Surgery, Gastroenterology and Oncology

|
|
Background: Hepatocellular carcinoma (HCC) is the sixth most common cancer worldwide and the third most common cause of cancer mortality. Underlying liver cirrhosis is the strongest predisposing factor for HCC. Many patients with HCC are diagnosed at an advanced stage when curative treatment is no longer feasible. Early detection of HCC is crucial for providing effective initial treatments. Fibroscan, a non-invasive, reproducible tool for assessing liver stiffness, may help identify patients with liver cirrhosis at high risk of developing HCC. Objective: To evaluate the role of Fibroscan-measured liver stiffness in predicting hepatocellular carcinoma among liver cirrhotic patients.
Methods: This cross-sectional analytical study was conducted in the Department of Hepatology, Bangladesh Medical University (BMU), Dhaka, from May 2022 to August 2023, involving 40 liver cirrhotic patients (20 with HCC and 20 without HCC). HCC was diagnosed by measuring serum alpha-fetoprotein (AFP) level and triphasic CT (computed tomography) scan of HBS. The liver stiffness was measured for patients using echosens fibroscan machine.
Results: The mean age of participants was 48.88±12.50 years; 77.5% were male. Liver stiffness and Alpha-fetoprotein were significantly higher in the HCC group. A cut-off liver stiffness ³46.05 kPa can significantly predict HCC with 70% Sensitivity, 80% specificity, and 75.00% accuracy.
Conclusion: Liver stiffness ³46.05 kPa measured by Fibroscan efficiently predicts hepatocellular carcinoma in liver cirrhotic patients.
INTRODUCTION
Liver cancer is the sixth leading cause of cancer and the third leading cause of cancer-related deaths globally (GLOBOCAN) (1). HCC is the most common type of primary liver cancer, accounting for 75-86 % of cases (2). Men are affected approximately two to three times more than women, with higher incidence and mortality in most countries (3). The strongest risk factor for developing HCC is cirrhosis of liver from any etiology, including viral hepatitis, non alcoholic steatohepatitis (NASH) and alcohol related hepatitis, which is present in over 80% of patients with HCC (4). Patients with cirrhosis of liver from any etiology typically have a ?2% annual risk of developing HCC (5).
Early identification of high risk individuals amongst the liver cirrhotic patients are crucial, as prognosis and treatment outcomes largely depend on the stage of HCC at diagnosis (6). Many patients who are diagnosed cases of HCC, have advanced disease, and are not candidates for curative treatment. However, early detection of HCC through surveillance methods have increased patient survival by providing effective initial treatments such as primary curative hepatectomy, locoregional ablative therapy or liver transplantation. Conventional surveillance tools, such as abdominal ultrasonography with or without serum alpha-fetoptotein (AFP) measurement, have limited sensitivity for early HCC detection, particularly in obese or advanced liver cirrhotic patients (7). Therefore, there is an increasing need for more reliable, non-invasive methods to stratify the risk of HCC development in this patients. The degree of liver fibrosis is the strongest indicator of HCC risk; therefore, liver stiffness measurement is helpful in identifying patients at high risk for HCC (8). The liver biopsy is the gold standard for quantitative assessment of fibrosis. However, it is associated with several problems such as invasiveness, sampling errors and diagnostic differences between pathologists (9). Noninvasive assessment of liver fibrosis with transient elastography using Fibroscan is emerging as a reliable, reproducible and noninvasive tool for evaluating liver stiffness (10). Liver stiffness measurement (LSM) reflects the degree of hepatic fibrosis and indirectly correlates with portal hypertension, both of which are established risk factors for HCC development (11). Several studies have demonstrated that higher baseline LSM values are associated with an increased risk of HCC in patients with liver cirrhosis, independent of other clinical factors (12,13). Consequently, Fibroscan not only aids in staging fibrosis but may also serve as a valuable predictor of HCC risk, enabling tailored surveillance and management strategies in high risk populations (14). This article explores the role of Fibroscan in predicting hepatocellular carcinoma in cirrhotic patients.
