Surgery, Gastroenterology and Oncology

|
|
Background: Among the most common malignancies worldwide, bladder cancer is among the most common. Iraq has seen a dramatic spike in the number of reported cases and deaths from this illness over the last few decades. Aim. Examining interleukin-10 expression in human bladder cancer with the main objective of discovering relevant connections between it and clinic-pathological variables is the fundamental purpose of this study. Using this marker to differentiate between basal and luminal bladder tumors is another goal.
Material and Methods: We hope that by studying interleukin-10 expression in bladder cancer, we may better understand the biological activity of this disease in connection to grade and muscle invasiveness, and so improve diagnostic and prognostic tools. This research looks at how interleukin-10 expression in bladder cancer may be detected using immunohistochemistry. The AL Hussein Teaching Hospital in the city of AL-Nasiriya in southern Iraq was the site of the random sample collection. One hundred tissues were used in the study; seventy of these were bladder cancer tissues and thirty were benign control tissues.
Results: Fifty out of seventy samples showed positive interleukin-10 expression. Compared to low-grade bladder cancer, which had an interleukin-10 expression level of 59.26%, high-grade bladder cancer had an enhanced level of 79.07%. Furthermore, interleukin-10 expression is significantly elevated in the T2 stage (76.22%), followed by Ta (54.89%), T1 (66.67%), T3 (two instances), and T4 (one case). In instances where muscle invasion has occurred, interleukin-10 expression is high (80.95%), whereas in circumstances where no such invasion has occurred, it is low (57.14%).
Conclusion: In conclusion. One way to measure how aggressive bladder cancer is is by looking for interleukin-10. In light of the paucity of prior work on the subject in Iraq, this study represents a significant step forward. More research on the function of interleukin-10 in bladder cancer biology is urgently required.
INTRODUCTION
The most common kind of tumor in the urinary system is bladder cancer, which requires a battery of immunological testing and a battery of other procedures to diagnose. Even with all the new medical technology, a biopsy of the bladder tissue is still necessary for a proper diagnosis (1). Cancers of the bladder may be classified into two groups based on how deeply they penetrate the bladder wall: muscle-invasive bladder carcinoma (MIBC) and non-muscle-invasive bladder cancer (NMIBC). Invasive carcinomas limited to the lamina propria (stage pT1), carcinoma in situ ("CIS"), non-muscle invasive bladder cancer (NMIBC), or carcinoma in situ (stage pT2) account for almost 70% of all bladder cancers that are first detected. The vast majority of bladder cancers are transitional cell carcinomas (TCCs), which make up 97% of all cases. Squamous cell carcinomas (SCCs) make up 2% of cases, while adenocarcinoma accounts for 1% (3). Bladder cancer ranks #4 among male malignancies and #8 among cancer-related fatalities in the US, claiming the lives of around 12,000 individuals yearly, with an estimated 60000 new-cases diagnosed annually. In 2017, the United States of America had 79,030 instances of bladder cancer, with 16,870 fatalities as a result (4). Iraq ranks fifth globally for bladder cancer incidence, third for men, and tenth for women. In 2010, 866 male cases and 297 female cases of bladder cancer were recorded by the Iraqi Cancer Registry (5). It will be the sixth most common malignancy overall in 2021 with an infection rate of 1,769—1,360 in men and 409 in women. Based on this imbalance, it appears that this illness will affect more males than women (6).
Although systemic cisplatin-based chemotherapy is now recommended for advanced bladder cancer, immunotherapy is emerging as a promising second line of defense when the first line of treatment was unsuccessful in halting the disease. Cells of the myeloid and lymphoid types, including dendritic cells, macrophages, eosinophils, neutrophils, and mast cells, respectively, generate this pleiotropic cytokine, which has extensive anti-inflammatory effects. When stimulated with dsDNA and lipopolysaccharide (LPS), macrophages and myeloid dendritic cells secrete IL-10 in response to MyD88 activation and TRIF-dependent Toll-like Receptor (TLR) pathways, namely TLR3 and TLR4, respectively. In addition, LPS triggers the formation of T regulatory cells by causing tolerogenic dendritic cells (CD11clowCD45RBhigh) to release copious quantities of IL-10 (7,8). An essential component of immunological homeostasis, IL-10 is produced by natural regulatory cells (nTreg) in reaction to IL-2 (9).
