Surgery, Gastroenterology and Oncology
|
|
Introduction: Breast cancer patients with multicentric disease will require mastectomy advances of screening and genetics have ended in many ladies may needs prophylactic mastectomy nipple and areola sparing is good option for these patients with higher levels of psychosocial wellbeing.
Patient and Methods: The 300 breast cancer patients all have NSM for the therapy of multicentric breast cancer. The primary end point was tumor recurrence post NSM either was local, regional or metastatic recurrence and, the Secondary end point for this study is to report disease free survival, loco-regional control and overall survival.
Results: All cases were multicentric invasive duct carcinoma, 6 cases (33.3%) were reported to have distant metastases to lung and bone and 12 cases (66.7%) were reported to have local recurrence within nipple areola complex, among reported recurrence cases the interval time between primary treatment and recurrence was 28 months in 6 cases (33.3%), 43 months in 6 cases (33.3%) and 50 months in 6 cases (33.3%). Among 300 cases that did NSM, 6 cases (2%) died from heavy lung metastases and 294 cases (98%) still alive within period of follow up ranging from 40-96 months.
Conclusion: This study revealed a low incidence of cancer recurrence at the NAC after NSM and immediate breast reconstruction. Patients with high stage disease, HER2 positive subtype, high grade lesions and extensive intraductal component had a significantly increased risk of cancer recurrence at the NAC.
INTRODUCTION
Breast cancer is the most widely recognized disease affecting ladies of any age and stays one of the main sources of death in ladies of any age with age occurrence in Egypt 10 years younger than mean age rate worldwide (1). Conservative breast surgery was done for about 41% of Egyptian patients diagnosed with breast invasive duct carcinoma (2). However many patients may present with multicenteric disease or extensive widespread DCIS, those patients will require mastectomy additionally, advances of screening and genetics have ended in many ladies requesting prophylactic mastectomy. Nipple sparing mastectomy will be the gold standard surgical option for those patients; however many patients were afraid of loco-regional recurrence post nipple sparing mastectomy (3). Patients with preserved nipple-areolar complexes reported higher levels of psychosocial wellbeing (4).
PATIENTS AND METHOD
Statistical analysis
Data was analyzed using IBM SPSS advanced statistics (Statistical Package for Social Sciences), version 24 (SPSS Inc., Chicago, IL). Numerical data was described as median and range or mean and standard deviation as appropriate, while qualitative data were described as number and percentage. Chi-square (Fisher's exact) test was used to examine the relation between qualitative variables as appropriate. Survival analysis was done using Kaplan-Meier method. Comparison between two survival curves was done using log rank test. Bonferonni corrections of p value was done to avoid hyperinflation of type 1 error which arises from multiple testing A p-value less than or equal to 0.05 was considered statistically significant. All tests were two tailed Overall survival (OS) was calculated from date of diagnosis till date of death or last follow up. Disease free survival (DFS) was calculated from date of Complete remission till date of relapse, metastasis, death or last follow up.
Patient consent information
All patients have explained surgery and its drawbacks and possible complications and explained another surgical options or procedure and consent taken for every patient after discussion and MDT decision. This study included 300 Ladies diagnosed with breast invasive carcinoma who did nipple sparing mastectomy with immediate reconstruction, after obtaining ethical committee approval clinical records were obtained and analyzed. All patients went through NSM for the therapy of multicenteric breast cancer. Patients involved in this study were female patients, their age ranged from 20 to 60 years old, with practically no associated clinical co-morbidities, desiring breast reconstruction post nipple sparing mastectomy. Tumor to nipple distance was not restricting factor for performing NSM provided that negative intra-operative pathological frozen assessment of the terminal ductal system. The essential end point for this study is to report tumor recurrence post NSM either was local, regional or metastatic recurrence and to highlight the risk factors for this recurrence via univariate analysis and multivariate analysis of Tumor characteristics (type, size, grade, distance from nipple & areola complex and hormonal receptors)way of axillary lymph node dissection either complete axillary evacuation or sentinel lymph node biopsy, technical concern as type of incision and results of frozen section from terminal ductal system and treatment modality as receiving neoadjuvant chemotherapy or adjuvant therapy either chemotherapy, radiotherapy and hormonal therapy. Secondary end point for this study is to report disease free survival, loco-regional control and overall survival.
