Surgery, Gastroenterology and Oncology

|
|
Introduction. Nephrolithiasis is one of the complications seen after bariatric surgery. In Iraq, a survey that involved 3916 adults showed that 31.8% were overweight, and 33.9% are obese. Purposes. This study prospectively examined the incidence of nephrolithiasis and possible predictors in a laparoscopic sleeve gastrectomy cohort of patients for one year.
Methods. A prospective cohort study involving 300 participants they were divided into two cohorts: the sleeve gastrectomy (SG) cohort consisted of 165 morbidly obese patients undergoing SG surgery, and the second cohort (control cohort) included 165 morbidly obese who did not undergo surgery. Each cohort was followed 6 and 12 months postoperatively. 24-hour total urinary analysis was taken for both cohorts at baseline and after 1 year; this included total volume, urinary calcium, oxalate, uric acid, and citrate.
Results. The overall stone formation in the 12 months of follow-up in SG vs. control cohorts (13.3 vs. 6.7%, p-value = 0.044). After 12 months of follow-up, several urinary parameters were significantly reduced in the SG cohort group, including urinary calcium, urinary uric acid, urinary citrate, and total urine volume (mean reduction was -45 mg/day, -255.0 mg/day, -155.2 mg/day, and -297.7 ml/day; respectively), while urinary oxalate significantly increased in the SG group (mean increase by 5.6 mg/day).
Conclusion. Patients undergoing laparoscopic sleeve surgery are at increased risk of developing kidney stones, with a risk as high as twice that of match control patients who did not undergo a surgical procedure. Urinary risk factors include low urinary calcium, uric acid, citrate, total urine volume, and elevated urinary oxalate.
Keywords: sleeve surgery, calcium oxalate, control-cohort, morbid obesity, urinalysis
Introduction
The global incidence of obesity has been steadily increasing since 1980, regardless of geographical location, socioeconomic level, age, or sex (1). The global prevalence of obesity, defined as having a body mass index (BMI) of 30 kg/m2 or more, is 6.2% among men and 7.0% among women (2). In Iraq, a survey that involved 3916 adults showed that 31.8% were overweight, and 33.9% were obese aged 40–49 years; living in an urban residence and having hypertension were predictors of obesity, which indicates the prudent of obesity in the Iraqi population in general is high (3). The concurrent rise in obesity rates across nearly all nations appears to be primarily influenced by transformations in the worldwide food system, generating more processed, cost-effective, and well-promoted food products than ever before (4). Obesity primarily impacts middle-aged adults, particularly women, from prosperous metropolitan areas in low-income countries. In higher-income nations, obesity affects individuals of all ages and genders but is more prevalent among disadvantaged populations (4).
Kidney stone disease, or nephrolithiasis, is a long-standing medical condition with a rich historical record. The prevalence and incidence of kidney stone development is said to be increasing worldwide, with an estimated 1-15% of individuals experiencing it at some point in their lifetime (5-7). Nephrolithiasis has become more common globally in recent decades. Nephro-lithiasis is a chronic condition that frequently reoccurs during a person's life. The recurrence rate is 50% within 5-10 years and 75% within 20 years (7,8). Untreated kidney stones can lead to obstruction of the ureter, hematuria, recurrent urinary tract infections, emesis, or dysuria, ultimately resulting in irreversible renal impairment (9).
The mineralogical composition of kidney stones can be categorized into five primary types: calcium oxalate (CaOx; 65.9%), carbapatite (15.6%), urate (12.4%), struvite (magnesium ammonium phosphate, 2.7%), and brushite (1.7%) (10,11). Males have a higher incidence of CaOx and urate stones, while females have a higher prevalence of carbapatite and struvite stones compared to males (10,12). Nevertheless, the precise understanding of how sex differences contribute to the pathophysiological mechanisms of urinary stone disease remains incomplete (13).
