Surgery, Gastroenterology and Oncology

|
|
Introduction/Background: Colposcopy-directed targeted biopsy has been established as a proven technique for identifying cervical precancerous lesions. The qualifications of the colposcopist significantly impact the subjective evaluation of the affected cervical areaduring colposcopy. The Large Loop Excision of the Transformation Zone (LLETZ) procedure is the most commonly used excisional method for treating cervical precancerous lesions. The present study aimed to investigate the correlation between colposcopic (colposcopic impression) and histological diagnosis following LLETZ procedure with respect to cervical precancerous lesions - low- and high grade squamous intraepithelial lesionLSIL, HSIL and to calculate the sensitivity (negative predictive value – NPV) and specificity (positive predictive value – PPV) of colposcopic diagnosis with respect to HSIL in the final histological result after LLETZ.
Material and Methodology: Prospective study (01/01/2017 – 31/07/2021) including 189 patients with cervical precancerous lesions treated by the LLETZ procedure in outpatient
settings. One gynaecologic oncologist performed the biopsies under colposcopic control and LLETZ procedures and one histopathologist diagnosed histological specimens from the biopsy and LLETZ.
Results: The sensitivity and negative predictive value of colposcopic diagnosis regarding HSIL in the final histological result after LLETZ were outstanding (96 and 91%, respectively), while the specificity and positive predictive value were much lower (53 and 73%, respectively). All patients diagnose as in situ and microinvasive cervical cancer by LLETZ were reported as HSIL by colposcopy - the sensitivity and positive predictive value was 100% in this group. Only 4 (3.7%) patients histological diagnosed as HSIL are reported as LSIL by colposcopy.
Conclusion: Colposcopic evaluation and diagnosis of precancerous changes in the cervix showed higher sensitivity and negative predictive value compared with the specificity and positive predictive value of the method concerning HSIL in the final histological result after LLETZ.
INTRODUCTION
Colposcopy is a specialized test to which screening-referred patients are referred (most commonly after abnormal cervical cytology) (1). The assessment of colposcopic findings is primarily subjective. Hence, colposcopic findings are known as colposcopic impressions and are a function of knowledge, skill, experience, and maintaining competence with continuous and case-rich practice (2).
Objective
To investigate the correlation between colposcopic diagnosis (colposcopic impression) and histological diagnosis after large loop excision of the transformation zone (LLETZ) procedure regarding cervical precancerous lesions - low-grade and high-grade squamous intraepithelial lesion (LSIL, HSIL) and to calculate the sensitivity (negative predictive value – NPV) and specificity (positive predictive value – PPV) of colposcopic diagnosis regarding HSIL in the final histological result after LLETZ.
MATERIAL AND METHODS
Study population
A prospective study from January 1st, 2017, to July 31st, 2021 with total of 189 patients with cervical precancerous lesions diagnosed by colposcopy and biopsy and after that received LLETZ treatment (loop electrosurgical excision) at Prof. Yavor Kornovski Medical Centre in an outpatient setting. An ethical committee approval (number 12-????/12.08.2020) was obtained from all patients.
The indications for the LLETZ procedure are: histologically proven by targeted biopsy under videocolposcopic control HSIL and LSIL, which colposcopically enters the cervical canal and its distal border is not visualized.
The patients’ characteristics are shown table 1.