RESULTS
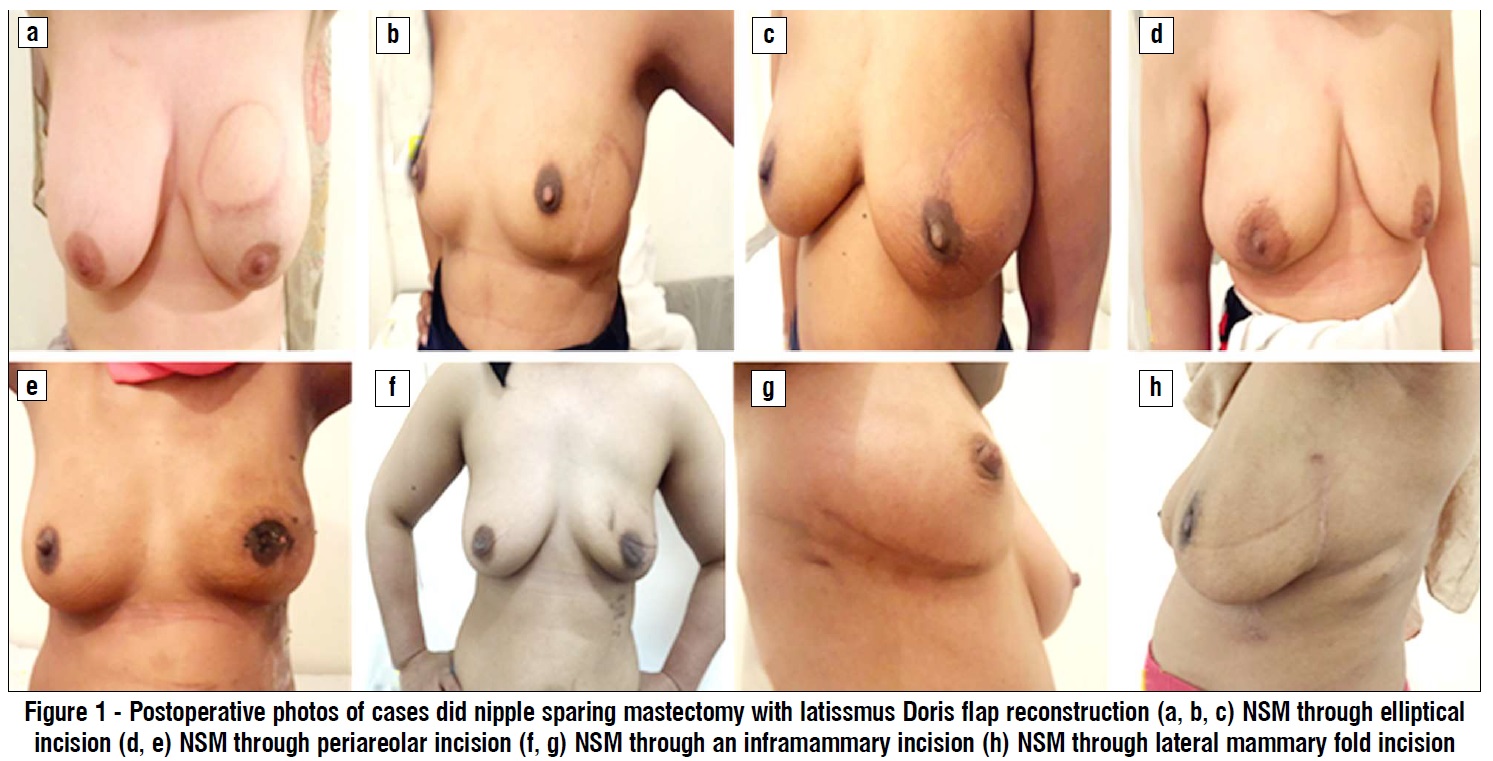
Among the reported cases who did nipple sparing mastectomy, 186 cases (62%) were under 30 years and 114 cases (38%) were over 30 years, 144 cases (48%) were in the left breast and 156 cases (52%) were in the right breast, 150 cases (50%) were staged as T1 and 150 cases (50%) were staged as T2, 90 cases (30%) were staged as N1 and 210 cases (70%) were staged as N0, all cases were staged as M0, multicentricity was present in all cases clinically and radiologically. NSM was done through an elliptical incision in 138 cases (46%), through an infra-mammary incision in 18 cases (6%), through lateral mammary fold incision in 54 cases (18%) and through periareolar incision in 90 cases (30%) (fig. 1).
