee KT, Juo SH. Polymorphism at the mucin-like protocadherin gene influences susceptibility to gallstone disease. Clin Chim Acta. 2011;412(23-24):2089-93.
-
Li L, Qiao X, Wang X, Liu D, Xue Q, Han L, et al. The association between apolipoprotein E and gallstone disease: an updated meta-analysis. BMC Med Genet. 2019;20(1):109.
-
Dixit M, Srivastava A, Choudhuri G, Mittal B. Higher alleles of apolipoprotein B gene 3’ VNTR: Risk for gallstone disease. Indian J Clin Biochem: IJCB. 2008;23(2):123-9.
-
Zhu H, Yu L, Feng L. Association of apolipoprotein B XbaI (rs693) polymorphism and gallstone disease risk based on a comprehensive analysis. Genes Environ. 2021;43(1):17.
Full Text Sources:
Abstract:
Views: 1533

For Authors
Journal Subscriptions

Dec 2025
Supplements
Instructions for authors
Online submission
Contact
e-ISSN: 2601 - 1700 (online)
ISSN-L: 2559 - 723X
Journal Abbreviation: Surg. Gastroenterol. Oncol.
Surgery, Gastroenterology and Oncology (SGO) is indexed in:
- SCOPUS
- EBSCO
- DOI/Crossref
- Google Scholar
- SCImago
- Harvard Library
- Open Academic Journals Index (OAJI)
Surgery, Gastroenterology and Oncology (SGO) is an open-access, peer-reviewed online journal published by Celsius Publishing House. The journal allows readers to read, download, copy, distribute, print, search, or link to the full text of its articles.
Time to first editorial decision: 25 days
Rejection rate: 61%
CiteScore: 0.2

Meetings and Courses in 2025
Meetings and Courses in 2024
Meetings and Courses in 2023
Meetings and Courses in 2022
Meetings and Courses in 2021
Meetings and Courses in 2020
Meetings and Courses in 2019
Verona expert meeting 2019

Surgery, Gastroenterology and Oncology applies the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits readers to copy and redistribute the material in any medium or format, remix, adapt, build upon the published works non-commercially, and license the derivative works on different terms, provided the original material is properly cited and the use is non-commercial. Please see: https://creativecommons.org/licenses/by-nc/4.0/
Publisher’s Note:
The opinions, statements, and data contained in article are solely those of the authors and not of Surgery, Gastroenterology and Oncology journal or the editors. Publisher and the editors disclaim responsibility for any damage resulting from any ideas, instructions, methods, or products referred to in the content.
 IASGO Society News
IASGO Society News