Table 1 - The characteristics of patients included in the study

Methodology
For this study we used the following methods:
Videocolposcopy
Videocolposcopy – all procedures were performed by one specialist with additional qualification in colposcopy. Until Feb. 10th, 2020 an Alyn Welch device was used, and after that a Leisegang video colposcope with original software and monitor, model 2020. The examination was performed after treatment of the cervix with 5% acetic acid solution, prepared every 2 days, and Lugol’s solution, replaced every month. The colposcopic examination of each patient was saved, documented, and archived in the medical centre's patient database. The colposcopic examination provides information on: adequacy (colposcopy is satisfactory and unsatisfactory); type of TZ; type of cervical lesion (its location relative to the exo- and endocervix); colposcopic diagnosis and cervical lesion size. Colposcopy is satisfactory if the junction between the squamous epithelium and the columnar epithelium (a line called the squamocolumnar junction (SCJ) is fully visible, and unsatisfactory if SCJ is partially visible or invisibe. The type of TZ is divided to type 1 - the SCJ is on the ectocervix; type 2 the SCJ is on the ectocervix; and type 3 - the SCJ is in the cervical canal and invisible. The types of cervical lesion are also divided to three types: type 1 - located entirely on the ectocervix; type 2 - the distal end of the lesion is on the endocervix but is visible; and type 3 - the distal end of the lesion is invisible. Colposcopic diagnosis (colposcopic impression) are reported asLSIL/grade 1 colposcopic findings andHSIL/grade 2 colposcopic findings. Lesion size relative to its involvement of the cervical circumference are reported as up to 1/3; up to 2/3; over 2/3.2.
Histological examination
Histological examination of the biopsy: It is taken with biopsy forceps from the most affected area without anaesthesia. It is performed by the same gynaecologic oncologist who performed the colposcopies. Bleeding is controlled by gauze-based pressure, with a pack soaked in Monsel's solution, by surgical insertion and, as a last resort, by tamponade with a sterile roll gauze for several hours or one day. The histological preparation is placed in a 10% formalin solution. The result is is reported as LSIL [for cervical intraepithelial neoplasia (CIN)1] and HSIL (for CIN2 and CIN3); the same highly qualified pathologist performed the histological examination. Histological examination of the specimen after the LLETZ procedure – it is performed by the same highly qualified pathologist who examined the previous biopsyLLETZ procedure
The procedure was performed by the same gynaecologic oncologist who performed the biopsy under local infiltration anaesthesia with lidocaine. SURTRON electrosurgical apparatus was used (cutting mode: cutting – 100W, and coagulation – 60W; coagulation mode – SOFT 100W 60W), and smoke evacuation apparatus.
Statistical analisys
Data were entered and processed with IBM SPSS Statistics 25.0. and MedCalc Version 19.6.3 statistical package. The significance level for rejecting the null hypothesis was established as p < 0.05. The statistical methods used to assess correlation, sensitivity, specificity, positive and negative predictive values were Fisher-Freeman-Halton exact test p<0.001 and Cramer's test).
RESULTS
The mean age of the included women is of 37.06 ± 8.12 years ranging between 21 and 66.By colposcopy 47 (24.87%) patients are reported as LSIL and 142 (75.13%) as HSIL. The histological results after the LLETZ procedure are: LSIL in 60 patients (31.75%); HSIL in 108 (57.14%); In situ cervical cancer in 14 (7.41%); microinvasive cervical cancer in 7(3.7%).
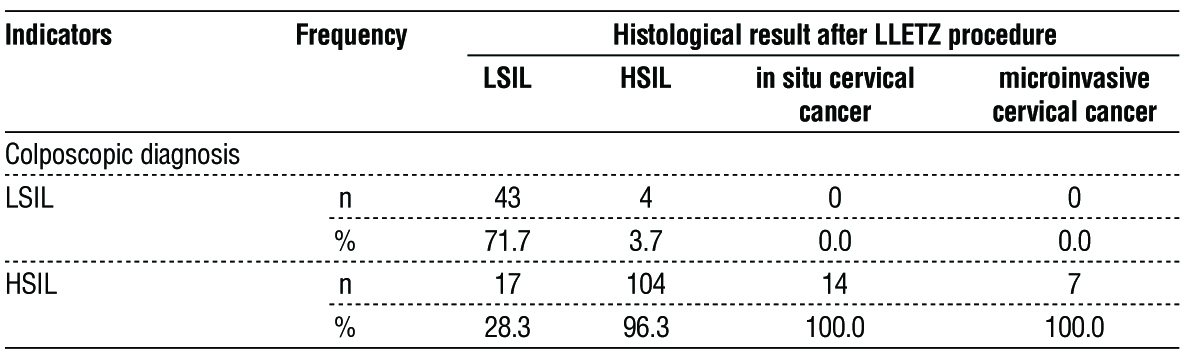
The correlation analysis between the histological result following LLETZ procedure and the colposcopic diagnosis showed that (table 2):
- There is a statistically significant correlation between the two outcomes studied;
- There is a strong correlation – the Cramer's contingency coefficient is 0.739.