Pathologically, all cases were multicenteric invasive duct carcinoma, 132 cases (44%) were luminal A, 120 cases (40%) were luminal B, 24 cases (8%) were Her.2 positive, 12 cases (4%) were triple negative and 12 cases (4%) were triple positive, 210 cases (70%) were grade 1&2 and 90 cases (30%) were grade 3, 90 cases (30%) showed marked anaplasia and mitosis with lymphovascular invasion. 120 cases (40%) showed an extensive intra-ductal component more than 20%. Recurrence was reported in 18 cases (6%), among those 18 cases, 6 cases (33.3%) were reported to have distant metastases to lung and bone and 12 cases (66.7%) were reported to have local recurrence within nipple areola complex, among reported recurrence cases the interval time between primary treatment and recurrence was 28 months in 6 cases (33.3%), 43 months in 6 cases (33.3%) and 50 months in 6 cases (33.3%), among recurrent cases, 6 cases (33.3%) were treated via chemotherapy and radiotherapy, 12 cases (66.7%) were dealt with surgery in the form of wide local excision of the local recurrence, after treatment of the recurrence follow up period was 12 months in 12 cases (66.7%) and 48 months in 6 cases (33.3%) with good response to treatment in 12 cases (66.7%) and poor response in 6 cases (33.3%). Among 300 cases that did NSM, 6 cases (2%) died from heavy lung metastases and 294 cases (98%) still alive within period of follow up ranging from 40-96 months (table 1).
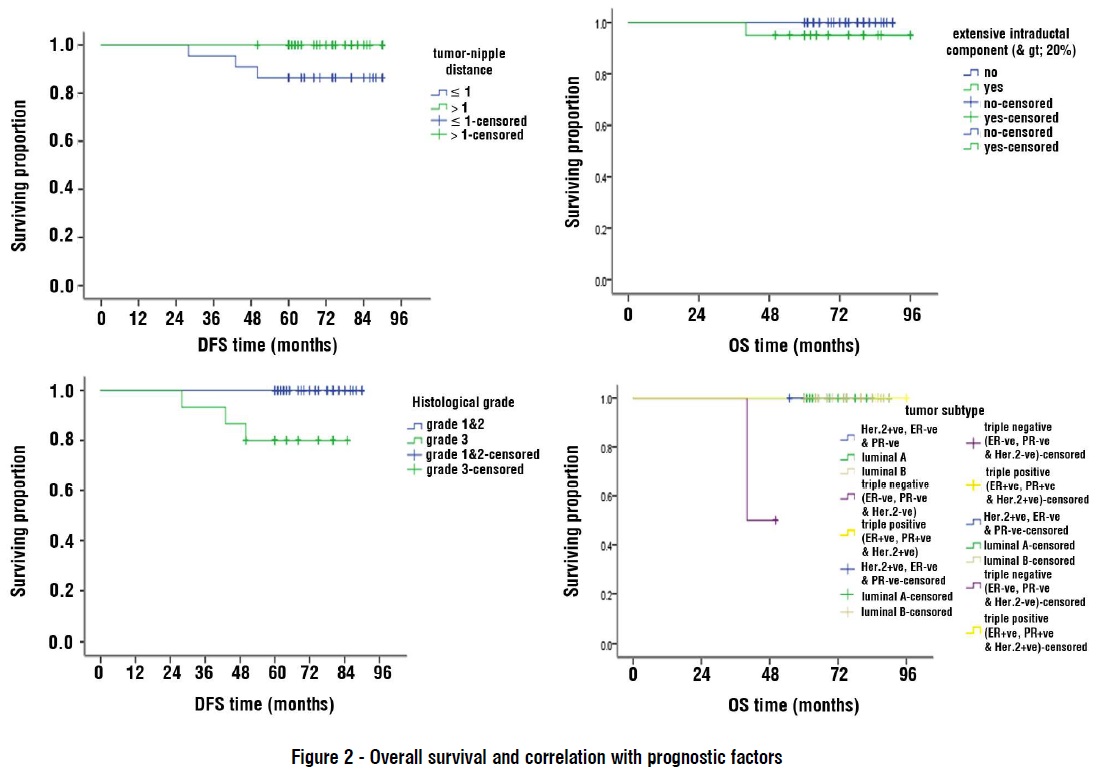
Regarding survival analysis throughout median follow up of 29 months (extending from 40-96 months). Disease free survival percentage at 5 years was 100% for T1 lesions and 92% for T2 lesions, it was 80% for grade 3 lesions and 100% for grade 1&2 lesions, it was 80% for N1 lesions and 100% for N0 lesions, it was 86.4% for lesions with distance in between tumor and nipple ex-ceding 1cm and was 100% for lesions with tumor nipple distance lesser than 1cm, it was 85% for tumors with extensive intraductal component and was 100% for tumors without extensive intraductal component, it was 100% for luminal A and luminal B lesions and was 75% for Her.2 positive lesions and was 50% for triple positive lesions and was not recorded for triple negative lesions with (P.value < 0.001). Overall survival proportion at 5 years (fig. 2) was 100% for T1 lesions and was 96% for T2 lesions; it was 95% for patients with extensive intraductal component lesions and was 93% for grade 3 lesions comparing with 100% for grade 1&2 lesions. Overall survival proportion at 5 years was 50% for patients with triple negative lesions and was 100% for patients with the other tumor subtypes (luminal A, luminal B, Her.2+ve & triple +ve) with (P value <0.001).
DISCUSSION