The 37 kDa physiologically active protein IL-10 forms a non-covalent homodimer with a molecular mass of 18.5 kDa per unit. We know it to be a three-dimensional domain switcher protein. Two subunits, IL-10R1 and IL-10R2, make up the IL-10 receptor (IL-10R), a class II cytokine receptor (10). An IL-10 secretory state and suppressive action in the anti-tumor immune response may be defining characteristics of a subset of functioning TAMs. Prior research has shown that IL-10+ TAMs help breast and gastric cancer cells avoid the immune system (11).
This study aimed to determine if IL-10 levels were associated with tumor grade and stage in bladder cancer patients by using immunohistochemistry (IHC) to evaluate IL-10 expression. The histological and clinical characteristics of bladder cancer in Iraqis were also intended to be documented.
MATERIAL AND METHODS
Collection of Samples
Two sets of 100 bladder tissue samples were used in this study. One group consisted of 70 samples from individuals with bladder cancer, while the other 30 samples were from healthy bladder lesions. The participants' ages ranged from 20 to 80 years. The materials used in the study were obtained from two locations in Thi-Qar province: Al-Hussein Hospital and a private laboratory. They were stored at room temperature with 10% formaldehyde until histopathological and immunohistochemically processing.
Immunohistochemistry Assays (IHC)
This experiment used the immunohistochemistry approach, following the previously described protocols (12). The initial step in deparaffination is to soak samples in xylene and then rehydrate them using a series of progressively stronger alcohols. After that, any leftover antigens will be removed by heating the slides in a Retrieval Solution. To counteract the tissue's natural peroxidase activity, peroxidase was injected after 20 minutes of room temperature cooling and left on for 10 minutes. After that, for a further ten minutes each time, the slides were washed with phosphate buffered saline. Afterwards, use a rabbit polyclonal antibody to incubate the sections with primary antibodies IL-10 (in a 1:100 ratio). The next day, the slides were immersed in PBS for ten minutes each of three washes. Afterwards, they were incubated at room temperature with a moderate vibrator for 30 minutes after being drenched with anti-mouse labeled polymer-HRP primary anti body. Following that, the slides were immersed in PBS buffer three times, with a five-minute interval between each wash. Following the Dako technique, the slides were incubated at room temperature for 10 minutes after being stained with drops of DAP chromogen. Then, after 10 minutes, the slides were rinsed with distilled water (D.W.). After that, the cell nucleus was brought to light by letting haematoxylin, a counter stain, incubate at room temperature for 2 minutes after being dropped over the slides. The next step was rinsing the slides under running water for two minutes and distilled water for another minute. This process was repeated three times. The next stage was to dry the slides by submerging them in ethanol at concentrations of70,95, and 100% for one minute each. The slides were considered dehydrated after being immersed in xylene for two minutes apiece. After that, 22 X 22 mm slides were placed over the slides and let to dry after DPX drops were applied.
Statistical Analysis
After analysed the data via SPSS, the results are shown as a percentage. Using the Pearson Chi-square test, one may determine whether two sets of qualitative data are significantly different. A p-value of less than 0.05 is considered a statistically significant finding.