Table 2 - Correlation analysis between histological results after LLETZ and colposcopic diagnosis (Fisher-Freeman-Halton exact test p < 0.001, Cramer's V = 0.739)
The sensitivity and negative predictive value of colposcopic diagnosis concerning HSIL in the final histological result after LLETZ were outstanding (96 and 91%, respectively), whereas the specificity and positive predictive value were much lower (53 and 73%, respectively). The magnitude estimation precision was also quite good at 78% (table 3).
Table 3 - Validation criteria values of colposcopic screening tests regarding HSIL in the final histological result after LLETZ

It is noteworthy that all patients diagnose as in situ and microinvasive cervical cancer by LLETZ procedure were reported at colposcopy as HSIL. The total number of these patients is 21 (11.11%) and the sensitivity and positive predictive value is 100%. This means the advanced changes in the cervix lead to an increase of sensitivity of colposcopy. This explain why only 4 (3.7%) patients histological diagnosed as HSIL are reported as LSIL by colposcopy. On the opposite 17 (8.99%) patients were reported as HSIL by colposcopy but were identified as LSIL by histological exam. In LSIL group the colposcopy has a lower sensitivity and positive predictive value.
DISCUSSION
The sensitivity of colposcopy for HSIL in the final histological result is 96%, and the negative predictive value is 91%. The sensitivity of colposcopy for CIN 2+ ranges from 30 to 60% (3-5). A higher sensitivity for HSIL has been reported in two other publications, 85.2% and 85%, respectively (6,7). To increase sensitivity (diagnostic accuracy), some authors recommend multiple biopsies and biopsies from normal-appearing colposcopic areas (8–13). Endocervical curettage (ECC) is not recommended if the squamocolumnar junction (SCJ) is visible (14).
A study by Kabaca et al. confirmed that ECC does not benefit the diagnosis of both LSIL and HSIL (15).
We believe that the most critical factor in increasing both the sensitivity and specificity of colposcopic examination is good expertise, extensive experience of the colposcopist, the use of freshly prepared 3% – 5% acetic acid and good optics with up to 30X magnification.
The specificity of colposcopic diagnosis in our study with respect to HSIL in the final histological result was 53%, and the positive predictive value of colposcopic diagnosis was 73%. With regard to the specificity of colposcopy for HSIL diagnosis, literature data also range widely. A team from Germany reported a 38% PPV of colposcopy (16). Three other studies found 69%, 72.9%, and 80% specificity of the method for CIN 2+ detection, respectively (3,6,7). The introduction of colposcopic indices is a way to quantify, a criterion and an opportunity to reduce subjectivism in the interpretation of colposcopic findings and thus increase the specificity of the method. One colposcopic index is the Swede Colposcopic Index (SCI) introduced by a Swedish group of scientists. Despite its efficacy, this index has some inconveniences related to measuring the cervical lesion size in millimetres and assessing the response to Lugol's solution (17). In a paper in the Journal of Gynecologic Oncology, Rodpenpear et Pataradool proposed a modified SCI (MSCI), replacing the response to Lugol's solution with another sign-localization of the lesion relative to the squamocolumnar junction (SCJ). They suggested that the lesion size be assessed by quadrants of the cervix rather than in millimetres (18). MSCI includes 5 colposcopic features: acetic acid reaction; margins and surface; vessels (mosaic, punctate area); lesion size (1/4, 2/4, >3/4 in a single lesion, or 2 quadrants or 3-4 quadrants in multiple lesions); lesion localization (outer 1/2 of TZ, outer and inner 1/2 of TZ, enters the cervical canal). Each of these factors (criteria) is scored 1 to 3 points.