Nipple-sparing mastectomy has acquired expanded acceptance, with good esthetic results and without affecting oncological safety. Follow-up data regards to the oncologic safety of modern NSM concerning locoregional recurrence and survival are variable. Among 99 cases did NSM Jensen et al., delineated no recurrence at NAC during a mean follow up of 60.2 months (5). Among 981 cases Wang et al., enumerated no case of cancer recurrence at the NAC during time of follow up of 29 months (6). Sakurai et al. delineated 3.7 % recurrence rate in NAC among 788 cases went through NSM during time of follow up of 78 months (7). Zhen et al., stated 5-year local recurrence at the NAC of 3.5% in patients went through NSM between 2003 and 2015 (8). Newman et al. detailed a 6.2% local recurrence rate during during time of follow up of 5 years (9). Vaughan et al. stated a 5.3% local recurrence rate during time of follow up of 5 years (10). Medina-Franco et al. detailed a 4.5% local recurrence rate during time of follow up of 8 years (11).
Gerber et al. during period of follow up of 10 years reported one case of local recurrence within the NAC among 112 cases underwent NSM (12). Incidence of loco-regional recurrence post neoadjuvant CTH then NSM was ranging from 3.2% and 10.3% (13). Among our study, we reported 18 cases of recurrence (6%) among 300 patients did NSM and during period of follow up ranging from 40:96 months, among those 18 cases, 6 cases (33.3%) were reported to have distant metastases to lung and bone and 12 cases (66.7%) were reported to have local recurrence within nipple areola complex. Variation in the rate of the local recurrence can be explained by the different inclusion criteria among different studies. Among 934 cases went through NSM for invasive breast cancer and within period of follow up of 50 months Petit et al., stated 11 patients with local recurrence with conclusion that tumor biological type was the main predictor for recurrence risk in multivariate analysis (14). Lohsiriwat et al. enumerated 7 cases of local recurrence within NAC post NSM and detailed that extensive intraductal component, negative hormonal receptor status, high pathological grade and HER.2 overexpression were all related to an increased risk of cancer recurrence at the NAC after NSM (15). Other studies detailed that young age, estrogen receptor negativity and HER.2 Over-expression were related to an increased risk of loco-regional recurrence (16). Tumor size, number of positive nodes, histological type, grade and the biological features of disease (HER2/neu, Ki-67, vascular invasion) are viewed as risk factors for local recurrence post NSM (17). Lari and Kuerer stated that phenotype HER2+, ER−, Ki67+ was a strong determinant of local recurrence post NSM (18). Gabos et al. detailed that triple positive, triple negative and (hormonal receptor) +/HER2+ subtypes were accompanied by an increased risk of loco-regional recurrence in univariate analysis. While tumor size and nodal involvement were risk factors for local recurrence in multivariate analysis (19). Scheiden et al. revealed that the validity of pathological frozen evaluation especially in central breast lesions (20). Wu et al, featured the impact of margin status on local recurrence and NAC recurrence, they stated 6.5% and 6.5% of the patients with positive or close margins (less than 1 mm), respectively, and 4.6% and 2% of the patients with clear margins (more than 1 mm), respectively.