Obesity is a well-established risk factor for kidney stones, with a reported incidence ranging from 4.3% to 4.6% (14,15). There is no apparent increase in frequency after undergoing treatments that are deemed to be more restricted in nature, where the reported incidence rate is 3.7% (16). An occurrence rate of 7.7-8.1% has been observed following Roux-en-Y gastric bypass (RYGB) (14,16). Two meta-analyses concluded that the risk of developing kidney stones was 0.37 and 0.29, respectively, for strictly restrictive surgeries such as sleeve gastrectomy (SG). In contrast, the related risk for developing kidney stones was 1.73 and 1.79 for RYGB surgeries (17,18).
The specific causes contributing to the increasing prevalence and recurrence of urolithiasis have not yet been determined. Kidney stone disease has a significant impact on both individuals and society due to its high occurrence in working-age adults. It has become a public health concern, especially in areas with a hot and dry climate, such as Iraq (19,20). This study uniquely identifies the risk of nephrolithiasis in only one type of bariatric surgery: sleeve surgery. All other research in the literature examined the other types of bariatric surgery, and sleeve surgery is sometimes presented as a minority of cases in other studies. Thus, this study aimed to examine the incidence of kidney stones and possible predictors in a special cohort of patients: laparoscopic sleeve gastrectomy patients.
Methods
Study Design and Setting
In a prospective cohort study involving 300 participants, they were divided into two cohorts: the sleeve gastrectomy (SG) cohort consisted of 165 morbidly obese patients undergoing SG surgery, and the second cohort (control cohort) included 165 morbidly obese who did not undergo surgery. Each cohort was followed 6 and 12 months postoperatively.
24-hour total urinary analysis was taken for both cohorts at baseline and after 1 year; this included total volume, urinary calcium, oxalate, uric acid, and citrate.