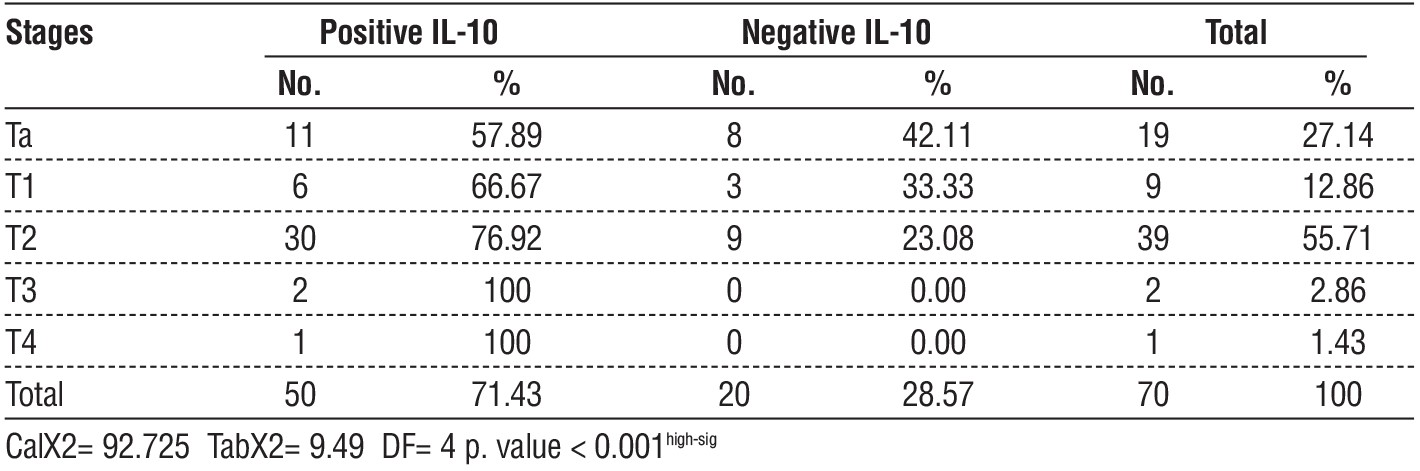
Table 1 - relationship between IL-10 and T staging of bladder cancer
RESULTS
Thirty bladder cancer patients at stage T2 had elevated IL-10 levels (p=0.001), compared to two out of thirty controls who tested positive for the protein. Fifty cases out of 70 exhibited overexpression of IL-10 (71.43%). Referring to the data in table 1, there were eleven instances of stage Ta, six instances of stage T1, two instances of stage T3, and one instance of stage T4. According to the data in the table 2, this study found that low-grade urothelial carcinoma accounted for 16 of the 50 instances of positive IL-10 expression in bladder cancer, whereas high-grade bladder cancer accounted for 34 cases of
high-expression IL-10 (P= 0.02). According to the data in the table 3, this study found that IL-10 (showed fig. 1-3) was overexpressed in 34 out of 50 cases of visive bladder cancer (P=0.01), despite the fact that 16 instances of bladder cancer that did not invade muscle showed underexpression.
Table 2 - relationship between IL-10 and grade tumor of bladder cancer
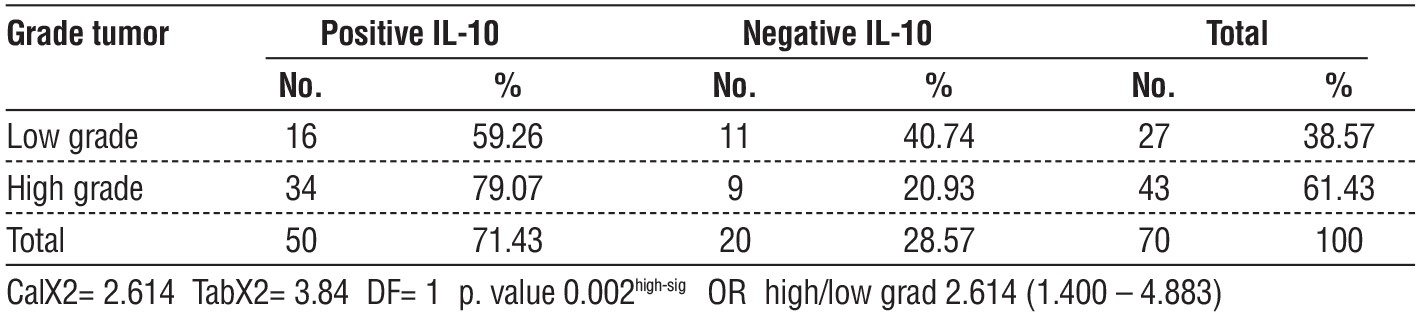
Table 3 - relationship between IL-10 and type of tumor of bladder cancer
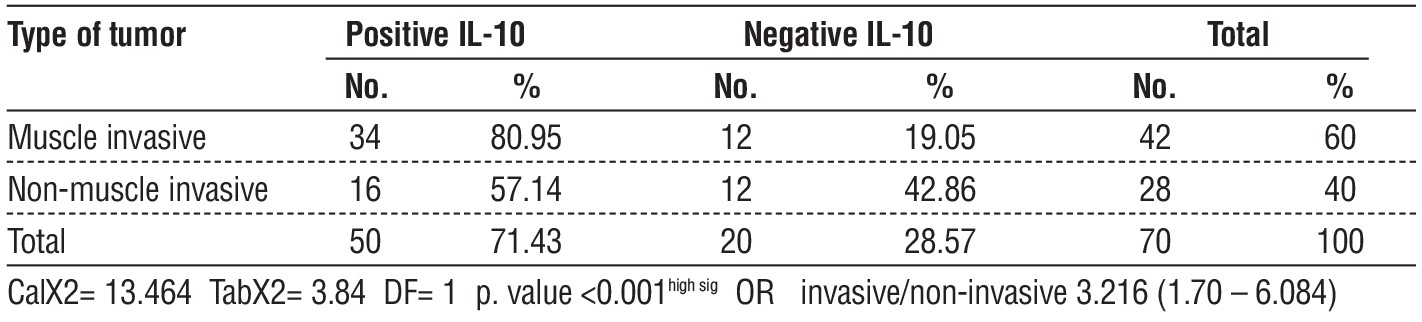
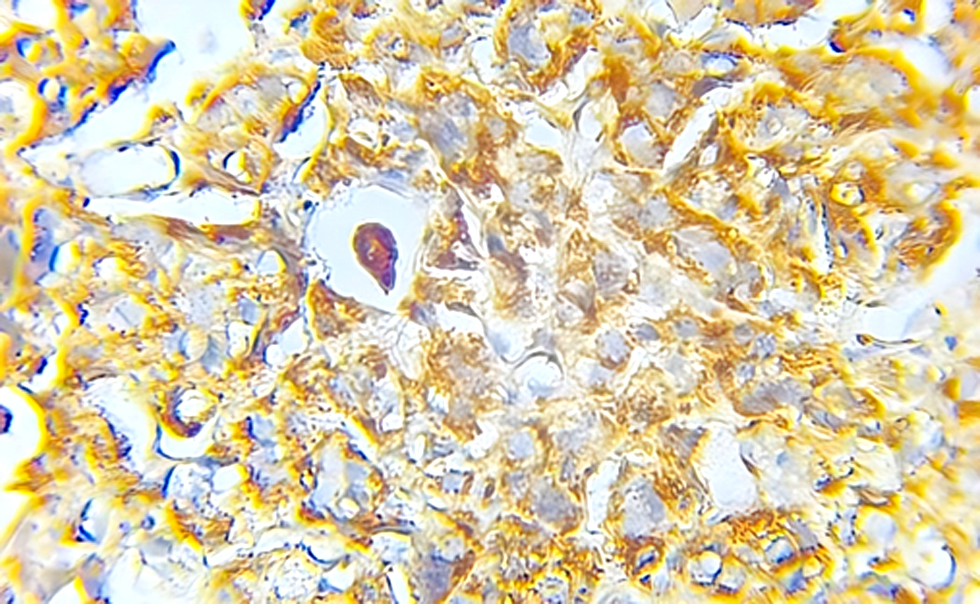
Figure 1 - High-grade urothelial carcinoma, show high expression of IL-10

With an interval from 0 to 8, the sum of PS and IS is called a Total Score (TS). The definition of a positive result for IL-10, which has been confirmed in several major clinical investigations, is TS ≥ 3.
DISCUSSION