Transient elastography using Fibroscan is a non-invasive method for measuring liver stiffness. An ultrasound transducer probe is mounted on the axis of a vibrator. Low-frequency, mild-amplitude vibrations from the vibrator are transmitted to the tissues via the transducer, thereby inducing an elastic shear wave that propagates through the tissue. In the meantime, pulse-echo ultrasound acquisitions allow the propagation of the shear wave to be followed and its velocity to be measured, as these are directly related to tissue stiffness: the stiffer the tissue, the faster the shear wave propagates (15). The results are expressed in kilopascals (kPa). It is painless, rapid (less than 5 minutes), and easy to perform at the bedside or in the outpatient clinic (16). The validity of the liver stiffness results also depends on three important parameters: the interquartile range (IQR), which reflects the variability of the validated measures, should not exceed 20 – 30% of the median value; a minimum of 10 valid individual measurements; and the success rate (the number of successful measurements divided by the total number of acquisitions) should be at least 60% (17). The result is presented as median stiffness, ranging from 1 to 75 kPa. Normal values are generally less than 5 kPa.
MATERIAL AND METHOD
This cross-sectional analytical study was conducted in the Department of Hepatology at Bangladesh Medical University (BMU), Dhaka, from August 2022 to August 2023. Ethical approval for the study was obtained from the Institutional Review Board (IRB) of BMU. A total 40 patients of liver cirrhosis with or without hepatocellular carcinoma were included in the study according to inclusion and exclusion criteria.
Inclusion Criteria
Patients with cirrhosis of liver with or without hepatocellular carcinoma (HCC) who are above 18 years old.
Exclusion Criteria
Hepatic encephalopathy, ascites, hepatorenal syndrome or hepatopulmonary syndrome, right-sided heart failure (liver congestion), extrahepatic tumour, history of liver transplantation, cholestasis and pregnancy.
Patients were selected using purposive sampling and grouped into the hepatocellular carcinoma group (n1=20) and the non-hepatocellular carcinoma group (n2=20). Diagnosis of liver cirrhosis was based on following criteria: Clinically stigmata of cirrhosis (vascular spider, palmer erythema, gynecomastia, leukonychia, clubbing, testicular atrophy) and laboratory features (reduced platelet count, prolonged prothrombin time, reduced serum albumin, alter AST/ALT ratio), inhomogenous or coarse liver parenchyma with or without enlarged spleen on abdominal ultrasound, any grade of varix or portal hypertensive gastropathy on upper GIT endoscopy or liver histology support cirrhosis of liver.
HCC was diagnosed by measuring the serum alpha-fetoprotein (AFP) level, imaging such as ultrasonography (USG) and triphasic CT (computed tomography) of the HBS (hepatobiliary system), and/or Fine Needle Aspiration Cytology (FNAC). A thorough clinical history was taken and a physical examination was performed. At baseline, for each patient, demographic and clinical data including age, sex, stigmata of cirrhosis and complications of cirrhosis like ascites, variceal bleeding, hepatorenal syndrome or hepatic encephalopathy was evaluated and obtained. Then other investigations like complete blood count (CBC); liver function tests like - serum bilirubin, alanine aminotransferase (ALT), aspartate aminotransferase (AST); serum albumin, prothrombin time; viral markers like - HBsAg, Anti HBc(T), Anti HCV; serum creatinine, serum electrolytes, blood sugar, alpha-fetoprotein; ultrasonography (USG) of whole abdomen, and endoscopy of upper GIT were done. The potential benefits and risks of use of Fibroscan were explained to the patients. Then, liver stiffness was measured using the Echosens Fibroscan machine. Fibroscan was done using M probe with maintaining following criteria- (a) At least 10 valid individual measurements were taken (b) Success rate was greater than 60%(c) An interquartile range to median ratio was less than 20%.
Statistical Analysis
Following data collection, data were entered. The collected data was assessed for completeness, accuracy and consistency before analysis. Statistical analysis was carried out using the Statistical Package for the Social Sciences (SPSS) version 27. Normality of the data was checked by Shapiro-Wilk test. Data were presented as mean ± standard deviation (SD), median (range), or number (%) unless specified. The difference in quantitative data between the two groups (HCC or non-HCC) was assessed using the Student's t-test or the Mann-Whitney U test, depending on the data distribution. Association between categorical variables was assessed by Chi-square test/fisher’s exact test. The cut-off values of FibroScan and alpha-fetoprotein (AFP) for predicting HCC were assessed using the Youden index. Based on the cutoff value, the predictive usefulness of FibroScan was determined. A p-value of < 0.05 was considered to constitute a statistically significant difference.
RESULTS
The majority (55%) of the participants were in the 41-60 age group. The median (range) age of the participants was 49.5 (25-79) years. About 78% of the patients were male (table 1).