The study was conducted at Al-Sadr Teaching Hospital in the Misan governorate (a teaching hospital associated with the College of Medicine/Misan University), Baghdad Teaching Hospital/Medical City Complex, and Beirut Private Hospital/Baghdad between January 2021 and September 2023. Written and informed consent was obtained from all the patients participating in this study.
Inclusion Criteria
Adult patients aged 18 years and older underwent sleeve gastrectomy and BMI ? 35 kg/m2.
Exclusion Criteria
Patients with a previous history of kidney stones, history of inflammatory bowel disease, and BMI < 35.0 kg/m2, chronic kidney and liver diseases.
Statistical Analysis
An Independent t-test was used to assess the differences in age and BMI. In contrast, the chi-square test was used to assess the differences in sex, hypertension, diabetes, and stone formation rate/ repeated measure two-way ANOVA was used to assess the differences in change of urinary parameters between baseline and after 12 months of follow-up divided according to the study cohorts, as analysis carried out using GraphPad prism 10.2, the p-value was considered to be significant if ?0.05.
Results
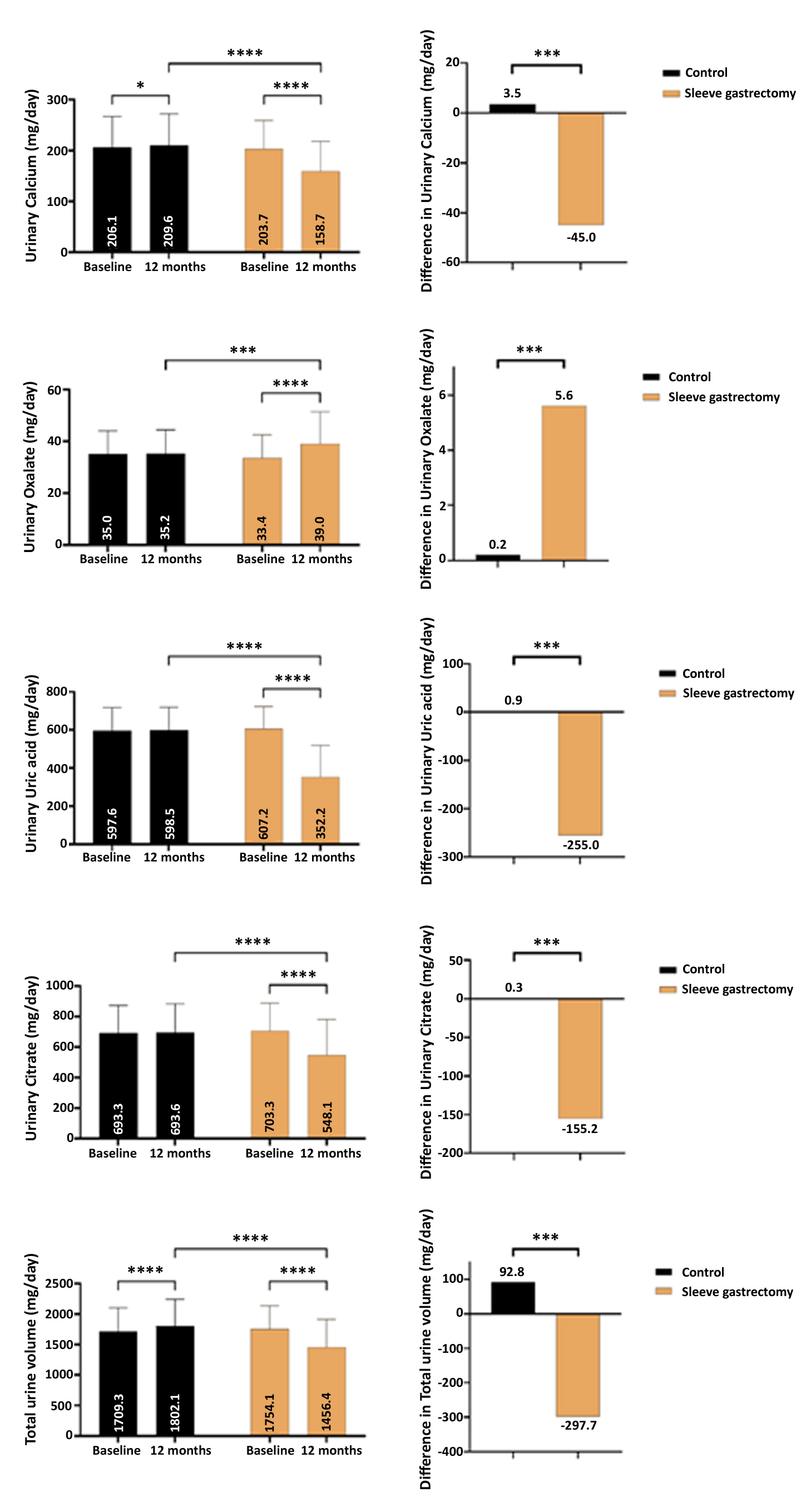
As illustrated in table 1, there was no significant difference in mean age, body mass index (BMI), sex, hypertension, and diabetes mellitus at baseline.
Table 1 - Assessment of baseline variables
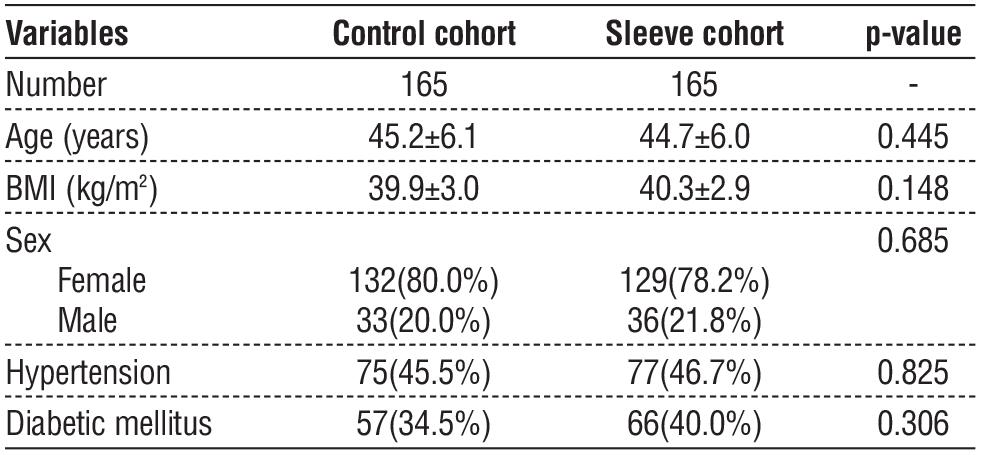
We followed up with the patients after 6 and 12 months of their operation for the SG cohort; at the same time, we also prospectively followed the control cohort (without surgery) and compared the rate of kidney stone formation, the rate of stone formation was slightly higher in SG cohort during the first 6 months of follow-up (9.1 vs. 4.2%) during the second 6 months of follow-up the rate of stone formation was also higher in SG cohort (4.2 vs. 2.4%). Still, it did not reach statistical significance; when we examined the overall stone formation in the 12 months of follow-up (13.3 vs. 6.7%), the difference between both cohorts reached statistical significance, as seen in fig. 1.