Fifty of the seventy bladder cancer cases (71.43%) had IL-10 overexpression, whereas two of the thirty control subjects tested positive for the protein. This study's results proved. A significant level of IL-10 expression is seen in stage T2 (30) patients (p < 0.001). The present analysis identified eleven instances of stage Ta, six cases of stage T1, two cases of stage T3, and one case of stage T4 to be in concordance (13). Patients whose bladder cancer has spread to their lymph nodes have a poor prognosis due to the dramatic increase in the number of regulatory T (Treg) cells (14). For that reason, it is thought that the presence of Treg cells stands alone as a sign of a high likelihood of treatment-induced bladder cancer (14).
Figure 2 - High-grade urothelial carcinoma, show high expression of IL-10
Activated B cells, monocytes, macrophages, and helper T lymphocytes (Th2) are the principal secretors of interleukin (IL)-10, a multifunctional negative regulator. As an immunosuppressant, IL-10 promotes tumor development and immune escape by reducing the involvement of tumor-immune cytokines (15).
Although 34 individuals tested positive for IL-10, none of them were considered to have advanced bladder cancer. Another option is that 16 cases of low-grade urothelial carcinoma tested positive for IL-10, which is statistically significant (13). To get this conclusion, the following procedure may be required: The production of IL-2, TNF-α, and INF-γ is decreased when tumor cells, tumor-associated macrophages, and regulatory T cells in the tumor microenvironment release IL-10. This, in turn, suppresses the proliferation and killing abilities of T cells (16, 17).