Table 1 - Distribution of the participants according to demographic characteristics (n=40)
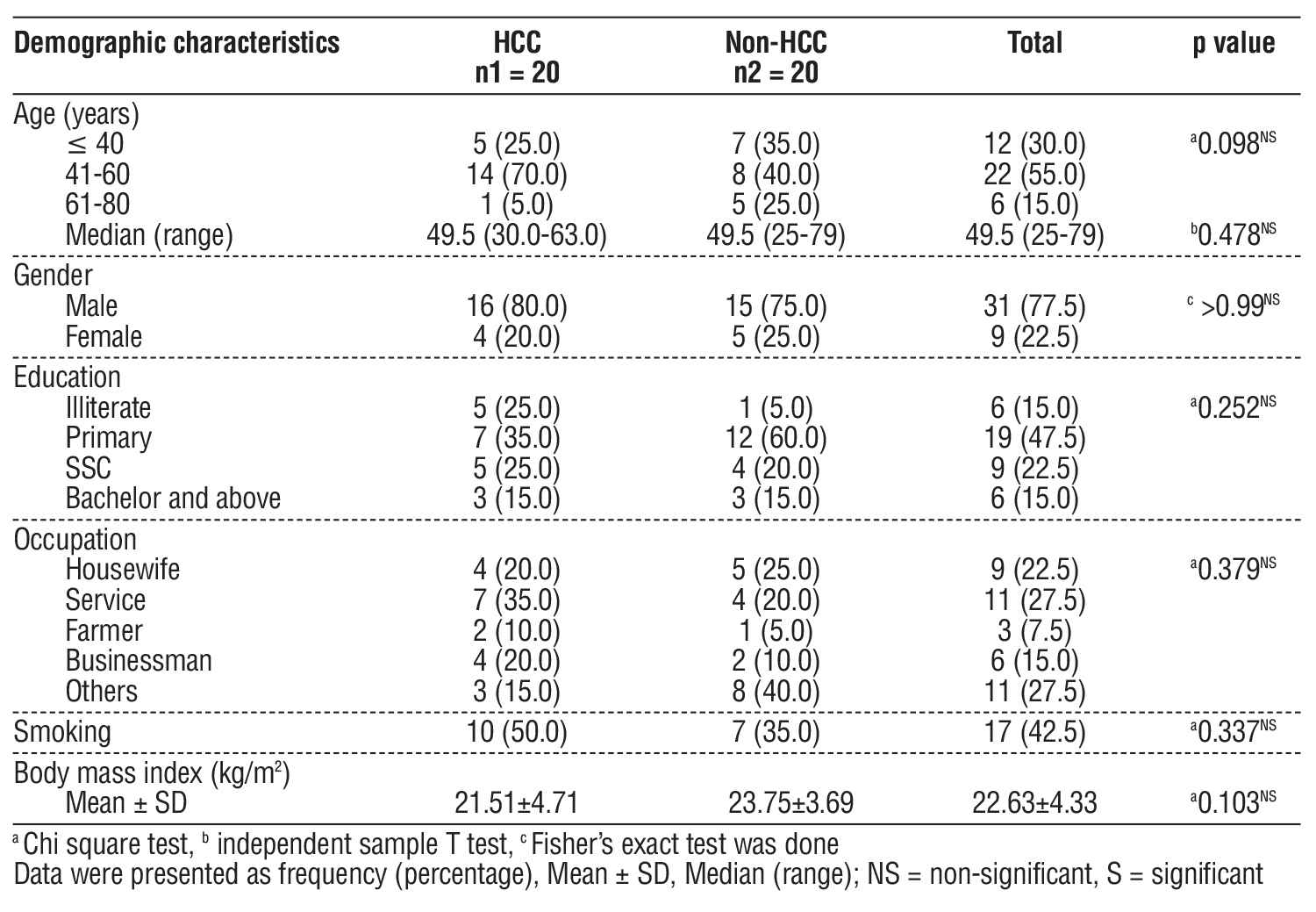
Fig. 1 showing the aetiology of liver cirrhosis in the participants with or without hepatocellular carcinoma. The etiologies were Hepatitis B, Hepatitis C, NASH, and cryptogenic cause. In the HCC group, 75% of patients were suffering from Hepatitis B virus infection, whereas in the non-HCC group it was 50%.
Figure 1 - Etiology of Liver cirrhosis of the participants with or without hepatocellular carcinoma (n=40)
The median of Alpha fetoprotein 409 ng/dl in HCC group of patients and 5 ng/dl in non-HCC group of patients. The median (range) LSM among HCC group was 69.45 (15.10-75.00), whereas the median (range) of LSM among non-HCC group was 25.85 (16.80-73.50). Liver stiffness was significantly higher in HCC group than the non-HCC group (table 2).