Figure 1 - Percentage of new stones formation during the study follow-up
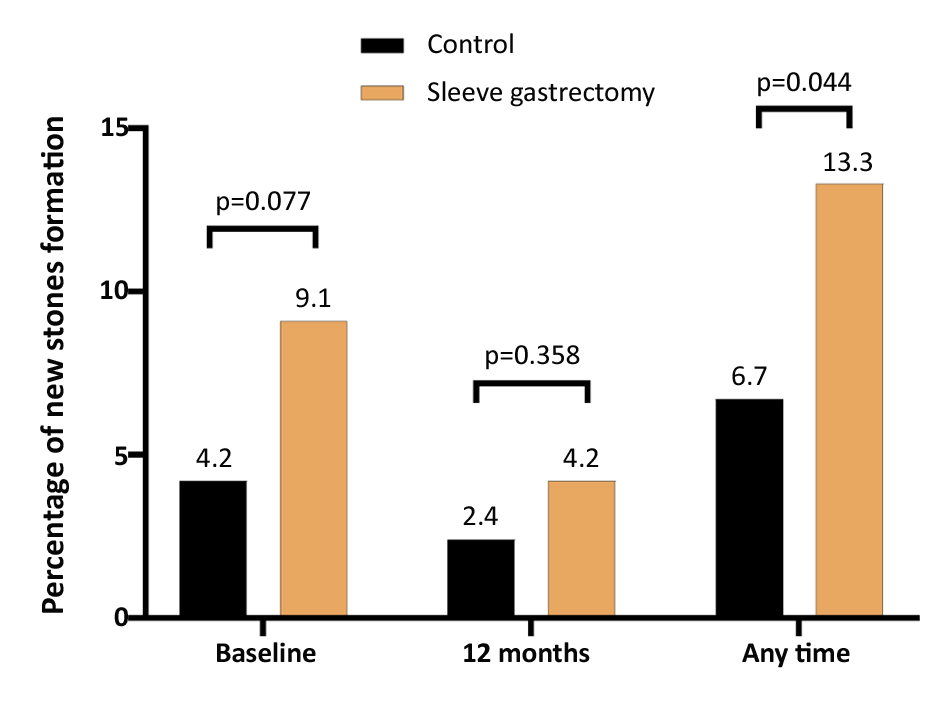
There was no significant difference (p-value = 0.899) in the type of stones according to the study cohort; in the control cohort, 8 developed calcium oxalate, 3 developed hydroxyapatite stones, while for the SG cohort, 11 developed calcium oxalate, 5 developed hydroxyapatite stones, 2 developed Struvite, and one developed uric acid stones, as illustrated in fig. 2.
