Surgery, Gastroenterology and Oncology
Official journal of the International Association of Surgeons, Gastroenterologists and Oncologists

|
|
Spontaneous Intramural Duodenal Hematoma - A Rare Entity
Spontaneous duodenal hematoma is a rare condition, mainly occurring in relationship to the
administration of anticoagulants. Rare case reports have described an association with
pancreatitis. This paper aims to present the case of a patient with type A hemophilia who
developed spontaneous intramural duodenal hematoma during all-oral antiviral therapy for
chronic HCV hepatitis. Furthermore, the paper reviews existing data on incidence, diagnosis
and management of this condition. Direct acting antiviral agents have proven very safe and
effective in curing hepatitis C (HCV) infection. However, interactions with other drugs and
therapeutic management in case of on-treatment complications have been insufficiently
studied.
INTRODUCTION
Intramural duodenal hematoma is an uncommon condition, mainly resulting from abdominal blunt trauma. Spontaneous hematomas occur in patients with coagulation disorders, endoscopic procedures or pancreatic disease (1). One hospital survey revealed an incidence of 1 in 2500 patients undergoing anticoagulation therapy (2). A retrospective study of published case reports revealed that, out of 10 patients with duodenal hematoma 6 had a history of abdominal trauma and the remaining 4 had a hypocagulatory state or were under anticoagulation therapy (3). The clinical findings of the patients were unspecific, and the diagnosis was made by abdominal computer tomograpy and upper gastrointestinal endoscopy.
In recent years, the development of all oral antiviral therapies for chronic HCV infection has raised the expectations regarding the possibility of HCV erradiacation (4). These therapies have very high efficacy (over 90%), are administered for a short period of time and few adverse reactions are reported.
Currently, treatment options include pan-genotypic and genotype-specific regimens. For genotype 1b treatment with ombitarsvir/paritaprevir/ ritonavir and dasabuvir is associated with sustained virologic response rates of 98%. However, the metabolic particularities of these antiviral agents are associated with complex drug-drug interactions as well as a close relationship to diet and meal hours. In these conditions, the development of a new gastrointestinal condition can create significant management problems.
CASE REPORT
We present the case of a 52-year-old with a history of type A haemophilia and HCV chronic hepatitis, nonresponder to interferon-based therapy. He had been under replacement therapy with factor VIII as needed and he began antiviral therapy with ombitarsvir/paritaprevir/ ritonavir and dasabuvir in February 2016. At initiation of therapy the patient had an HCV-RNA of 200000 U/mL and an F3 degree of fibrosis evaluated by Fibroscan.
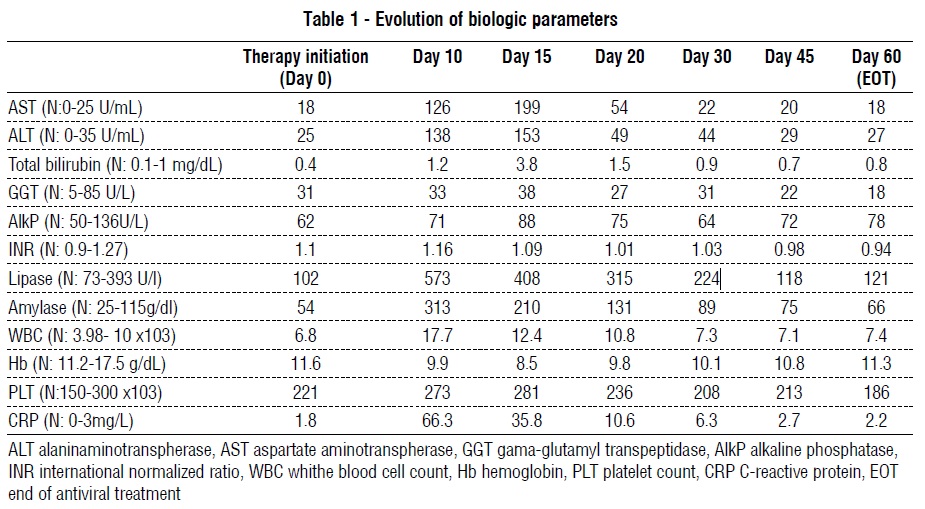
Ten days after therapy initiation, he was admitted with a continuous abdominal pain, mainly in the epigastrium, vomiting and absence of intestinal transit for 4 days. Serum biochemistry and hematology revealed pancreatitis and inflammatory syndrome, associated with liver cytolysis and cholestasis. Biologic parameters are presented in table 1.