Other risk factors for local recurrence enumerated by their study are large tumor size (more than 4 cm), high grade, and negative hormonal status and HER2 positivity. Other studies did not find any relation between margin status and local recurrence (21). Among our study, we couldn't assess the impact of margin status on locoregional recurrence as among all of our cases who did NSM, the margin was clear (negative margin). Some studies considered that tumor nipple distance is a predictor for nipple involvement (22). Wu ZY et al. among 319 NSM cases done after neoadjuvant CTH revealed that patients less than 50 years old, tumor size more than 5 cm, positive LNs and molecular subtype were risk factors for loco-regional recurrence in univariate analysis while tumor grade and lympho-vascular invasion were risk factors for locoregional recurrence in multivariate analysis (23). Voltura et al. delineated that tumor subtype was the most important risk factor for local recurrence (24). Among our study, we reported that tumor size, nuclear grade, nodal status, tumor nipple distance with cutoff point (1 cm), extensive intraductal component and tumor subtype were risk factors for tumor local recurrence. We reported also that tumor biology (tumor subtype) was the most important risk factor for local recurrence. Langstein et al. detailed that overall survival rate is much better in patients with subcutaneous recurrence (61%) compared to those with chest wall recurrence (45%) in a period of follow up of 80.8 months (25). Among our study we reported 12 cases (66.7%) with local recurrence and all of those cases had the recurrence within the NAC. Wu et al, reported the impact of the interval time between NSM and the local recurrence on prognosis regarding post recurrence disease free survival and distant metastases free survival with cutoff point (2 years), they reported that 5-year post-recurrence disease free survival rates for patients with an interval time (less than 2 years) versus those with (more than 2 years) were 61.9% and 79.5%, respectively,
The 5-year post-recurrence distant metastases free survival rates for patients with an interval time (less than 2 years) versus those with (more than 2 years) were 69.6% and 84.5%, respectively (26). Among recurrent cases within our study (18 cases), the interval time between primary treatment and recurrence was 28 months in 6 cases (33.3%), 43 months in 6 cases (33.3%) and 50 months in 6 cases (33.3%) with observation of improved prognosis regarding post recurrence disease free survival in patients with local recurrence with prolonged time interval period between the NSM and the local recurrence, we also observed very poor outcome with patient with recurrence in the form of distant metastases post NSM. Surgical excision of the local recurrence is considered the gold standard option for local recurrence as far as there was no evidence of distant metastases (27). Among our study, surgical excision was the primary treatment for patients with local recurrence (12 cases) and was in the form of wide local excision of the local recurrence with negative margins while chemotherapy was the treatment of choice for patients presented with distant metastases post NSM (6 cases). Regarding the very restricted available data on the outcomes of LRR following NSM and immediate reconstruction, our study results provide significant initial insights that will help with the suitable management and surveillance of this patient population.
CONCLUSION
This study revealed a low incidence of cancer recurrence at the NAC after NSM and immediate breast reconstruction. Patients with high stage disease, HER2 neu–positive subtype, high histologic grade, lymphovascular invasion and positive extensive intraductal component had a significantly increased risk of cancer recurrence at the NAC. These findings suggest that factors should be considered when planning for NSM. Most patients with cancer recurrence at the NAC had a favorable prognosis after receiving appropriate comprehensive treatment.
Conflict of interest
No conflict of interset.
Source of found
This study didn’t receive any fund.
Ethical approval
Approved from ethical committee.
REFERENCES
1. National Cancer Registry Program of Egypt. http://www.cancerregistry. gov.eg/
2. Ibrahim AS, Khaled HM, Mikhail NNH, Baraka H and Kamel H. Egypt National Cancer Registry: Aswan Profile—2008. National Cancer Registry Program of Egypt. Publication Number RR1, Cairo. 2010.