Figure 2 - Percentage of stones type according to study groups
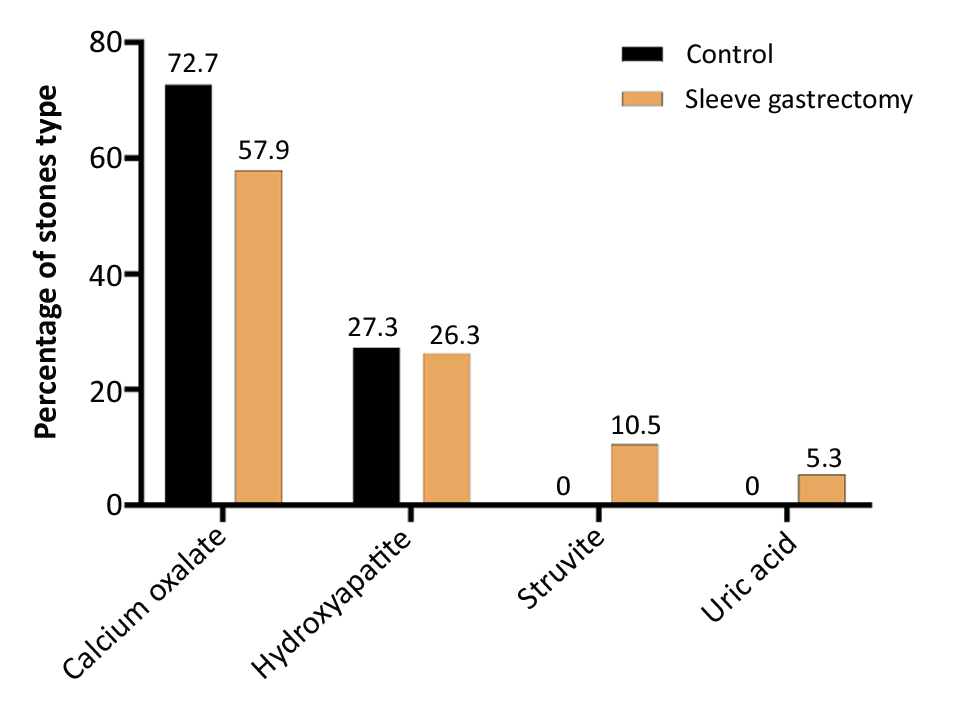
After 12 months of follow-up, several urinary parameters were significantly reduced in the SG cohort group, including urinary calcium, urinary uric acid, urinary citrate, and total urine volume (mean reduction was -45 mg/day, -255.0 mg/day, -155.2 mg/day, and -297.7 ml/day; respectively), while urinary oxalate significantly increased in the SG group (mean increase by 5.6 mg/day), as seen in fig. 3.
Figure 3 - 24-hour urine analysis at baseline and after 12 months of follow-up, (a) urinary calcium, (b) urinary oxalate, (c) urinary uric acid, (d) urinary citrate, and (e) total urinary volume.
Discussion
In the present study, we examined prospectively the incidence of new nephrolithiasis in a cohort of patients undergoing sleeve gastrectomy. We compared this cohort with morbidly obese individuals who did not undergo a surgical procedure. The rate of stone formation was significantly higher after one year (13.3 vs. 6.7%). This provides clinical evidence of the increased risk of kidney stones even after low-risk restrictive surgery like sleeve surgery; it is established that from the many types of bariatric surgeries, the greatest risk of kidney stones in malabsorptive procedures, intermediate risk in standard Roux-en-Y gastric bypass surgery (RYGB), and least risk in restrictive procedures (15).
In the present study, urinary calcium, uric acid, citrate, and total urine volume were significantly reduced after one year of follow-up compared to the control cohort. In contrast, urinary oxalate significantly increased in the SG group compared to the control cohort.
Several studies examined nephrolithiasis risk after bariatric surgeries; during a median follow-up period of around 4 years, 7.65% of patients who underwent bariatric surgery developed a stone, but only 4.63% of obese individuals in the control group experienced the same. The average duration between bariatric surgery and the occurrence of the stone incident was 1.5 years. However, it seems that laparoscopic adjustable gastric band surgery or sleeve gastrectomy did not affect the chance of developing stones. A possible explanation for this discrepancy is that different populations were examined, and the study did not control for risk factors like hypertension and diabetes, which the current study controlled for (21,22).