Table 2 - Distribution of laboratory parameters between study groups (n=40)
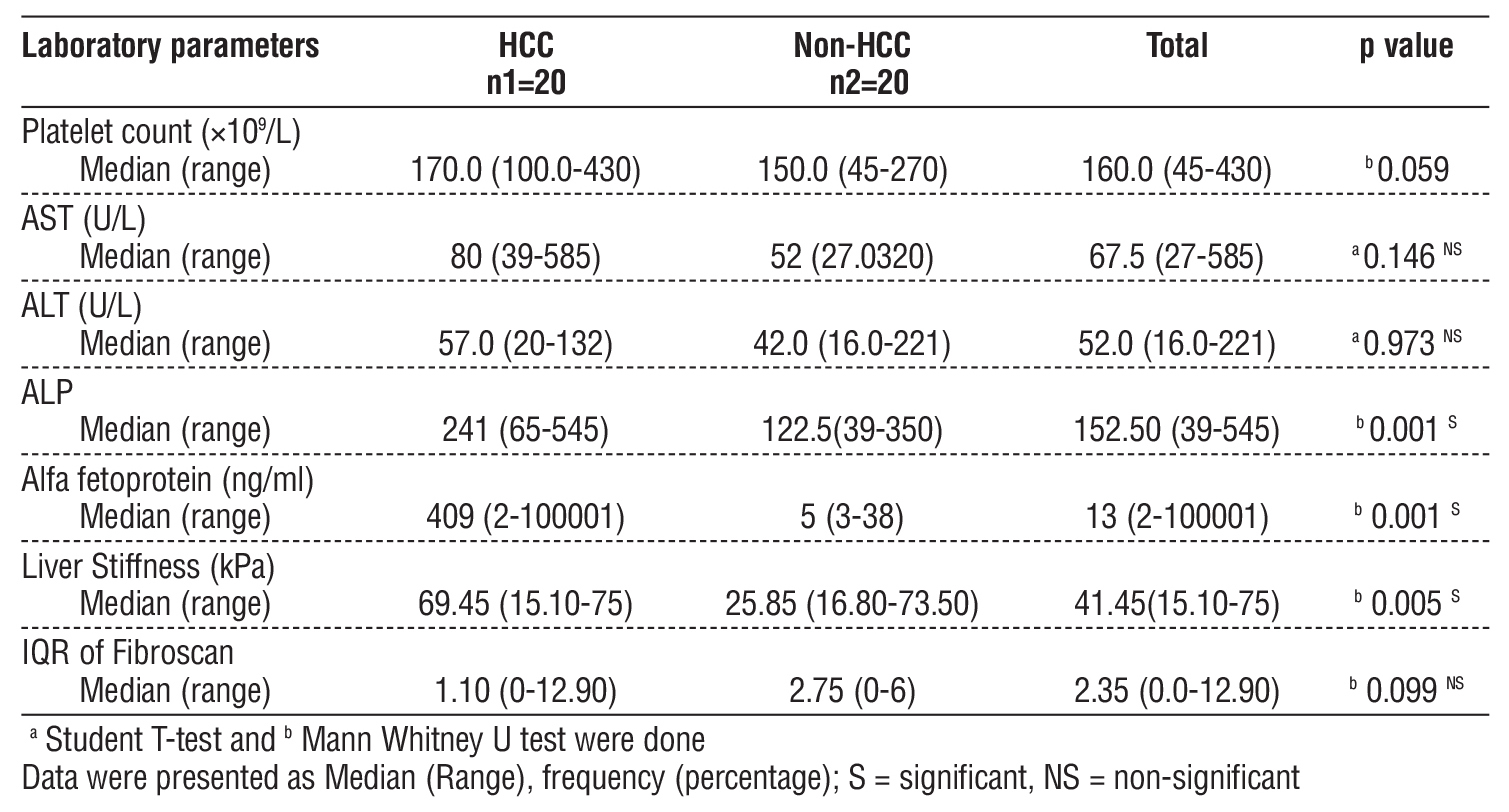
Table 3 - Determination of cut-off value of liver stiffness with Youden index
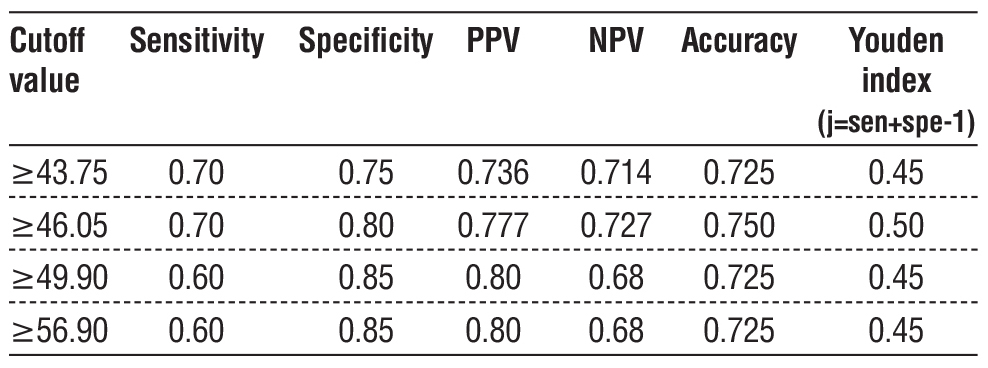
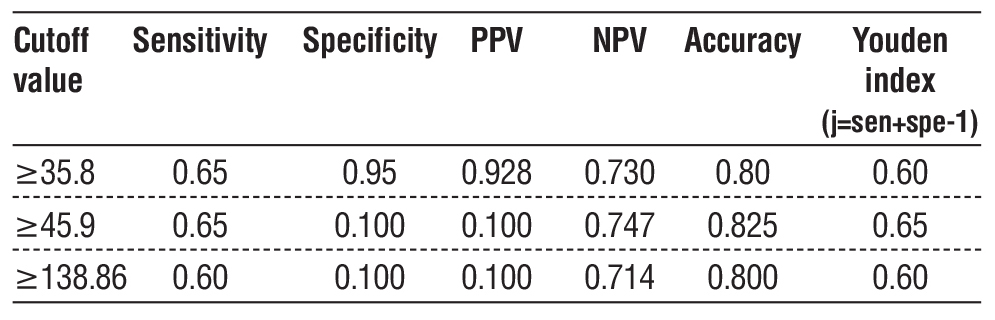
A cut-off value of liver stiffness ?46.05 yielded the highest Youden index (0.50), with 70% sensitivity, 80% specificity, and an accuracy of 75%. The cut-off value of liver stiffness ?46.05 also showed PPV and NPV of 77.78% and 72.73% respectively (table 3).
A cut-off value of AFP ?45.9 yielded the highest Youden index (0.65), with 65% sensitivity, 100% specificity, and an accuracy of 82.5%. Moreover, the cut-off value of AFP ?45.9 showed PPV and NPV of 100% and 74.70% respectively (table 4). An AFP value?45.9 ng/mL can predict HCC with 65% sensitivity and 100% specificity.
Table 4 - Determination of cut-off value of AFP with Youden index
No significant association or difference in Child Turcotte Pugh score and MELD score was observed between HCC and non-HCC group (p-value: >0.05) (table 5).
Table 5 - Child Turcotte Pugh, MELD score in different categories of study participants