Figure 3 - Low-grade urothelial carcinoma, show high expression of IL-10

Tumors are able to grow and spread more easily because IL-10 blocks the antitumor immune response by interacting with immunocytes. By increasing myeloid cell PDL1 expression, IL-10 decreases the antitumor effectiveness of T cells via its interaction with the T cell inhibitory receptor PD1. The current study's findings also indicated that IL-10 might encourage the proliferation of bladder cancer cells. Therefore, it may one day serve as a diagnostic tool for breast cancer and a treatment target for bladder cancer. Since monocytes, Th2 cells, macrophages, and activated B cells might all produce IL-10, more studies using the Th2-trace-labeling method are required to determine its origin (19). In 28 out of 33 cases of muscular bladder cancer (p. value < 0.001), the research found that IL-10 is overexpressed, whereas in 22 out of 37 cases of non-muscle bladder cancer, it is under-expressed. According to the results of the prior research, everyone is in agreement (13).
Increased IL-10 expression in colorectal cancer is linked to worse prognosis, shorter survival time, and increased likelihood of cancer recurrence (Li et al., 2020). Accordingly, IL-10 shows promise as a colorectal cancer prognostic marker. We may also look at how IL-10 affects bladder cancer prognosis (20).
CONCLUSION
Important prognostic factors associated with IL-10 expression include stage and grade. The expression of IL-10 was discovered to be significantly associated with tumor grade and invasion degree, suggesting that it might be a valuable prognostic marker for urothelial carcinoma. IL-10 immuno-expression is greater in invasive urothelial carcinoma patients compared to those without invasive muscle, and this is due to the fact that these patients exhibit a greater number of tumor features.
Conflict of interest
The author declare that him have no conflict of interest.
Funding
No funding sources.
Ethical Approval
This study was approved by Ethical Reviewer Board Committee of Department of Histopathology, College of Medicine, University of Thi-Qar (No. 78 in 2023).
REFERENCES
1. Powles T, Kockx M, Rodriguez-Vida A, Duran I, Crabb SJ, Van Der Heijden MS, et al. Clinical efficacy and biomarker analysis of neo-adjuvant atezolizumab in operable urothelial carcinoma in the
ABACUS trial. Nat Med. 2019;25(11):1706-1714.
2. Sjödahl G, Lauss M, Lövgren K, Chebil G, Gudjonsson S, Veerla S, et al. A molecular taxonomy for urothelial carcinoma. Clin Cancer Res. 2012;18(12):3377-86.
3. Beukers W, Kandimalla R, van Houwelingen D, Kovacic H, Chin JF, Lingsma HF, et al. The use of molecular analyses in voided urine for the assessment of patients with hematuria. PLoS One. 2013;8(10): e77657.
4. Siegel RL, Miller KD, Jemal A. CA Cancer J Clin. 2018;68(1):7-30.
5. Iraqi Cancer Registry (2011) Baghdad-Iraq. https://ghdx.healthdata.org/record/iraq-cancer-registry-2011
6. Iraqi Cancer Registry (2021) Baghdad-Iraq. https://ghdx.healthdata.org/record/iraq-cancer-registry-2011
7. Boonstra A, Rajsbaum R, Holman M, Marques R, Asselin-Paturel C, Pereira JP, et al. Macrophages and myeloid dendritic cells, but not plasmacytoid dendritic cells, produce IL-10 in response to MyD88-and TRIF-dependent TLR signals, and TLR-independent signals. J Immunol. 2006;177(11):7551-8.
8. Comi M, Amodio G, Gregori S. Interleukin-10-producing DC-10 is a unique tool to promote tolerance via antigen-specific T regulatory type 1 cells. Front Immunol. 2018;9:682.
9. Ouyang W, O’Garra A. IL-10 family cytokines IL-10 and IL-22: from basic science to clinical translation. Immunity. 2019;50(4):871-891.
10. Zigmond E, Bernshtein B, Friedlander G, Walker CR, Yona S, Kim KW, et al. Macrophage-restricted interleukin-10 receptor deficiency, but not IL-10 deficiency, causes severe spontaneous colitis. Immunity. 2014;40(5):720-33.