After 3 years of follow-up, the risk of nephrolithiasis was 7.7% in RYGB surgery compared to 4.6% for control (without surgery) in a national private insurance claims database study (14), another study that is similar in design to the current study, after 6 years of follow-up and 14% risk for nephrolithiasis compared to 7% in control after 10 years of follow-up, the risk of stones formation was 11% compared to 4.3% for control, 78% of the patients performed RYGB, 14% performed very long limb RYGB or biliopancreatic diversion/duodenal switch, while 7% performed laparoscopic banding or sleeve gastrectomy (15).
In a small study (n=18) with a short follow-up that examined gastric banding (n=14) and SG surgery (n=4), urinary oxalate was reduced more in the restrictive surgery compared to RYGB. Overall, there is no evidence to suggest that restrictive bariatric surgery is linked to a higher likelihood of developing kidney stone disease (23). Another small study (n=39) reported similar findings (27 performed RYGB and 12 gastric banding procedures. Individuals who underwent gastric banding placement exhibited reduced urine quantities (24). A study analyzing the occurrence rates in 85 people with a specific medical condition indicated that just 1% of patients formed a stone after an average follow-up period of slightly more than 2 years (21).
In the current study, the most common type of stone is calcium oxalate, which is agreed by others (15, 25,26). This was associated with increased levels of 24-hour urinary oxalate, which also agrees with other studies (18,26,27). The results align with previous evidence from non-bariatric surgery patients, indicating that a higher urine calcium oxalate supersaturation is linked to an increased likelihood of developing kidney stones (28). Therefore, comprehending the alterations in urine calcium oxalate and its primary factors following surgery might offer valuable understanding as to why certain individuals develop kidney stones post-surgery. The main constituents of urine that affect the level of calcium oxalate are urine calcium, oxalate, citrate, and volume (29). Elevated concentrations of calcium or oxalate in urine, as well as decreased urine volume or citrate levels, independently contribute to an increased risk of calcium oxalate formation in urine and the development of kidney stones (30).
Not every individual with gastrointestinal issues marked by fat malabsorption will experience the formation of kidney stones. A study examined a group of 51 individuals who suffered from difficulty absorbing fats. Within this group, 10 patients had a history of forming kidney stones, while the remaining 41 patients did not. The group that forms stones had higher levels of urine oxalate excretion, lower levels of urinary citrate excretion, and higher levels of calcium oxalate supersaturation.
Conclusions
Patients undergoing laparoscopic sleeve surgery are at increased risk of developing kidney stones, with a risk as high as twice that of match control patients who did not undergo a surgical procedure. Urinary risk factors include low urinary calcium, uric acid, citrate, total urine volume, and elevated urinary oxalate.
Author’s Contributions
Thaer Saleh Sabor Al-Omary; Conceptualization; Investigation; Methodology; Project administration; Resources; Supervision; Validation; Visualization; Writing – original draft and Writing – review & editing. Muhammed Ghanim Alwan; Conceptualization; Project administration; Resources; Software; Visualization; Writing – original draft and Writing – review & editing. Aseel Mahmood Jawad; Conceptualization; Data Curation; Investigation; Methodology; Project administration; Writing – original draft and Writing – review & editing.
Conflict of Interests
All author declare that they have no conflict of interest.
Funding
None.
Ethical Consideration
This work approved by Department of Surgery, Collage of Medicine, University of Misan (no. 401 in 3/12/2023).
References
1. Hoffman K, Shah R, Ismail M, Satyavada S, Alkhayyat M, Mansoor E, et al. Incidence of Kidney Stones After Bariatric Surgeries: Comparing Roux-en-Y Gastric Bypass and Sleeve Gastrectomy. J Gastrointest Surg. 2023;27(11):2336-2341.
2. NCD Risk Factor Collaboration (NCD-RisC). Worldwide trends in underweight and obesity from 1990 to 2022: a pooled analysis of 3663 population-representative studies with 222 million children, adolescents, and adults. Lancet. 2024;403(10431):1027-50.