Figure 2 - The Forest plot of adjusted odds ratios
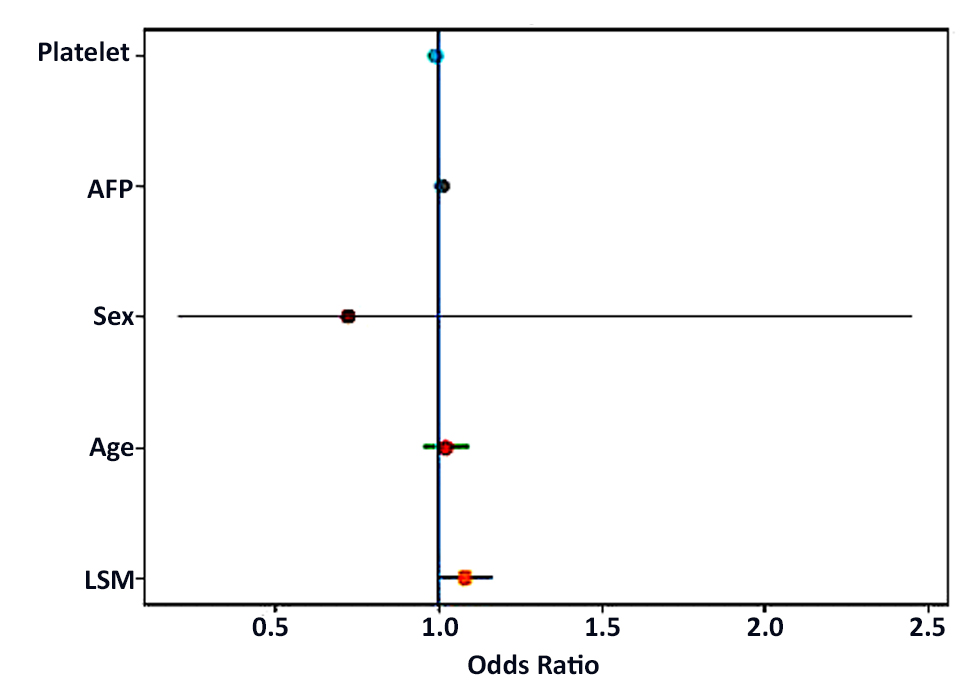
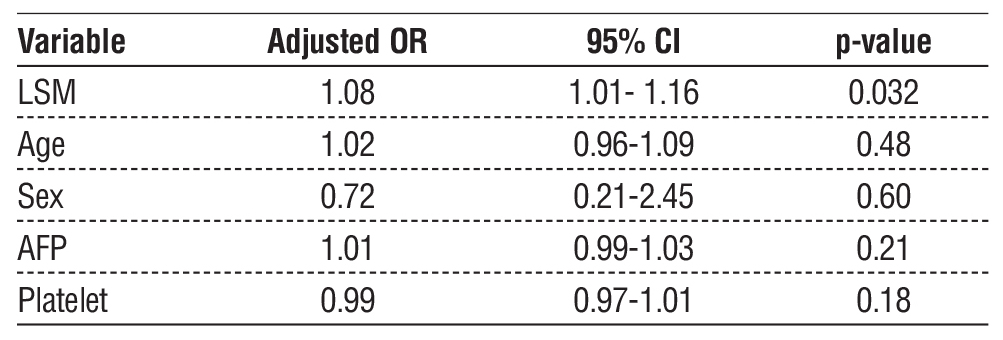
The Forest plot illustrates the adjusted odds ratios (ORs) with 95% confidence intervals (CIs) for variables included in the multivariate logistic regression model for hepatocellular carcinoma (HCC) (fig. 2). Among the variables analyzed, liver stiffness measurement (LSM) showed a statistically significant association with HCC (adjusted OR 1.08, 95% CI: 1.01-1.16, p value 0.032). In contrast, age, sex, AFP and platelet count were not significantly associated with HCC (table 6).
Table 6 - Multivariate Logistic Regression Analysis for HCC
DISCUSSION
This cross-sectional analytical study was conducted in the Department of Hepatology at Bangladesh Medical University, Dhaka, from May 2022 to August 2023. Total 40 patients with cirrhosis of liver were included, among them 20 patients were with HCC and 20 were without HCC. In our study, the median (range) age of patients with HCC was 49.5 (25-79) years, which is similar to that reported by Mahtab et al. (18), who reported a mean age of 41.92 years. Another study reported by Khan et al. (19) found the average age of HCC patients was 57.9 years, and a study by Badran et al. (8) found that, the mean age was 53.5 years in patients with HCC. In our study, the median AFP value was 409 ng/mL in the HCC group and 5 ng/mL in the non-HCC group. The median AFP value was significantly higher in the HCC group than in the non-HCC group. Ebrahim et al. (9) reported that the average AFP level in HCC patients was 317.5 ng/ml, which was statistically higher than in patients with cirrhosis (10.8 ng/ml). These results also consistent with Jiang et al. (20) who reported that AFP in cases of liver cancer had an average value of 384.6 ng/ml which was statistically higher than patients with cirrhosis (6.04 ng/ml). In this study, we found that an AFP value?45.9 ng/mL can predict HCC with 65% sensitivity and 100% specificity. Ebrahim et al. (9) reported that the sensitivity of AFP was 79% and the specificity was 89.5% at a cut-off of 26.15 ng/mL for HCC. Jiang et al. (20) reported that the sensitivity of AFP was around 60% and the specificity was about 79.9% at a cut-off of 10 ng/mL in cases of liver cancer.
