Surgery, Gastroenterology and Oncology
Official journal of the International Association of Surgeons, Gastroenterologists and Oncologists

|
|
Temperature, Neutrophils and Multiple Organ Failure Score: A Simple Scoring System to Predict Mortality Following Perforated Peptic Ulcer
Purposes: Patients with perforated peptic ulcer (PPU) present with signs of sepsis and
appropriate management can be offered to achieve an optimal outcome of disease. We
propose evaluating the severity of intra-abdominal sepsis in case of PPU with a new score
called TNM, name borrowed by cancer staging, with the aim of assess its predictive value.
Methods: We included 183 patients with diagnosis of complicated PPU. We defined categories T (Temperature), N (Neutrophils count) and M (MOF); then, patients were grouped in stages (0-IV). Variables analysed were age, sex, ASA, blood transfusion, causes of sepsis, temperature, neutrophils count, preoperative organ failure, immune-compromised status, stage (0-IV).
Results: Patients were grouped as follows: none at stage 0; 6 at stage I; 72 at stage II, 72 at stage III; 33 at stage IV. ASA score, neutrophils count, preoperative organ failure, stage IIIIV emerged as statistically significant different prognostic factors. ASA score and stage were significant independent predictors of post-operative mortality in multivariate analysis.
Conclusion: Our proposed system could define and help to assess the mortality risk.
INTRODUCTION
Peptic ulcer disease, both duodenal and gastric, despite the widespread availability of effective acid reduction agents and antibiotic therapy for Helicobacter pylori (1), is associated with potentially life-threatening complications, including bleeding, perforation, penetration and obstruction. Intraabdominal sepsis (IAS) after perforation is the second most frequent complication after bleeding (2,3). A high risk for morbidity (20-50%) and mortality (1.3-40%) is encountered in surgically treated perforated peptic ulcer (PPU) patients (4-9). Patients with PPU present with signs of sepsis and by a careful preoperative assessment of the patients’ severity grade, appropriate management can be offered to achieve an optimal outcome of disease (10,11). Many scoring systems (Boey score, Peptic Ulcer Perforated (PULP) Score, American Society of Anaesthesiologists (ASA) score) have been proposed to predict mortality after PPU (12-14). PULP score seems to be the most reliable, but it is very complex to use (12). Boey score is easier but its predictability value is not consistent (12,15-17). ASA score is a general surgical risk score not intended for PPU patients in particular, and its major drawback is its subjective assessment (12,15). Nowadays, in the clinical practice the grading systems are not always employed for PPU, although they seem to give precise clinical indications, because some of them are too complicated (PULP score) and others are too aspecific (ASA score). In our work, we tried to assess the severity of IAS as a complication of PPU using a new TNM score: T indicates Temperature, N Neutrophil count and M Multiple organ failure (MOF) (18,19). In this study we aimed to evaluate significance of this score to predict mortality of patients with complicated PPU.
MATERIALS AND METHODS
The TNM system was studied in 183 patients with complicated PPU and IAS, managed in General Surgery and Hepato-biliopancreatic Surgery at our Department of Surgery in the period between April 2012 and December 2019. Pregnant women, patients aged < 18, immune-compromised patients and those who underwent laparoscopic surgery were excluded.

At the presentation, patients were clinically evaluated; blood tests and imaging exams were performed. Intravenous antibiotic therapy was set up: Ciprofloxacin 200 mg or Amoxicillin-clavulanic 2 gr and Metronidazole 500 mg.