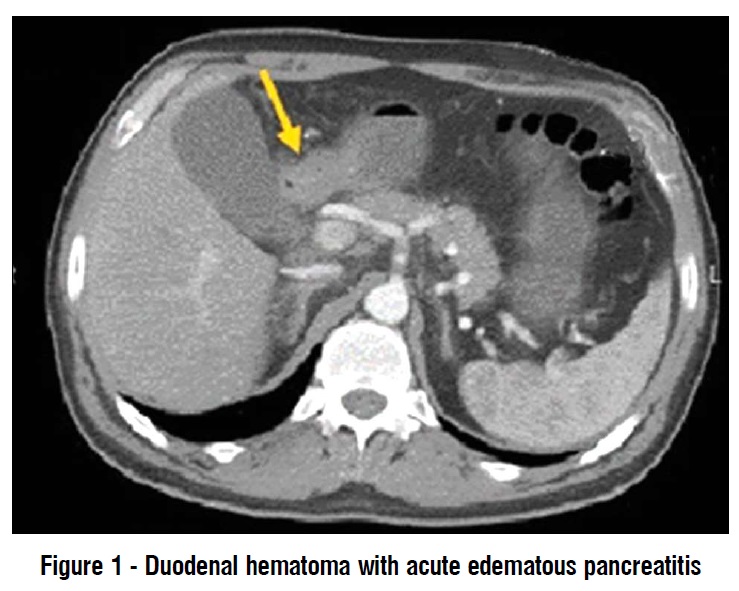
Abdominal ultrasound showed only accelerated intestinal motility. The upper endoscopy revealed stomach with bile content, duodenal edema and duodenal stenosis. A contrast enhanced CT scan of the abdomen was performed. This examination revealed an important duodenal parietal thickening of about 24 mm and acute edematous pancreatitis, a large heterogeneous peri-duodenal mass in the retroperitoneum (fig. 1), obstructing the duodenum (fig. 2).
The patients was given antibiotic therapy (third generation cephalosporin), Factor VIII replacement therapy, proton pump inhibitors 3 times a day, octerotide and symptomatic treatment. The oral antiviral treatment was administered via a nasogastric tube, two hours apart from all other therapies, by fragmentation and dilution in water, despite the producer`s recommendations. To our knowledge, this is the first case of an alternative means of administration for these drugs. Under treatment, serum pancreatic enzymes and inflammatory syndrome diminished, and serologic values normalized in 3 weeks. The nasogastric tube was suppressed and antiviral treatment was continued orally. A month later upper endoscopy and abdominal CT scan were repeated and there were no signs of pancreatitis or hematoma, the duodenum was fully permeable.
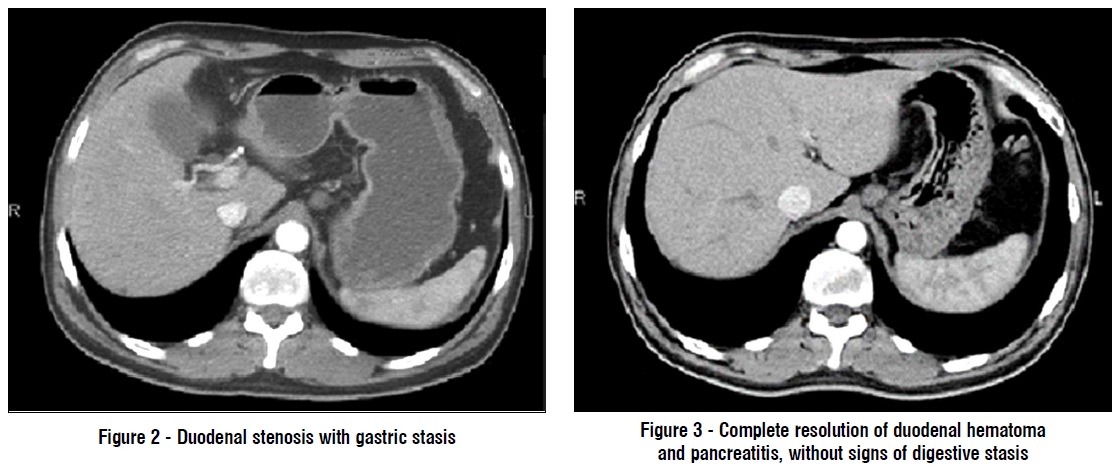
After 3 months the patient was admitted for reevaluation; the CT scan and the MRI scan confirmed the complete resolution of duodenal hematoma and acute pancreatitis (fig. 3). Meanwhile, the antiviral treatment was continued and in August 2016, the patient was declared cured of HCV infection, with sustained virologic response.