In our study, we found that liver stiffness was significantly higher in the HCC group than in the non-HCC group. A study conducted by Badran et al. (8) reported that liver stiffness measured by Fibroscan were significantly higher in HCC group. In their study, Badran et al. (8) found the cutoff value of LSM to predict HCC was 30.4 kPa, with a sensitivity of 72%, specificity of 84%, positive predictive value of 81.82%, and negative predictive value of 75%. In our study, we found that the cutoff value for liver stiffness in predicting HCC was ?46.05 kPa, with sensitivity of 70.00% and specificity of 80.0%. The cutoff value for predicting HCC in our study was higher than that reported by Badran et al. (8) but slightly lower than that reported by Foucher et al. (21), who stated that the cutoff value for HCC was 53.7 kPa. Very much lower liver stiffness value (24 kPa)for HCC was reported by Ebrahim et al. (9). Tatsumi et al. (22) reported liver stiffness measurement for risk assessment of hepatocellular carcinoma and found that, in HCV related CLD, liver stiffness of more than 12.0 kPa was an independent risk factor for new HCC development. In this study, the multivariate logistic regression model also showed the liver stiffness measurement (LSM) significantly associated with hepatocellular carcinoma.
CONCLUSION
Transient Elastography using Fibroscan-measured liver stiffness is a reliable, noninvasive marker for predicting HCC risk in cirrhotic patients.In this study, a liver stiffness measurement (LSM) cut-off value of ?46.05 kPa demonstrated a 70% sensitivity and 80% specificity for predicting HCC development. These findings suggest that higher LSM values are significantly associated with increased risk of HCC, highlighting the potential use of Fibroscan as an effective tool for risk stratification and guiding surveillance strategies in cirrhotic patients.
Limitation of Study
It was a single-center study with a small sample size. We recommend that a multicenter study with a large number of cases and long-term follow-up is required to evaluate the use of Fibroscan to predict HCC more accurately.
Acknowledgements
The authors acknowledge the patients who participated in the study for their cooperation.
Conflict of Interest
All the authors state that they have no conflicts of interest to report.
Funding Information
Support and funding for the work were shared by all the authors.
REFERRENCES
1. Bray F, Ferlay j, Soerjomataram I, Siegal RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA: A Cancer J Clinicians 2018;68(6):394–424.
2. Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, et al. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin. 2021;71:209–49.
3. Amini M, Looha MA, Zarean E, Pourhoseingholi MA. Global pattern of trends in incidence, mortality, and mortality-toincidence ratio rates related to liver cancer, 1990-2019: a longitudinal analysis based on the global burden of disease study. BMC Public Health. 2022;22(1):604.
4. Valery PC, Laversanne M, Clark PJ, Petrick JL, McGlynn KA, Bray F. Projections of primary liver cancer to 2030 in 30 countries worldwide. Hepatology. 2018;67(2):600-611.
5. Kanwal F, Khaderi S, Singal AG, Marrero JA, Loo N, Asrani SK, et al. Risk factors for HCC in contemporary cohorts of patients with cirrhosis. Hepatology. 2023;77(3):997-1005.
6. Llovet JM, Kelley RK, Villanueva A, Singal AG, Pikarsky E, Roayaie S, Lencioni R, Koike K, Zucman-Rossi J and Finn RS. Hepatocellular carcinoma. Nat Rev Dis Primers. 2021;7(1):6.
7. Singal AG, Lampertico P, Nahon P. Epidemiology and surveillance for Hepatocellular carcinoma: New trends. J Hepatol. 2020; 72(2):250-261.
8. Badran AGA, EI-Tokhy N, EI-Tokhy R, Mohamad S, Baioumy HA. Role of Transient Elastography in Early Detection of HCC in Cirrhotic patients. Afro-Egyptian Journal of infection & Endemic Diseases 2017;7(3):159-168.
9. Ebrahim AE, Ahned Helmy Shehata M, Sabry AS, Manal fathy H, Sherief AE, Yousuf M. Role of fibroscan in early detection of hepatocellur carcinoma in hepatitis C cirrhotic patients. Egyptian Journal of Radiology & Nuclear medicine 2020;51:134.
10. Castéra L, Vergniol J, Foucher J, Le Bail B, Chanteloup E, Haaser M, et al. Prospective comparison of transient elastography, Fibrotest, APRI, and liver biopsy for the assessment of fibrosis in chronic hepatitis C. Gastroenterology. 2005; 128(2):343-50.
11. Masuzaki R, Ohki T, Tateishi R, Yoshida H, Goto E, Sato T. Prospective risk assessment for hepatocellular carcinoma development in patients with chronic hepatitis C by transient elastography. Hepatology. 2009;49(6):1954-61.
12. Jung Ks, Kim SU, Ahn SH, Park YN, Kim DY, Park JY, et al. Risk assessment of hepatitis B virus-related hepatocellular carcinoma development using liver stiffness measurement (Fibroscan). Hepatology. 2011;53(3):885-94.