3. Chen W, Lv X, Xu X, Gao X, Wang B. Meta-analysis for psychological impact of breast reconstruction in patients with breast cancer. Breast Cancer. 2018;25(4):464-469.
4. De Alcantara Filho P, Capko D, Barry JM, Morrow M, Pusic A. and Sacchini VS. Nipple-sparing mastectomy for breast cancer and risk-reducing surgery: the Memorial Sloan-Kettering Cancer Center experience. Ann Surg Oncol. 2011;18(11):3117-22.
5. Jensen JA, Orringer JS, Giuliano AE. Nipple-sparing mastectomy in 99 patients with a mean follow-up of 5 years. Ann Surg Oncol. 2011; 18(6):1665-70.
6. Wang F, Peled AW, Garwood E, Stover Fiscalini A, Sbitany H, RD Foster, et al. Total skin-sparing mastectomy and immediate breast reconstruction: an evolution of technique and assessment of outcomes. Ann Surg Oncol. 2014;21(10):3223-30.
7. Sakurai T, Zhang N, Suzuma T, Umemura T, Yoshimura G, Sakurai T, et al. (2013) Long-term follow-up of nipple-sparing mastectomy without radiotherapy: a single center study at a Japanese institution. Med Oncol. 2013;30(1):481.
8. Wu ZY, Kim HJ, Lee JW, Chung IY, Kim JS, Sae-Byul Lee et al. (2019) Breast Cancer Recurrence in the Nipple-Areola Complex After Nipple-Sparing Mastectomy With Immediate Breast Reconstruction for Invasive Breast Cancer. JAMA Surg. 2019;154(11):1030-1037.
9. Newman LA, Kuerer HM, Hunt KK, Kroll SS, Ames FC, Ross MI, et al. Presentation, treatment, and outcome of local recurrence after skin-sparing mastectomy and immediate breast reconstruction. Ann Surg Oncol. 1998;5(7):620-6.
10. Vaughan A, Dietz JR, Aft R, Gillanders WE, Eberlein TJ, Freer P, et al. Scientific Presentation Award. Patterns of local breast cancer recurrence after skin-sparing mastectomy and immediate breast reconstruction. Am J Surg. 2007;194(4):438-43.
11. Medina-Franco H, Vasconez LO, Fix RJ, Heslin MJ, Beenken SW, Bland KI, et al. Factors associated with local recurrence after skinsparing mastectomy and immediate breast reconstruction for invasive breast cancer. Ann Surg. 2002; 235(6):814-9.
12. Gerber B, Krause A, Dieterich M, Kundt G, and T. Reimer. The oncological safety of skin sparing mastectomy with conservation of the nipple-areola complex and autologous reconstruction: an extended follow-up study. Ann Surg. 2009;249(3):461-8.
13. Wengler CA, Valente SA, Al-Hilli Z, Woody NM, Muntean JH, Abraham J, et al. Determinants of short and long term outcomes in patients undergoing immediate breast reconstruction following neoadjuvant chemotherapy. J Surg Oncol. 2017;116(7):797-802.
14. Petit JY, Veronesi U, Orecchia R, Curigliano G, Rey PC, Botteri E, et al. (2012) Risk factors associated with recurrence after nipplesparing mastectomy for invasive and intraepithelial neoplasia. Ann Oncol. 2012;23(8):2053-2058.
15. Lohsiriwat V, Martella S, Rietjens M, Botteri E, Rotmensz N, Mastropasqua MG, et al. Paget’s disease as a local recurrence after nipple-sparing mastectomy: clinical presentation, treatment, outcome, and risk factor analysis. Ann Surg Oncol. 2012;19(6):1850-5.
16. Shimo A, Tsugawa K, Tsuchiya S, Yoshie R, Tsuchiya K, Uejima T, et al. Oncologic outcomes and technical considerations of nipplesparing mastectomies in breast cancer: experience of 425 cases from a single institution. Breast Cancer. 2016;23(6):851-860.
17. Wallgren A, Bonetti M, Gelber RD, Goldhirsch A, Castiglione-Gertsch M, Holmberg SB, et al. Risk factors for locoregional recurrence among breast cancer patients: results from International Breast Cancer Study Group Trials I through VII. J Clin Oncol. 2003; 21(7):1205-13.
18. Lari SA, Kuerer HM Biological markers in DCIS and risk of breast recurrence: a systematic review. J Cancer. 2011;2:232-61.