11. Zhang H, Li R, Cao Y, Gu Y, Lin C, Liu X, et al. Poor clinical outcomes and immunoevasive contexture in intratumoral IL-10-producing macrophages enriched gastric cancer patients. Ann Surg. 2022; 275(4):e626-e635.
12. Zhang H, Li R, Cao Y, Gu Y, Lin C, Liu X, et al. Poor clinical outcomes and immunoevasive contexture in intratumoral IL-10-producing macrophages enriched gastric cancer patients. Ann Surg. 2022; 275(4):e626-e635.
13. Ma T, Kong M.Interleukin-18 and -10 may be associated with lymph node metastasis in breast cancer. Oncol Lett. 2021;21(4):253.
14. Hashemi V, Maleki LA, Esmaily M, Masjedi A, Ghalamfarsa G, Namdar A, et al. Regulatory T cells in breast cancer as a potent anti-cancer therapeutic target. Int Immunopharmacol. 2020; 78:106087.
15. Giúdice AD, Pagura L, Capitani MC, Mainetti LE, Scharovsky OG, Di Masso RJ, et al. Nonclassical roles for IFN-g and IL-10 in a murine model of immunoedition. Future Sci OA. 2020;6(10):FSO589.
16. Song H, Liu A, Liu G, Wu F, Li Z. T follicular regulatory cells suppress Tfh-mediated B cell help and synergistically increase IL-10-producing B cells in breast carcinoma. Immunol Res. 2019;67(4-5):416-423.
17. Rahimi Kalateh Shah Mohammad G, Ghahremanloo A, Soltani A, Fathi E, Hashemy SI. Cytokines as potential combination agents with PD-1/PD-L1 blockade for cancer treatment. J Cell Physiol. 2020; 235(7-8):5449-5460.
18. Butte MJ, Keir ME, Phamduy TB, Sharpe AH, Freeman GJ. Programmed death-1 ligand 1 interacts specifically with the B7-1 costimulatory molecule to inhibit T cell responses. Immunity. 2007;27(1):111-22.
19. Lee M, Lee YH, Song J, Kim G, Jo Y, Min H, et al. Deep-learning-based three-dimensional label-free tracking and analysis of immunological synapses of CAR-T cells. Elife. 2020;9:e49023.
20. Li H, Zhang Q, Shuman L, Kaag M, Raman JD, Merrill S, et al. Evaluation of PD-L1 and other immune markers in bladder urothelial carcinoma stratified by histologic variants and molecular subtypes. Sci Rep. 2020;10(1):1439.
Full Text Sources:
Abstract:
Views: 1228

For Authors
Journal Subscriptions

Dec 2025
Supplements
Instructions for authors
Online submission
Contact
e-ISSN: 2601 - 1700 (online)
ISSN-L: 2559 - 723X
Journal Abbreviation: Surg. Gastroenterol. Oncol.
Surgery, Gastroenterology and Oncology (SGO) is indexed in:
- SCOPUS
- EBSCO
- DOI/Crossref
- Google Scholar
- SCImago
- Harvard Library
- Open Academic Journals Index (OAJI)
Surgery, Gastroenterology and Oncology (SGO) is an open-access, peer-reviewed online journal published by Celsius Publishing House. The journal allows readers to read, download, copy, distribute, print, search, or link to the full text of its articles.
Time to first editorial decision: 25 days
Rejection rate: 61%
CiteScore: 0.2

Meetings and Courses in 2025
Meetings and Courses in 2024
Meetings and Courses in 2023
Meetings and Courses in 2022
Meetings and Courses in 2021
Meetings and Courses in 2020
Meetings and Courses in 2019
Verona expert meeting 2019

Surgery, Gastroenterology and Oncology applies the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits readers to copy and redistribute the material in any medium or format, remix, adapt, build upon the published works non-commercially, and license the derivative works on different terms, provided the original material is properly cited and the use is non-commercial. Please see: https://creativecommons.org/licenses/by-nc/4.0/
Publisher’s Note:
The opinions, statements, and data contained in article are solely those of the authors and not of Surgery, Gastroenterology and Oncology journal or the editors. Publisher and the editors disclaim responsibility for any damage resulting from any ideas, instructions, methods, or products referred to in the content.
 IASGO Society News
IASGO Society News