13. Singh S, Fujii LL, Murad MH, Wang Z, Asrani SK, Ehman RL, et al. Liver stiffness associated withrisk of decompensation, liver cancer and death in patient with chronic liver disease: a systematic review and meta-analysis. Clin Gastroenterol Hepatol. 2013 ;11(12):1573-84.e1-2; quiz e88-9.
14. Wong GL, Chan HL, Wong CK, Leung C, Chan CY, Ho PP-L, et al. Liver stiffness-based optimization of hepatocellular carcinoma risk score in patients with chronic hepatitis B. J Hepatol. 2014;60(2): 339-45.
15. Sandrin L, Fourquet B, Hasquenoph JM, Yon S, Fournier C, Mal F, et al. Transient elastography: a new noninvasive method for assessment of hepatic fibrosis. Ultrasound Med Biol. 2003;29(12): 1705-13.
16. Castera L, Forns X, Alberti A. Non-invasive evaluation of liver fibrosis using transient elastography. J Hepatol. 2008;48(5):835-47.
17. Elzawawy MS, Hassainein SA, El Nomrosy RM. The role of fibroscan in assessment of liver cirrhosis in patients with chronic liver disease. Menoufia medical journal. 2018;131(2):520-524.
18. Mahtab MA, Karim MF, Rahman S, Shrestha A, Khan M. Aetiology of HCC in Bangladesh. Hepatol Int. 2009;3(1):140.
19. Khan A M, Islam MR and Alam MJ. Epidemiology of hepatocellular carcinoma (HCC) in tertiary level hospitals in Bangladesh. Annals of Oncology. 2020;31:1304.
20. Jiang J, Wu C, Shen Y, Xu B, Zheng X, Li X, et al. Clinical application of determining serum AFP-IgM complexes for diagnosis of small hepatocellular carcinoma. Anticancer Res. 2011;31(2):687-91.
21. Foucher J, Castera L, Chanteloup E, Vergniol J, Le Bjil B, Adhoute X, et al. Diagnosis of cirrhosis by Transient elastography (Fibroscan): a prospective study. Gut. 2006;55(3):403-8.
22. Tatsumi A, Maekawa S, Sato M, Komatsu N, Miura M, Amemiya F, et al. Stiffness measurement for risk assessment of hepatocellular carcinoma. Hepatol Res. 2015;45(5):523-32.
23. Masuzaki R, Tateishi R, Haruhiko Y, Hideo Y, Sato S, Kato N, et al. Risk assessment of hepatocellular carcinoma in chronic hepatitis C patients by transient elastography. J Clin Gastroenterol. 2008;42(7): 839-43.
24. Kuo Y-H, Lu S-N, Hung C-H, Kee K-M, Chen C-H, Hu T-H, et al. Liver stiffness measurement in the risk assessment of hepatocellular carcinoma for patients with chronic hepatitis. Hepatol Int. 2010;4(4): 700-6.
Full Text Sources:
Abstract:
Views: 205

For Authors
Journal Subscriptions

Dec 2025
Supplements
Instructions for authors
Online submission
Contact
e-ISSN: 2601 - 1700 (online)
ISSN-L: 2559 - 723X
Journal Abbreviation: Surg. Gastroenterol. Oncol.
Surgery, Gastroenterology and Oncology (SGO) is indexed in:
- SCOPUS
- EBSCO
- DOI/Crossref
- Google Scholar
- SCImago
- Harvard Library
- Open Academic Journals Index (OAJI)
Surgery, Gastroenterology and Oncology (SGO) is an open-access, peer-reviewed online journal published by Celsius Publishing House. The journal allows readers to read, download, copy, distribute, print, search, or link to the full text of its articles.
Time to first editorial decision: 25 days
Rejection rate: 61%
CiteScore: 0.2

Meetings and Courses in 2025
Meetings and Courses in 2024
Meetings and Courses in 2023
Meetings and Courses in 2022
Meetings and Courses in 2021
Meetings and Courses in 2020
Meetings and Courses in 2019
Verona expert meeting 2019

Surgery, Gastroenterology and Oncology applies the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits readers to copy and redistribute the material in any medium or format, remix, adapt, build upon the published works non-commercially, and license the derivative works on different terms, provided the original material is properly cited and the use is non-commercial. Please see: https://creativecommons.org/licenses/by-nc/4.0/
Publisher’s Note:
The opinions, statements, and data contained in article are solely those of the authors and not of Surgery, Gastroenterology and Oncology journal or the editors. Publisher and the editors disclaim responsibility for any damage resulting from any ideas, instructions, methods, or products referred to in the content.
 IASGO Society News
IASGO Society News