DISCUSSION
Intramural duodenal hematoma was initially describe in 1838 by McLauchlan as a "false aneurysmal tumor occupying nearly the whole of the duodenum" at the authopsy of a 49-year-old who died of duodenal obstruction. In 1948 Liverud first describe the radiographic findings associated with intramural intestinal hematoma (5).
Duodenal hematoma is a very rare condition, with rare cases reported in patients with no risk factors or patients undergoing anticoagulant treatment (6,7). In our case, we consider that the patient’s hematoma appeared in the context of hemophilia, irrespective of the antiviral treatment. Symptoms of duodenal hematomas are non-specific, including abdominal pain and small bowel obstruction; hematemesis is a rare occurence (8). This is consistent with the clinical presentation of our patient. Furthermore, cholestasis and pancreatitis may appear due to the obstruction of the papilla of Vater, as was the case we described (9).
The diagnosis requires abdominal CT, usually revealing a large homogenous mass (with hematic densities 50-60 HU) located in the duodenum (3). MRI may be used for a better evaluation of biliary ducts and pancreatic abnormalities (as potential cause or secondary to the hematoma). In our case, contrast-enhanced CT was able to identify pancreatic edema associated to the obstruction of bile ducts and MRI was not required. Therapeutic management for duodenal hematonas is mainly conservative, if patients are in a stable condition. Surgical management is presented in some case reports ( 10,11) describing good clinical evolution but prolonged hospitalization. Another therapeutic option is transarterial embolization, if active bleeding can is visualized (12).
The challenge in our case was tailoring the medical treatment in order to prevent interferences with the antiviral treatment. Antiviral drugs were administered via a naso-gastric tube and in association with increased doses of proton pump inhibitors, with the risk of impaired absorption. However, the follow-up of the patient revealed sustained virologic response. In addition, concomitant administration of antibiotics increased the risk of altered antiviral metabolism. On the other hand, antiviral treatment has been associated with liver cytolysis and cholestasis during the first weeks of therapy with spontaneous remission (13). As such, in our case, the biologic alterations described may have had a dual cause.
CONCLUSION
In conclusion, we strongly advise that the management of HCV infected patients should include active monitoring of commorbidities, during and after antiviral therapy, as unrelated complication may arise, imposing treatment difficulties and increased risks.
Conflict of interests
The authors declare no conflicts of interests.
Ethical approval
All procedures performed were in accordance with the ethical standards of the 1964 Helsinki Declaration and its later amendments.
REFERENCES
1. Eurboonyanun C, Somsap K, Ruangwannasak S, Sripanaskul A. Spontaneous intramural duodenal hematoma: pancreatitis, obstructive jaundice, and upper intestinal obstruction. Case Rep Surg. 2016;2016:5321081.
2. Bettler S, Montani S, Bachmann F. Incidence of intramural digestive system hematoma in anticoagulation. Epidemiologic study and clinical aspects of 59 cases observed in Switzerland (1970-1975). Schweiz Med Wochenschr. 1983;113(17):630-6. French
3. Niehues SM, Denecke T, Bassir C, Hamm B, Haas M. Intramural duodenal hematoma: clinical course and imaging findings. Acta Radiol Open. 2019;8(4):2058460119836256.
4. European Association for the Study of the Liver. EASL recommendations on treatment of hepatitis C: Final update of the series. J Hepatol. 2020;73(5):1170-1218
5. Jones WR, Hardin WJ, Davis JT, Hardy JD. Intramural hematoma of the duodenum: a review of the literature and case report. Ann Surg. 1971;173(4):534-44.
6. Carrilero Zaragoza G, Egea Valenzuela J, Moya Arnao M, Muñoz Tornero M, Jijón Crespín R, Tomas Pujante P, et al. Spontaneous retroperitoneal hematoma in a patient under anticoagulant agents presenting as upper gastrointestinal bleeding. Rev Esp Enferm Dig. 2016;108(12):817-818.