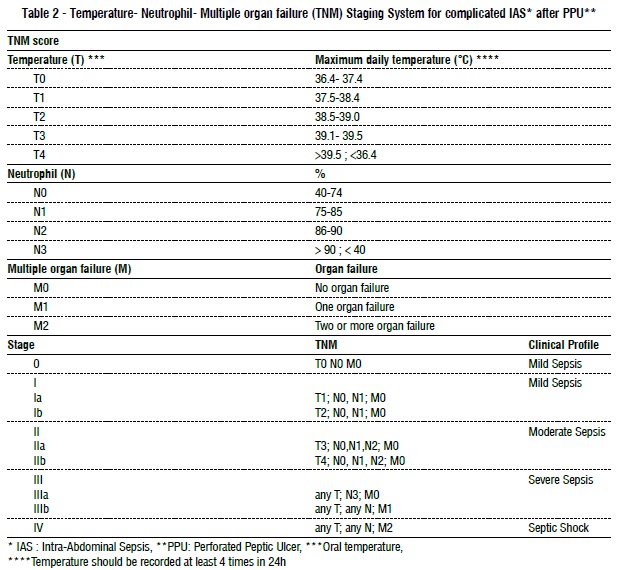
The anthropometric data were collected in an electronic database. According to clinical and laboratory characteristics, the patients were classified based on our system. Table 1 resumes the definitions. The classes of the patients is showed in table 2, which also shows the groupings in stages (stage 0-IV).
For the study of this system, we used retrospective data of 102 patients between January 2001 and January 2012 (control group); the study group was prospectively evaluated. TNM stage was firstly evaluated at the time of the presentation and then every day of recovery. The primary endpoint was to assess the efficacy of TNM score in forecasting mortality at 30 days. The work has been reported in line with the STROCSS criteria (20).
Statistical analysis
The characteristics of the study sample were analysed with descriptive statistics; the discrete and nominal variables were expressed using frequencies and percentages; for continuous variables, medians and range were reported. The frequency distribution of prognostic factors (age classes, sex, ASA score, blood transfusion, causes of sepsis, fever, neutrophil count, pre-operative organ failure, immuno-compromised status, TNM stage) were examined between outcome groups (alive or dead). Chi square ( 2) test was used to analyse statistical differences. Variables significantly different between the two groups were introduced in the multivariate logistic model to obtain independent predictors of death, with associations reported as odds ratios (ORs) and 95% confidence intervals (CIs). Model discrimination was evaluated using the receiver operating characteristics (ROC) curve. All data were electronically recorded; statistical analyses were performed using the Stata Statistical Software (Release 15/IC, College Station, TX: Stata Corp LP). All the tests were two-tailed, and p < 0.05 was considered statistically significant.
RESULTS
One hundred eighty-three consecutive patients were included; they had a mean age of 67.0 years (range 23 to 86). No significative differences of age between the sexes was reported. One hundred and seventeen patients (63.9%) were diagnosed with localized peritonitis or abscesses and sixty-six (36%) with generalized peritonitis.
The model has a good predictive power being the area under the ROC curve equal to 0.8058 (standard error 0.0342) (fig. 1). In the control group retrospectively analysed death occurred in 33.3% of patients, with no significant difference from the study group. The mortality increased among stages (13.1% at stage II, 28.5% at stage III and 100% at stage IV).
DISCUSSION
Mortality is a serious complication in PPU. PPU carries a mortality ranging from 1.3% to 40% (4-9,21, 22). The mortality rate is as high as 12%-47% in elderly patients undergoing PPU surgery (23-25). Significant risk factors that lead to death are presence of patients factors (age > 65 years-old, female, underweight, presence of comorbidities, delay in presentation more than 24h, non-steroidal anti-inflammatory or steroid use), disease factors (shock at presentation, elevated urea or creatinine, metabolic acidosis, anemia, hypoalbuminemia), and treatment factors (resection surgery, blood transfusion, intensive care units) (26-33). Several different scoring systems used to predict outcome in PPU can be identified through the literature: the Boey score, the ASA score, the Sepsis score (SS), the Charlson Comorbidity Index (CCI), the Mannheim Peritonitis Index (MPI), the Acute Physiology and Chronic Health Evaluation II (APACHE II), the Simplified Acute Physiology score II (SAPS II), the Physiology and Operative Severity Score for the Enumeration of Mortality and Morbidity Physical Sub-score ( POSSUMphys score), the Mortality Probability Models II (MPM II), the PULP score, the Hacettepe score (HS), the Jabalpur score (JS), the Practical Scoring System of Mortality in Patients with Perforated Peptic Ulcer (POMPP) score, and the American Association for the Surgery of Trauma (AAST) Emergency General Surgical (EGS) grading system (AAST EGS grade) (34-37).