3. Pengpid S, Peltzer K. Overweight and Obesity among Adults in Iraq: Prevalence and Correlates from a National Survey in 2015. Int J Environ Res Public Health. 2021;18(8):4198.
4. Swinburn BA, Sacks G, Hall KD, McPherson K, Finegood DT, Moodie ML, et al. The global obesity pandemic: shaped by global drivers and local environments. Lancet. 2011;378(9793):804-14.
5. Romero V, Akpinar H, Assimos DG. Kidney stones: a global picture of prevalence, incidence, and associated risk factors. Rev Urol. 2010;12(2-3):e86-96,
6. Morgan MS, Pearle MS. Medical management of renal stones. BMJ. 2016;352:i52.
7. Alwan MG, Nima MH, Alquraishi FS, Rashid NR. Deciding on a novel predictive value to gauge how well patients with lower ureteric stones respond to medical expulsive therapy. Urolithiasis. 2024; 52(1):41.
8. Eisner BH, Goldfarb DS. A nomogram for the prediction of kidney stone recurrence. J Am Soc Nephrol. 2014;25(12):2685-7.
9. Alwan MG, Nima MH, Al-Omary T SS. A new variable emerges to guide the decision when choosing surgical options for renal stones larger than 20 mm. Ukrainian Journal of Nephrology and Dialysis, 2024;(3(83):25-32.
10. Ye Z, Zeng G, Yang H, Li J, Tang K, Wang G, et al. The status and characteristics of urinary stone composition in China. BJU Int. 2020; 125(6):801-809.
11. Parmar MS. Kidney stones. BMJ. 2004;328(7453):1420-4.
12. Sun X, Shen L, Cong X, Zhu H, He L, Lu J. Infrared spectroscopic analysis of 5,248 urinary stones from Chinese patients presenting with the first stone episode. Urol Res. 2011;39(5):339-43.
13. Wang Z, Zhang Y, Zhang J, Deng Q, Liang H. Recent advances on the mechanisms of kidney stone formation (Review). Int J Mol Med. 2021;48(2):149.
14. Matlaga BR, Shore AD, Magnuson T, Clark JM, Johns R, Makary MA. Effect of gastric bypass surgery on kidney stone disease. J Urol. 2009;181(6):2573-7.
15. Lieske JC, Mehta RA, Milliner DS, Rule AD, Bergstralh EJ, Sarr MG. Kidney stones are common after bariatric surgery. Kidney Int. 2015;87(4):839-45.
16. Mishra T, Shapiro JB, Ramirez L, Kallies KJ, Kothari SN, Londergan TA. Nephrolithiasis after bariatric surgery: a comparison of laparoscopic Roux-en-Y gastric bypass and sleeve gastrectomy. Am J Surg. 2020;219(6):952-957.
17. Thongprayoon C, Cheungpasitporn W, Vijayvargiya P, Anthanont P, Erickson SB. The risk of kidney stones following bariatric surgery: a systematic review and meta-analysis. Renal failure. 2016;38(3): 424-30.
18. Upala S, Jaruvongvanich V, Sanguankeo A. Risk of nephrolithiasis, hyperoxaluria, and calcium oxalate supersaturation increased after Roux-en-Y gastric bypass surgery: a systematic review and meta-analysis. Surg Obes Relat Dis. 2016;12(8):1513-21.
19. Abeywickarama B, Ralapanawa U, Chandrajith R. Geoenvironmental factors related to high incidence of human urinary calculi (kidney stones) in Central Highlands of Sri Lanka. Environ Geochem Health. 2016;38(5):1203-14.
20. Wang Z, Zhang JW, Zhang Y, Zhang SP, Hu QY, Liang H. Analyses of long non-coding RNA and mRNA profiling using RNA sequencing in calcium oxalate monohydrate-stimulated renal tubular epithelial cells. Urolithiasis. 2019;47(3):225-34.