The authors found that the MSCI of 11 pts provided 84.2% sensitivity and 96.2% specificity for HSIL. The MSCI has a high specificity; thus, the overdiagnosis and overtreatment were only 4%, respectively. According to the Cochrane Colposcopy and Cervical Cytopathology Collaborative Group, an acceptable value for overtreatment should not exceed 10% (19).
The correlation rate between colposcopy and histological outcome after LLETZ with respect to LSIL in our study was 91.5%. Only 8.5% of the colposcopic findings with evidence of low-grade dysplasia were diagnosed as high-grade lesions in the final histology. This is probably due to the invisible all-cervical lesion entering the cervical canal. Regarding the colposcopic findings showing high-grade dysplasia, 88.1% showed a correlation with the final histology, i.e. HSIL, and 11.9% (17/142) showed LSIL. The inconsistency could be attributed to the utilization of colposcope equipment with lower magnification prior to 2019, including some lesions being perceived as high-grade by colposcopic criteria but actually being low-grade. Another interpretation could be the accumulation of colposcopic expertise throughout the duration of the study. In any case, these results support the recommendation for histologic verification by targeted biopsy in colposcopic atypia, including that which is suspective for HSIL. Very often, conditions such as atrophy and inflammation impact the colposcopic diagnosis and cause overdiagnosis. Performing LLETZ without prior biopsy in these cases would be overtreatment. Further evidence of this are the results on the specificity and positive predictive value of colposcopic examination with respect to HSIL versus the final histological outcome.
The literature provides additional evidence on the correlation between the histological result of colposcopically directed biopsy and that of the LLETZ procedure (20–24). In these cases, we can consider the biopsy result as a function of the colposcopic assessment of the cervical lesion. In the study of Kabaca (2014), the histological result of the targeted biopsy showed a lower degree of cervical change compared to the result after LLETZ in 22.9% for CIN1, 37.03% for CIN2 and 12.72% for CIN3/Ca colli uteri in situ (22). In the same study, the biopsy result was superior at 29.16% for CIN1, 40.74% for CIN2, and 15.45% for CIN3/Ca colli uteri in situ. In a publication by Stoler et al., the correlation between colposcopically guided biopsy histology results and the final histology from LLETZ was 42%, with 21% of biopsies showing a lower grade and 36% showing a higher grade of cervical change (23). Factors underlying the correlation were: age, cervical lesion size, number of biopsies and HPV 18 and 18 strain infection. The factors underlying the lower biopsy degrees of cervical alteration compared to histological results from LLETZ were the number of biopsies and HPV 16 and 18 strain infection. The importance of the number of biopsies in the detection and diagnosis of high-grade lesions has also been established in another study (25). According to the authors, performing 3 biopsies from 3 sites increases the sensitivity of biopsy to 96%, regardless of cytological result, HPV status and colposcopic impression. Costa et al. emphasized the factors of age, invisible SCJ, number of quadrants affected (lesion size), and cone width for the diagnosis of severe precancerous changes (26). Much of the discrepancies were due to the lower pre-cancer grade from the targeted biopsy compared to that after the excisional procedure (hypodiagnosis from biopsy). Thus, in a prospective study comparing histological results from biopsy and LLETZ in 170 cases, the authors found frequent inconsistency, primarily due to hypodiagnosis from biopsy (27). The sensitivity and specificity of targeted biopsy for detecting HSIL were estimated to be 74% and 91%, respectively, and the negative and positive predictive values were 48% and 97%, respectively. The biopsy failed to diagnose one case of microinvasive carcinoma and one case of high-grade glandular lesion. The authors concluded that targeted biopsy is unnecessary in colposcopic evidence of HSIL and recommended excisional procedures in these cases. In another study, the sensitivity of biopsy under colposcopic control versus the histological result of LLETZ concerning LSIL (CIN1) and HSIL (CIN2–3) was 50–70% and 55–90%, respectively (28).