Anbalakan K. et al have validated ASA score, Boey’s score, MPI and PULP score and found that all the four systems have moderate accuracy of predicting mortality with area under the receiver operator curve of 72%- 77.2% (37). Other scoring systems are not widely used due a lack of validation or their complexity in clinical use. Our new scoring system (18,19) is simple to use and it seems to be a good predictor of mortality. We believe that the initial TNM stage can be easily adopted in the clinical practice to predict the surgical mortality of PPU patients. Early detection of patients at higher risk could be useful to choose other treatment strategies except surgery to decrease the risk of mortality. More consistent and careful perioperative cares should be adopted, among which respiratory support, circulatory stabilization and frequent monitorization (12,38). To early stage patients, a simple grading system may provide reduction in mortality rates.
The death rates related to complicated IAS is reported to be about 1% (39), 6.7% (40) up to 60% (41-50). The most important variable to explains the difference could be the heterogeneous population of patients and procedures (41,43,51-62). Both the anatomic source of infections and the physiologic impairment affect the outcome (63-67). In our present study we selected a homogeneous sample with the same diagnosis (complicated peptic ulcer), same operation (urgent open repair), same surgical incision (midline laparotomy).
Our results showed that TNM could help to classify patients based on their mortality risk. Moreover, some variables seem to be related to mortality: TNM stages III-IV, ASA score III-IV, neutrophil count and preoperative organ failure. Multivariate analysis, in fact, showed that TNM stage IV and ASA score IV themselves significantly influenced the mortality. Indeed, 90.9% (30/33) of the patients at stage IV died, and the high mortality rate (100%) for M2 patients was mainly reported for patients in the first period of the study (retrospective analysis), when treatment was still not so aggressive as in the last cases considered.
Our grading systems is simple and it allows a reevaluation of the patients based on the clinical picture. Some limitations have to be underlined. The prolonged period of data collection and the small sample size are the main ones, because these factors may influence the evaluation of the TNM. Indeed, our study population was only 183 patients, but this number was noticeable when compared with other studies in the literature (6,68-75), except cohort study of Møller 12 and the study of Hernandez (36). A large-scale clinical trial should be evaluated.
CONCLUSION
In our preliminary study, we want to describe our results about the use of TNM score to assess IAS after PPU. This “transfer” of TNM from cancer pathology to septic pathology could prove, if other studies confirm our results, to be extremely effective to define the mortality risk in patients with IAS after PPU.
Supported and conflict of interest statement
The authors declare no dedicated source of funding and no conflicts of interest related to this publication.
Ethics approval and consent to participate
This is an observational clinical study, so ethics approval is not required. Informed consent was obtained from all individual participants included in the study.
Competing interest
The authors declare that they have no conflict of interest.
Funding: No funding.
Authors contribution
M.S. and F.C. provided study conception and design. B.P., L.R., A.G. have acquired the data. A.M. analysed and interpreted these data. L.R. drafted the manuscript. All authors revised, read and approved the final manuscript.
REFERENCES
1. Testini M, Portincasa P, Piccinni G, Lissidini G, Pellegrini F, Greco L. Significant factors associated with fatal outcome in emergency open surgery for perforated peptic ulcer. World J Gastroenterol. 2003; 9(10):2338-40.
2. Milosavljevic T, Kostic-Milosavljevic M, Jovanovic I, Krstic M. Complications of peptic ulcer disease. Dig Dis 2011; 29(5):491–493.
3. Beatrice P, Lucia R, Antonio G, Domenico G, Mario S, Francesco C, et al. Rare case of upper gastrointestinal bleeding: Dieulafoy' s lesion of duodenum. A case report. Ann Med Surg (Lond). 2019; 45:19-21.