21. Chen T, Godebu E, Horgan S, Mirheydar HS, Sur RL. The effect of restrictive bariatric surgery on urolithiasis. J Endourol. 2013;27(2): 242-4.
22. Semins MJ, Matlaga BR, Shore AD, Steele K, Magnuson T, Johns R, et al. The effect of gastric banding on kidney stone disease. Urology. 2009;74(4):746-9.
23. Semins MJ, Asplin JR, Steele K, Assimos DG, Lingeman JE, Donahue S, et al. The effect of restrictive bariatric surgery on urinary stone risk factors. Urology. 2010;76(4):826-9.
24. Penniston KL, Kaplon DM, Gould JC, Nakada SY. Gastric band placement for obesity is not associated with increased urinary risk of urolithiasis compared to bypass. J Urol. 2009;182(5):2340-6.
25. Nelson WK, Houghton SG, Milliner DS, Lieske JC, Sarr MG. Enteric hyperoxaluria, nephrolithiasis, and oxalate nephropathy: potentially serious and unappreciated complications of Roux-en-Y gastric bypass. Surg Obes Relat Dis. 2005;1(5):481-5.
26. Sinha MK, Collazo-Clavell ML, Rule A, Milliner DS, Nelson W, Sarr MG, et al. Hyperoxaluric nephrolithiasis is a complication of Roux-en-Y gastric bypass surgery. Kidney Int. 2007;72(1):100-7.
27. Agrawal V, Liu XJ, Campfield T, Romanelli J, Silva JE, Braden GL. Calcium oxalate supersaturation increases early after Roux-en-Y gastric bypass. Surg Obes Relat Dis. 2014;10(1):88-94.
28. Prochaska M, Taylor E, Ferraro PM, Curhan G. Relative Supersaturation of 24-Hour Urine and Likelihood of Kidney Stones. J Urol. 2018;199(5):1262-1266.
29. Werness PG, Brown CM, Smith LH, Finlayson B. EQUIL2: a BASIC computer program for the calculation of urinary saturation. J Urol. 1985;134(6):1242-4.
30. Curhan GC, Willett WC, Speizer FE, Stampfer MJ. Twenty-four-hour urine chemistries and the risk of kidney stones among women and men. Kidney Int. 2001;59(6):2290-8.
Full Text Sources:
Abstract:
Views: 1274

For Authors
Journal Subscriptions

Dec 2025
Supplements
Instructions for authors
Online submission
Contact
e-ISSN: 2601 - 1700 (online)
ISSN-L: 2559 - 723X
Journal Abbreviation: Surg. Gastroenterol. Oncol.
Surgery, Gastroenterology and Oncology (SGO) is indexed in:
- SCOPUS
- EBSCO
- DOI/Crossref
- Google Scholar
- SCImago
- Harvard Library
- Open Academic Journals Index (OAJI)
Surgery, Gastroenterology and Oncology (SGO) is an open-access, peer-reviewed online journal published by Celsius Publishing House. The journal allows readers to read, download, copy, distribute, print, search, or link to the full text of its articles.
Time to first editorial decision: 25 days
Rejection rate: 61%
CiteScore: 0.2

Meetings and Courses in 2025
Meetings and Courses in 2024
Meetings and Courses in 2023
Meetings and Courses in 2022
Meetings and Courses in 2021
Meetings and Courses in 2020
Meetings and Courses in 2019
Verona expert meeting 2019

Surgery, Gastroenterology and Oncology applies the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits readers to copy and redistribute the material in any medium or format, remix, adapt, build upon the published works non-commercially, and license the derivative works on different terms, provided the original material is properly cited and the use is non-commercial. Please see: https://creativecommons.org/licenses/by-nc/4.0/
Publisher’s Note:
The opinions, statements, and data contained in article are solely those of the authors and not of Surgery, Gastroenterology and Oncology journal or the editors. Publisher and the editors disclaim responsibility for any damage resulting from any ideas, instructions, methods, or products referred to in the content.
 IASGO Society News
IASGO Society News