Three main factors have been cited as reasons for the hypodiagnosis of biopsy under colposcopic control: the skill of the colposcopist, the qualifications (expertise) of the histopathologist, and the invisible SCJ. The skill of the colposcopist has been shown to influence the colposcopic assessment of a cervical lesion (grade 1 or grade 2) (3). There has been a tendency toward hyperdiagnosis of cervical lesions when the colposcopist's training and experience are insufficient (29). More important remains the skill to recognize smaller areas with HSIL among larger lesions with LSIL from which to take a targeted biopsy. According to Massad et al.'s research, the colposcopic examination was performed on 2825 women, with 2112 having a targeted biopsy taken by trainee colposcopists. After comparing the colposcopic diagnosis (impression) and the histologic biopsy results, the following findings were found: positive predictive value of colposcopy (regardless of the degree and type of change) – 80%; negative predictive value (i.e., normal colposcopic findings) – 68%; sensitivity of colposcopic examination compared to histologic biopsy result – 89%; specificity of colposcopic examination to histologic biopsy result – 52%. The authors reported a low sensitivity with respect to CIN of 2–3 of 56%, demonstrating either a lack of sufficient skill on the part of the trainee colposcopist or deficiencies on the part of the equipment and consumables (3). In another study, the colposcopist's misjudgment in selecting the site from which to take biopsies from larger lesions appeared to be the cause of hypo-diagnosis compared to the histological outcome after LLETZ (30). Studies have demonstrated that targeted biopsy can miss 30 to 50% of existing high-grade lesions and be a misleading factor in the adequate treatment decision (31). To increase the correlation between biopsy and LLETZ results in such cases, the role of HPV status is increasing. Patients positive for HPV 16 and 18 strains are 35 times more likely to develop precancerous disease, even with normal cytology (32). At the same time, women negative for high-risk HPV strains before conization are more likely to have a histological result negative for precancerous disease after LLETZ (33). Therefore, immediate conization (excisional biopsies) is recommended for HPV 16 carriers rather than targeted biopsies, which may lead to hypodiagnosis and delayed treatment. Another critical factor in the hypodiagnostic value of the histologic result of the targeted biopsy versus the final histology after LLETZ is the ability of the histopathologist to differentiate CIN1 from CIN2, which is not always an easy task given the scarcity of tissue material in the histologic specimen from the pinch biopsy (34,35).
The third factor for hypodiagnosis of the targeted biopsy compared to the post-LLETZ biopsy is the invisible GCE. The exocervical lesion may be LGSIL, but as it enters the cervical canal, it may transition to HSIL (36).
Conversely, cases in which colposcopy-directed biopsy provides a higher grade of precancerous disease compared to the histological result after LLETZ can be explained by several hypotheses (34, 37-39). One reason for this type of discrepancy is the presence of a small area of high-grade lesion removed by the pinch biopsy, and no other such lesion exists in the post-LLETZ specimen. A second reason is the presence of multi-focal foci of high-grade dysplasia, especially in the cervical canal remaining outside the cone resection line. This is particularly typical of glandular lesions.
The third reason is the subjective evaluation of the preparations by the histopathologist. The role of the highly skilled and competent pathologist in these cases is significant.
CONCLUSION
The sensitivity of colposcopic diagnosis regarding HSIL in the final histological result was 96%. The specificity of the colposcopic diagnosis regarding HSIL in the final histological result was 53%. The positive predictive value of the colposcopic diagnosis was 73%, and the negative predictive value was 91%. The correlation rate between colposcopy and histological outcome after LLETZ with respect to LSIL in our study was 91.5%. The correlation percentage between colposcopy and histological results after LLETZ regarding HSIL was 88.1%. A biopsy is recommended for histological verification in case of colposcopic evidence of HSIL.
Author’s contributions
All authors have accepted responsibility for the entire content of this manuscript and approved its submission.
Competing interests
Authors state no conflict of interest.
Research funding: None declared.
Research ethics: Not applicable.
Informed consent: Not applicable.