7. Wong K, Thomas R. A curious case of spontaneous duodenal haematoma presenting a diagnostic challenge. BMJ Case Rep. 2014;2014:bcr2013200614. doi: 10.1136/bcr-2013-200614.
8. Veldt BJ, Haringsma J, Florijn KW, Kuipers EJ. Coumarin-induced intramural hematoma of the duodenum: case report and review of the literature. Scand J Gastroenterol. 2011;46(3):376-9.
9. Sadry F, Hauser H. Fatal pancreatitis secondary to iatrogenic intramural duodenal hematoma: a case report and review of the literature. Gastrointest Radiol. Fall 1990;15(4):296-8.
10. Han SJ, Tsai CC, Mo LR, Tseng LJ, Yau MP. Laparoscopic finding and imaging of the iatrogenic duodenal intramural hematoma. Hepatogastroenterology. 1997;44(13):139-42.
11. Touloukian RJ. Protocol for the nonoperative treatment of obstructing intramural duodenal hematoma during childhood. Am J Surg. 1983;145(3):330-4.
12. Dunne R, McCarthy E, Joyce E, McEniff N, Guiney M, Ryan JM, et al. Post-endoscopic biliary sphincterotomy bleeding: an interventional radiology approach. Acta Radiol. 2013;54(10):1159-64.
13. Welzel TM, Hinrichsen H, Sarrazin C, Buggisch P, Baumgarten A, Christensen S, et al. Real-world experience with the all-oral, interferon-free regimen of ombitasvir/paritaprevir/ritonavir and dasabuvir for the treatment of chronic hepatitis C virus infection in the German Hepatitis C Registry. J Viral Hepat. 2017;24(10): 840-849.
Full Text Sources:
Abstract:
Views: 3468

INTRODUCTION
Intramural duodenal hematoma is an uncommon condition, mainly resulting from abdominal blunt trauma. Spontaneous hematomas occur in patients with coagulation disorders, endoscopic procedures or pancreatic disease (1). One hospital survey revealed an incidence of 1 in 2500 patients undergoing anticoagulation therapy (2). A retrospective study of published case reports revealed that, out of 10 patients with duodenal hematoma 6 had a history of abdominal trauma and the remaining 4 had a hypocagulatory state or were under anticoagulation therapy (3). The clinical findings of the patients were unspecific, and the diagnosis was made by abdominal computer tomograpy and upper gastrointestinal endoscopy.
In recent years, the development of all oral antiviral therapies for chronic HCV infection has raised the expectations regarding the possibility of HCV erradiacation (4). These therapies have very high efficacy (over 90%), are administered for a short period of time and few adverse reactions are reported.
Currently, treatment options include pan-genotypic and genotype-specific regimens. For genotype 1b treatment with ombitarsvir/paritaprevir/ ritonavir and dasabuvir is associated with sustained virologic response rates of 98%. However, the metabolic particularities of these antiviral agents are associated with complex drug-drug interactions as well as a close relationship to diet and meal hours. In these conditions, the development of a new gastrointestinal condition can create significant management problems.

CASE REPORT
We present the case of a 52-year-old with a history of type A haemophilia and HCV chronic hepatitis, nonresponder to interferon-based therapy. He had been under replacement therapy with factor VIII as needed and he began antiviral therapy with ombitarsvir/paritaprevir/ ritonavir and dasabuvir in February 2016. At initiation of therapy the patient had an HCV-RNA of 200000 U/mL and an F3 degree of fibrosis evaluated by Fibroscan.
Ten days after therapy initiation, he was admitted with a continuous abdominal pain, mainly in the epigastrium, vomiting and absence of intestinal transit for 4 days. Serum biochemistry and hematology revealed pancreatitis and inflammatory syndrome, associated with liver cytolysis and cholestasis. Biologic parameters are presented in table 1.

Abdominal ultrasound showed only accelerated intestinal motility. The upper endoscopy revealed stomach with bile content, duodenal edema and duodenal stenosis. A contrast enhanced CT scan of the abdomen was performed. This examination revealed an important duodenal parietal thickening of about 24 mm and acute edematous pancreatitis, a large heterogeneous peri-duodenal mass in the retroperitoneum (fig. 1), obstructing the duodenum (fig. 2).