4. Christensen S, Riis A, Norgaard M, Sørensen HT, Thomsen RW. Short-term mortality after perforated or bleeding peptic ulcer among elderly patients: a population-based cohort study. BMC Geriatr 2007; 7:8.
5. Christiansen C, Christensen S, Riis A, Thomsen RW, Johnsen SP, Tonnesen E, et al. Antipsychotic drugs and short-term mortality after peptic ulcer perforation: a population-based cohort study. Aliment Pharmacol Ther 2008;28(7):895–902.
6. Thorsen K, Glomsaker TB, von Meer A, Soreide K, Soreide JA. Trends in diagnosis and surgical management of patients with perforated peptic ulcer. J Gastrointest Surg 2011;15(8):1329–1335.
7. Bertleff MJ, Lange JF. Perforated peptic ulcer disease: a review of history and treatment. Dig Surg 2010;27(3):161–169.
8. Lau JY, Sung J, Hill C, Henderson C, Howden CW, Metz DC. Systematic review of the epidemiology of complicated peptic ulcer disease: incidence, recurrence, risk factors and mortality. Digestion 2011;84(2):102–113.
9. Bae S, Shim KN, Kim N, Kang JM, Kim DS, Kim KM, et al. Incidence and short-term mortality from perforated peptic ulcer in korea: a population-based study. J Epidemiol 2012;22(6):508–516.
10. Moller MH, Shah K, Bendix J, Jensen AG, Zimmermann-Nielsen E, Adamsen S, et al. Risk factors in patients surgically treated for peptic ulcer perforation. Scand J Gastroenterol 2009; 44(2):145–152.
11. Moller MH, Adamsen S, Thomsen RW, Moller AM. Multicentre trial of a perioperative protocol to reduce mortality in patients with peptic ulcer perforation. Br J Surg 2011; 98(6):802–810.
12. Møller MH, Engebjerg MC, Adamsen S, Bendix J, Thomsen RW. The Peptic Ulcer Perforation (PULP) score: a predictor of mortality following peptic ulcer perforation. A cohort study. Acta Anaesthesiol Scand. 2012; 56:655–62.
13. Boey J, Choi SK, Poon A, Alagaratnam TT. Risk stratification in perforated duodenal ulcers. A prospective validation of predictive factors. Ann Surg 1987;205:22–6.
14. Mäkelä JT, Kiviniemi H, Ohtonen P, Laitinen SO. Factors that predict morbidity and mortality in patients with perforated peptic ulcers. Eur J Surg 2002;168:446–51.
15. Thorsen K, Søreide JA, Søreide K. Scoring systems for outcome prediction in patients with perforated peptic ulcer. Scand J Trauma Resusc Emerg Med 2013;21:25.
16. Thorsen K, Søreide JA, Søreide K. What is the best predictor of mortality in perforated peptic ulcer disease? A population-based, multivariable regression analysis including three clinical scoring systems. J Gastrointest Surg 2014;18:1261–8.
17. Mishra A, Sharma D, Raina VK. A simplified prognostic scoring system for peptic ulcer perforation in developing countries. Indian J Gastroenterol 2003;22:49–53.
18. Schietroma M, Pessia B, Mattei A, Romano L, Giuliani A, Carlei F. Temperature-Neutrophils-Multiple Organ Failure Grading for Complicated Intra-Abdominal Infections. Surg Infect (Larchmt) 2020;21:69-74.
19. Schietroma M, Romano L, Pessia B, Mattei A, Fiasca F, Carlei F, et al. TNM: a simple classification system for complicated intraabdominal sepsis after acute appendicitis [published online ahead of print, 2020 Aug 6]. Minerva Chir. 2020;10.23736/S0026- 4733.20.08274-7.
20. Agha R, Abdall-Razak A, Crossley E, Dowlut N, Iosifidis C, Mathew G; STROCSS Group. STROCSS 2019 Guideline: Strengthening the reporting of cohort studies in surgery. Int J Surg 2019;72:156-165.