Data availability
The authors declare that all related data are available concerning researchers by the corresponding author’s email.
REFERENCES
1.Ferris DG, Mayeux EJ, Jr, Thomas Cox J. American Society for Colposcopy and Cervical Pathology, editors. Modern colposcopy: textbook & atlas. 3rd ed. Philadelphia, PA: Wolters Kluwer Health/Lippincott Williams & Wilkins; 2012. The colposcopic examination; pp. 120–148.
2.Jordan JA. Colposcopy in the diagnosis of cervical cancer and precancer. Clin Obstet Gynaecol. 1985;12(1):67-76.
3.Massad LS, Collins YC. Strength of correlations between colposcopic impression and biopsy histology. Gynecol Oncol. 2003; 89(3):424-8.
4.Massad LS, Jeronimo J, Katki HA, Schiffman M. The accuracy of colposcopic grading for detection of high-grade cervical intraepithelial neoplasia. J Low Genit Tract Dis. 2009;13(3):137-44.
5.Bekkers RL, Nieuwenhof HPVD, Neesham DE, Hendriks JH, Tan J, Quinn MA. Does experience in colposcopy improve identification of high grade abnormalities? Eur J Obstet Gynecol Reprod Biol. 2008; 141(1):75-8.
6.?widerska-Kiec J, Czajkowski K, Zar?ba-Szczudlik J, Kacperczyk-Bartnik J, Bartnik P, Romejko-Wolniewicz E. Comparison of HPV Testing and Colposcopy in Detecting Cervical Dysplasia in Patients With Cytological Abnormalities. In Vivo. 2020;34(3):1307-1315.
7.Mitchell MF, Schottenfeld D, Tortolero-Luna G, Cantor SB, Richards-Kortum R. Colposcopy for the diagnosis of squamous intraepithelial lesions: a meta-analysis. Obstet Gynecol. 1998;91(4):626-31.
8.Gage JC, Hanson VW, Abbey K, Dippery S, Gardner S, Kubota J, et al. Number of cervical biopsies and sensitivity of colposcopy. Obstet Gynecol. 2006;108(2):264-72.
9.Nakamura Y, Matsumoto K, Satoh T, Nishide K, Nozue A, Shimabukuro K, et al. Optimizing biopsy procedures during colposcopy for women with abnormal cervical cancer screening results: a multicenter prospective study. Int J Clin Oncol. 2015; 20(3):579-85.
10. Jespersen MM, Booth BB, Petersen LK. Can biopsies be omitted after normal colposcopy in women referred with low-grade cervical cytology? A prospective cohort study. BMC Womens Health. 2021; 21(1):394.
11. Pretorius RG, Belinson JL, Burchette RJ, Hu S, Zhang X, Qiao Y-L. Regardless of skill, performing more biopsies increases thesensitivity of colposcopy. J Low Genit Tract Dis. 2011;15(3):180-8.
12. Huh WK, Sideri M, Stoler M, Zhang G, Feldman R, Behrens CM. Relevance of random biopsy at the transformation zone when colposcopy is negative. Obstet Gynecol. 2014;124(4):670-678.
13. Nam K, Chung S, Kwak J, Cha S, Kim J, Jeon S, et al. Random biopsy after colposcopy-directed biopsy improves the diagnosis of cervical intraepithelial neoplasia grade 2 or worse. J Low Genit Tract Dis. 2010;14(4):346-51.
14. Perkins RB, Guido RS, Castle PE, Chelmow D, Einstein MH, Garcia F, et al. 2019 ASCCP risk-based management consensus guidelines for abnormal cervical cancer screening tests and cancer precursors. J Low Genit Tract Dis. 2020; 24(2):102-131.
15. Kabaca C, Giray B, Guray Uzun M, Akis S, Emre Purut Y, Keles Peker E, et al. The meaning of high-risk HPV other than type 16/18 in women with negative cytology: Is it really safe to wait for 1 year? Diagn Cytopathol. 2021;49(4):480-486.