The patients was given antibiotic therapy (third generation cephalosporin), Factor VIII replacement therapy, proton pump inhibitors 3 times a day, octerotide and symptomatic treatment. The oral antiviral treatment was administered via a nasogastric tube, two hours apart from all other therapies, by fragmentation and dilution in water, despite the producer`s recommendations. To our knowledge, this is the first case of an alternative means of administration for these drugs. Under treatment, serum pancreatic enzymes and inflammatory syndrome diminished, and serologic values normalized in 3 weeks. The nasogastric tube was suppressed and antiviral treatment was continued orally. A month later upper endoscopy and abdominal CT scan were repeated and there were no signs of pancreatitis or hematoma, the duodenum was fully permeable.

After 3 months the patient was admitted for reevaluation; the CT scan and the MRI scan confirmed the complete resolution of duodenal hematoma and acute pancreatitis (fig. 3). Meanwhile, the antiviral treatment was continued and in August 2016, the patient was declared cured of HCV infection, with sustained virologic response.
DISCUSSION
Intramural duodenal hematoma was initially describe in 1838 by McLauchlan as a "false aneurysmal tumor occupying nearly the whole of the duodenum" at the authopsy of a 49-year-old who died of duodenal obstruction. In 1948 Liverud first describe the radiographic findings associated with intramural intestinal hematoma (5).
Duodenal hematoma is a very rare condition, with rare cases reported in patients with no risk factors or patients undergoing anticoagulant treatment (6,7). In our case, we consider that the patient’s hematoma appeared in the context of hemophilia, irrespective of the antiviral treatment. Symptoms of duodenal hematomas are non-specific, including abdominal pain and small bowel obstruction; hematemesis is a rare occurence (8). This is consistent with the clinical presentation of our patient. Furthermore, cholestasis and pancreatitis may appear due to the obstruction of the papilla of Vater, as was the case we described (9).
The diagnosis requires abdominal CT, usually revealing a large homogenous mass (with hematic densities 50-60 HU) located in the duodenum (3). MRI may be used for a better evaluation of biliary ducts and pancreatic abnormalities (as potential cause or secondary to the hematoma). In our case, contrast-enhanced CT was able to identify pancreatic edema associated to the obstruction of bile ducts and MRI was not required. Therapeutic management for duodenal hematonas is mainly conservative, if patients are in a stable condition. Surgical management is presented in some case reports ( 10,11) describing good clinical evolution but prolonged hospitalization. Another therapeutic option is transarterial embolization, if active bleeding can is visualized (12).
The challenge in our case was tailoring the medical treatment in order to prevent interferences with the antiviral treatment. Antiviral drugs were administered via a naso-gastric tube and in association with increased doses of proton pump inhibitors, with the risk of impaired absorption. However, the follow-up of the patient revealed sustained virologic response. In addition, concomitant administration of antibiotics increased the risk of altered antiviral metabolism. On the other hand, antiviral treatment has been associated with liver cytolysis and cholestasis during the first weeks of therapy with spontaneous remission (13). As such, in our case, the biologic alterations described may have had a dual cause.
CONCLUSION
In conclusion, we strongly advise that the management of HCV infected patients should include active monitoring of commorbidities, during and after antiviral therapy, as unrelated complication may arise, imposing treatment difficulties and increased risks.
Conflict of interests
The authors declare no conflicts of interests.
Ethical approval
All procedures performed were in accordance with the ethical standards of the 1964 Helsinki Declaration and its later amendments.
REFERENCES
1. Eurboonyanun C, Somsap K, Ruangwannasak S, Sripanaskul A. Spontaneous intramural duodenal hematoma: pancreatitis, obstructive jaundice, and upper intestinal obstruction. Case Rep Surg. 2016;2016:5321081.
2. Bettler S, Montani S, Bachmann F. Incidence of intramural digestive system hematoma in anticoagulation. Epidemiologic study and clinical aspects of 59 cases observed in Switzerland (1970-1975). Schweiz Med Wochenschr. 1983;113(17):630-6. French
3. Niehues SM, Denecke T, Bassir C, Hamm B, Haas M. Intramural duodenal hematoma: clinical course and imaging findings. Acta Radiol Open. 2019;8(4):2058460119836256.
4. European Association for the Study of the Liver. EASL recommendations on treatment of hepatitis C: Final update of the series. J Hepatol. 2020;73(5):1170-1218
5. Jones WR, Hardin WJ, Davis JT, Hardy JD. Intramural hematoma of the duodenum: a review of the literature and case report. Ann Surg. 1971;173(4):534-44.