19. Gabos Z, Thoms J, Ghosh S, Hanson J, Deschênes J, Sabri S, et al. The association between biological subtype and locoregional recurrence in newly diagnosed breast cancer. Breast Cancer Res Treat. 2010;124(1):187-94. .
20. Scheiden R, Sand J, Tanous AM, Knolle U, Capesius C, Wagnon MC, et al. Accuracy of frozen section diagnoses of breast lesions after introduction of a national programme in mammographic screening. Histopathology. 2001; 39(1):74-84.
21. Chan LW, Rabban J, Hwang ES, Bevan A, Alvarado M, Ewing C, et al. Is radiation indicated in patients with ductal carcinoma in situ and close or positive mastectomy margins? Int J Radiat Oncol Biol Phys. 2011;80(1):25-30.
22. Klein J, Kong I, Paszat L, Nofech-Mozes S, Hanna W, Thiruchelvam D, et al. Close or positive resection margins are not associated with an increased risk of chest wall recurrence in women with DCIS treated by mastectomy: a population-based analysis. Springerplus. 2015;4:335.
23. Wu ZY, Kim HJ, Lee JW, Chung IY, Kim JS, Lee SB, et al. Oncologic Outcomes of Nipple-Sparing Mastectomy and Immediate Reconstruction after Neoadjuvant Chemotherapy for Breast Cancer. Ann Surg. 2021;274(6):e1196-e1201.
24. Voltura AM, Tsangaris TN, Rosson GD, Jacobs LK, Flores JI, Singh NK, et al. Nipple-sparing mastectomy: critical assessment of 51 procedures and implications for selection criteria. Ann Surg Oncol. 2008;15(12):3396–401.
25. Langstein HN, Cheng MH, Singletary SE, Robb GL, Hoy E, Smith TL, et al. Breast cancer recurrence after immediate reconstruction: patterns and significance. Plast Reconstr Surg. 2003;111(2):712- 20; discussion 721-2.
26. Wu ZY, Han HH, Kim HL, Lee J, Chung IY, Kim J, et al. Locoregional recurrence following nipple-sparing mastectomy with immediate breast reconstruction: Patterns and prognostic significance. Eur J Surg Oncol. 2021;47(6):1309-1315.
27. Buchanan CL, Dorn PL, Fey J, Giron G, Naik A, Mendez J, et al. Locoregional recurrence after mastectomy: incidence and outcomes. J Am Coll Surg. 2006;203(4):469-74.
Full Text Sources:
Abstract:
Views: 3892

For Authors
Journal Subscriptions

Dec 2025
Supplements
Instructions for authors
Online submission
Contact
e-ISSN: 2601 - 1700 (online)
ISSN-L: 2559 - 723X
Journal Abbreviation: Surg. Gastroenterol. Oncol.
Surgery, Gastroenterology and Oncology (SGO) is indexed in:
- SCOPUS
- EBSCO
- DOI/Crossref
- Google Scholar
- SCImago
- Harvard Library
- Open Academic Journals Index (OAJI)
Surgery, Gastroenterology and Oncology (SGO) is an open-access, peer-reviewed online journal published by Celsius Publishing House. The journal allows readers to read, download, copy, distribute, print, search, or link to the full text of its articles.
Time to first editorial decision: 25 days
Rejection rate: 61%
CiteScore: 0.2

Meetings and Courses in 2025
Meetings and Courses in 2024
Meetings and Courses in 2023
Meetings and Courses in 2022
Meetings and Courses in 2021
Meetings and Courses in 2020
Meetings and Courses in 2019
Verona expert meeting 2019

Surgery, Gastroenterology and Oncology applies the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits readers to copy and redistribute the material in any medium or format, remix, adapt, build upon the published works non-commercially, and license the derivative works on different terms, provided the original material is properly cited and the use is non-commercial. Please see: https://creativecommons.org/licenses/by-nc/4.0/
Publisher’s Note:
The opinions, statements, and data contained in article are solely those of the authors and not of Surgery, Gastroenterology and Oncology journal or the editors. Publisher and the editors disclaim responsibility for any damage resulting from any ideas, instructions, methods, or products referred to in the content.
 IASGO Society News
IASGO Society News