16. Schneider A, Hoyer H, Lotz B, Leistritza S, Kühne-Heid R, Nindl I, et al. Screening for high-grade cervical intra-epithelial neoplasia and cancer by testing for high-risk HPV, routine cytology or colposcopy. Int J Cancer. 2000;89(6):529-34.
17. Boonlikit S. Correlation between Reid's colposcopic index and histologic results from colposcopically directed biopsy in differentiating high-grade from low-grade squamous intraepithelial lesion at Rajavithi Hospital. J Med Assoc Thai. 2011;94 Suppl 2:S59-65.
18. Rodpenpear N, Pataradool KJ. The efficacy of modified Swede Colposcopic Index in prediction of high-grade lesion and cancer of cervix. J Gynecol Oncol. 2019;30(5):e78.
19. Kyrgiou M, Tsoumpou I, Vrekoussis T, Martin-Hirsch P, Arbyn M, Prendiville W, et al. The up-to-date evidence on colposcopy practice and treatment of cervical intraepithelial neoplasia: the Cochrane Colposcopy & Cervical Cytopathology Collaborative Group (C5 group) approach. Cancer Treat Rev. 2006;32(7):516-23.
20. Chappatte OA, Byrne DL, Raju KS, Nayagam M, Kenney A. Histological differences between colposcopic-directed biopsy and loop excision of the transformation zone (LETZ): a cause for concern. Gynecol Oncol. 1991;43(1):46-50.
21. Kjellberg L, Tavelin B. ‘See and treat’ regime by LEEP conisation is a safe and time saving procedure among women with cytological high-grade squamous intraepithelial lesion. Acta Obstet Gynecol Scand. 2007;86(9):1140-4.
22. Kabaca C, Koleli I, Sariibrahim B, Karateke A, Gurbuz A, Kapudere B, et al. Is cervical punch biopsy enough for the management of low-grade cervical intraepithelial neoplasia? J Low Genit Tract Dis. 2014; 18(3):240-5.
23. Stoler MH, Vichnin MD, Ferenczy A, Ferris DG, Perez G, Paavonen J, et al. The accuracy of colposcopic biopsy: analyses from the placebo arm of the Gardasil clinical trials. Int J Cancer. 2011;128(6): 1354-62.
24. Jung Y, Lee AR, Lee SJ, Lee YS, Park DC, Park EK. Clinical factors that affect diagnostic discrepancy between colposcopically directed biopsies and loop electrosurgical excision procedure conization of the uterine cervix. Obstet Gynecol Sci. 2018;61(4):477-488.
25. Nicolas W, Joan LW, Michael AG, Katie MS, Rosemary EZ, Cara M, et al. Multiple biopsies and detection of cervical cancer precursors at colposcopy. J Clin Oncol. 2015;33(1):83-9.
26. Costa S, Nuzzo MD, Rubino A, Rambelli V, Marinelli M, Santini D, et al. Independent determinants of inaccuracy of colposcopically directed punch biopsy of the cervix. Gynecol Oncol. 2003;90(1):57-63.
27. Byrom J, Douce G, Jones PW, Tucker H, Millinship J, Dhar K, et al. Should punch biopsies be used when high-grade disease is suspected at initial colposcopic assessment? A prospective study. Int J Gynecol Cancer. 2006;16(1):253-6.
28. Duesing N, Schwarz J, Choschzick M, Jaenicke F, Gieseking F, Issa R, et al. Assessment of cervical intraepithelial neoplasia (CIN) with colposcopic biopsy and efficacy of loop electrosurgical excision procedure (LEEP). Arch Gynecol Obstet. 2012;286(6):1549-54.
29. Baum ME, Rader JS, Gibb RK, McAlister RP, Powell MA, Mutch DG, et al. Colposcopic accuracy of obstetrics and gynecology residents. Gynecol Oncol. 2006;103(3):966-70.
30. Buxton EJ, Luesley DM, Shafi MI, Rollason M. Colposcopically directed punch biopsy: a potentially misleading investigation. Br J Obstet Gynaecol. 1991;98(12):1273-6.