6. Carrilero Zaragoza G, Egea Valenzuela J, Moya Arnao M, Muñoz Tornero M, Jijón Crespín R, Tomas Pujante P, et al. Spontaneous retroperitoneal hematoma in a patient under anticoagulant agents presenting as upper gastrointestinal bleeding. Rev Esp Enferm Dig. 2016;108(12):817-818.
7. Wong K, Thomas R. A curious case of spontaneous duodenal haematoma presenting a diagnostic challenge. BMJ Case Rep. 2014;2014:bcr2013200614. doi: 10.1136/bcr-2013-200614.
8. Veldt BJ, Haringsma J, Florijn KW, Kuipers EJ. Coumarin-induced intramural hematoma of the duodenum: case report and review of the literature. Scand J Gastroenterol. 2011;46(3):376-9.
9. Sadry F, Hauser H. Fatal pancreatitis secondary to iatrogenic intramural duodenal hematoma: a case report and review of the literature. Gastrointest Radiol. Fall 1990;15(4):296-8.
10. Han SJ, Tsai CC, Mo LR, Tseng LJ, Yau MP. Laparoscopic finding and imaging of the iatrogenic duodenal intramural hematoma. Hepatogastroenterology. 1997;44(13):139-42.
11. Touloukian RJ. Protocol for the nonoperative treatment of obstructing intramural duodenal hematoma during childhood. Am J Surg. 1983;145(3):330-4.
12. Dunne R, McCarthy E, Joyce E, McEniff N, Guiney M, Ryan JM, et al. Post-endoscopic biliary sphincterotomy bleeding: an interventional radiology approach. Acta Radiol. 2013;54(10):1159-64.
13. Welzel TM, Hinrichsen H, Sarrazin C, Buggisch P, Baumgarten A, Christensen S, et al. Real-world experience with the all-oral, interferon-free regimen of ombitasvir/paritaprevir/ritonavir and dasabuvir for the treatment of chronic hepatitis C virus infection in the German Hepatitis C Registry. J Viral Hepat. 2017;24(10): 840-849.
Full Text Sources:
Abstract:
Views: 3468

Watch Video Articles
For Authors
Journal Subscriptions

Current Issue
Dec 2025
Supplements
Instructions for authors
Online submission
Contact
ISSN: 2559 - 723X (print)
e-ISSN: 2601 - 1700 (online)
ISSN-L: 2559 - 723X
Journal Abbreviation: Surg. Gastroenterol. Oncol.
Surgery, Gastroenterology and Oncology (SGO) is indexed in:
e-ISSN: 2601 - 1700 (online)
ISSN-L: 2559 - 723X
Journal Abbreviation: Surg. Gastroenterol. Oncol.
Surgery, Gastroenterology and Oncology (SGO) is indexed in:
- SCOPUS
- EBSCO
- DOI/Crossref
- Google Scholar
- SCImago
- Harvard Library
- Open Academic Journals Index (OAJI)
Open Access Statement
Surgery, Gastroenterology and Oncology (SGO) is an open-access, peer-reviewed online journal published by Celsius Publishing House. The journal allows readers to read, download, copy, distribute, print, search, or link to the full text of its articles.
Surgery, Gastroenterology and Oncology (SGO) is an open-access, peer-reviewed online journal published by Celsius Publishing House. The journal allows readers to read, download, copy, distribute, print, search, or link to the full text of its articles.
Journal Metrics
Time to first editorial decision: 25 days
Rejection rate: 61%
CiteScore: 0.2
Time to first editorial decision: 25 days
Rejection rate: 61%
CiteScore: 0.2

Meetings and Courses in 2025
Meetings and Courses in 2024
Meetings and Courses in 2023
Meetings and Courses in 2022
Meetings and Courses in 2021
Meetings and Courses in 2020
Meetings and Courses in 2019
Verona expert meeting 2019

Surgery, Gastroenterology and Oncology applies the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits readers to copy and redistribute the material in any medium or format, remix, adapt, build upon the published works non-commercially, and license the derivative works on different terms, provided the original material is properly cited and the use is non-commercial. Please see: https://creativecommons.org/licenses/by-nc/4.0/
Publisher’s Note:
The opinions, statements, and data contained in article are solely those of the authors and not of Surgery, Gastroenterology and Oncology journal or the editors. Publisher and the editors disclaim responsibility for any damage resulting from any ideas, instructions, methods, or products referred to in the content.
 IASGO Society News
IASGO Society News