31. Denny LA, Soeters R, Dehaeck K, Bloch B. Does colposcopically directed punch biopsy reduce the incidence of negative LLETZ? Br J Obstet Gynaecol. 1995;102(7):545-8.
32. Wright TC, Jr, Stoler MH, Behrens CM, Apple R, Derion T, Wright TL. The ATHENA human papillomavirus study: design, methods, and baseline results. Am J Obstet Gynecol. 2012;206(1):46.e1-46.e11.
33. Rodriguez-Manfredi A, Alonso I, del Pino M, Fusté P, Torné A, Ordi J. Predictors of absence of cervical intraepithelial neoplasia in the conization specimen. Gynecol Oncol. 2013;128(2):271-6.
34. Dalla Palma P, Giorgi Rossi P, Collina G, Buccoliero AM, Ghiringhello B, Gilioli E, et al. The reproducibility of CIN diagnoses among different pathologists: data from histology reviews from a multi-center randomized study. Am J Clin Pathol. 2009;132(1):125-32.
35. Martin CM, O‘Leary JJ. Histology of cervical intraepithelial neoplasia and the role of biomarkers. Best Pract Res Clin Obstet Gynaecol. 2011;25(5):605-15.
36. Killackey MA, Jones WB, Lewis JL Jr. Diagnostic conization of the cervix: review of 460 consecutive cases. Obstet Gynecol. 1986; 67(6):766-70.
37. Livasy CA, Moore DT, Van Le L. The clinical significance of a negative loop electrosurgical cone biopsy for high-grade dysplasia. Obstet Gynecol. 2004;104(2):250-4.
38. Ryu A, Nam K, Chung S, Kim J, Lee H, Koh E, et al. Absence of dysplasia in the excised cervix by a loop electrosurgical excision procedure in the treatment of cervical intraepithelial neoplasia. J Gynecol Oncol. 2010;21(2):87-92.
39. Carrigg A, Teschendorf C, Amaro D, Weidner N, Tipps A, Shabaik A, et al. Examination of sources of diagnostic error leading to cervical cone biopsies with no evidence of dysplasia. Am J Clin Pathol. 2013; 139(4):422-7.
Full Text Sources:
Abstract:
Views: 4922

For Authors
Journal Subscriptions

Dec 2025
Supplements
Instructions for authors
Online submission
Contact
e-ISSN: 2601 - 1700 (online)
ISSN-L: 2559 - 723X
Journal Abbreviation: Surg. Gastroenterol. Oncol.
Surgery, Gastroenterology and Oncology (SGO) is indexed in:
- SCOPUS
- EBSCO
- DOI/Crossref
- Google Scholar
- SCImago
- Harvard Library
- Open Academic Journals Index (OAJI)
Surgery, Gastroenterology and Oncology (SGO) is an open-access, peer-reviewed online journal published by Celsius Publishing House. The journal allows readers to read, download, copy, distribute, print, search, or link to the full text of its articles.
Time to first editorial decision: 25 days
Rejection rate: 61%
CiteScore: 0.2

Meetings and Courses in 2025
Meetings and Courses in 2024
Meetings and Courses in 2023
Meetings and Courses in 2022
Meetings and Courses in 2021
Meetings and Courses in 2020
Meetings and Courses in 2019
Verona expert meeting 2019

Surgery, Gastroenterology and Oncology applies the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits readers to copy and redistribute the material in any medium or format, remix, adapt, build upon the published works non-commercially, and license the derivative works on different terms, provided the original material is properly cited and the use is non-commercial. Please see: https://creativecommons.org/licenses/by-nc/4.0/
Publisher’s Note:
The opinions, statements, and data contained in article are solely those of the authors and not of Surgery, Gastroenterology and Oncology journal or the editors. Publisher and the editors disclaim responsibility for any damage resulting from any ideas, instructions, methods, or products referred to in the content.
 IASGO Society News
IASGO Society News