21. Hermansson M, Staël von Holstein C, Zilling T. Surgical approach and prognostic factors after peptic ulcer perforation. Eur J Surg 1999; 165: 566-572.
22. Rajesh V, Chandra SS, Smile SR. Risk factors predicting operative mortality in perforated peptic ulcer disease. Trop Gastroenterol 2003; 24: 148-150.
23. Blomgren LG. Perforated peptic ulcer: long-term results after simple closure in the elderly. World J Surg 1997; 21:412-414.
24. Svanes C, Salvesen H, Stangeland L, Svanes K, Søreide O. Perforated peptic ulcer over 56 years. Time trends in patients and disease characteristics. Gut 1993; 34:1666-1671.
25. Bulut OB, Rasmussen C, Fischer A. Acute surgical treatment of complicated peptic ulcers with special reference to the elderly. World J Surg 1996; 20: 574-577.
26. Buck DL, Møller MH. Influence of body mass index on mortality after surgery for perforated peptic ulcer. Br J Surg 2014; 101: 993-999.
27. Noguiera C, Silva AS, Santos JN, Silva AG, Ferreira J, Matos E, et al. Perforated peptic ulcer: main factors of morbidity and mortality. World J Surg 2003; 27:782-787.
28. Agrez MV, Henry DA, Senthiselvan S, Duggan JM. Changing trends in perforated peptic ulcer during the past 45 years. Aust N Z J Surg 1992; 62:729-732.
29. Svanes C, Lie RT, Lie SA, Kv?le G, Svanes K, S?reide O. Survival after peptic ulcer perforation: a time trend analysis. J Clin Epidemiol 1996; 49:1363-1371.
30. Walt R, Katschinski B, Logan R, Ashley J, Langman M. Rising frequency of ulcer perforation in elderly people in the United Kingdom. Lancet 1986; 1: 489-492.
31. Kocer B, Surmeli S, Solak C, Unal B, Bozkurt B, Yildirim O, et al. Factors affecting mortality and morbidity in patients with peptic ulcer perforation. J Gastroenterol Hepatol 2007;22:565-570.
32. Schietroma M, Colozzi S, Romano L, Pessia B, Giuliani A, Vicentini V, et al. Short- and long-term results after laparoscopic floppy Nissen fundoplication in elderly versus non-elderly patients. J Minim Access Surg. 2020;16(3):256 263.
33. Giuliani A, Romano L, Papale E, Puccica I, Di Furia M, Salvatorelli A, et al. Complications of postlaparoscopic sleeve gastric resection: review of surgical technique. Minerva Chir. 2019 Jun;74(3):213-217.
34. Knudsen NV, M?ller MH. Association of mortality with out-of-hours admission in patients with perforated peptic ulcer. Acta Anaesthesiol Scand 2015; 59: 248-254.
35. Menekse E, Kocer B, Topcu R, Olmez A, Tez M, Kayaalp C. A practical scoring system to predict mortality in patients with perforated peptic ulcer. World J Emerg Surg 2015 Feb 21;10:7.
36. Hernandez MC, Thorn MJ, Kong VY, Aho JM, Jenkins DH, Bruce JL, et al. Validation of the AAST EGS grading system for perforated peptic ulcer disease. Surgery 2018;164(4):738-745.
37. Anbalakan K, Chua D, Pandya GJ, Shelat VG. Five year experience in management of perforated peptic ulcer and validation of common mortality risk prediction models - are existing models sufficient? A retrospective cohort study. Int J Surg 2015; 14: 38-44.
38. M?ller MH, Adamsen S, Thomsen RW, M?ller AM. Peptic Ulcer Perforation (PULP) trial group. Multicentre trial of a perioperative protocol to reduce mortality in patients with peptic ulcer perforation. Br J Surg. 2011;98:802–10.
39. Teichmann W, Wittmann DH, Andreone PA. Scheduled reoperations (etappenlavage) for diffuse peritonitis. Arch Surg 1986;121: 147–52.
40. Penninckx FM, Kerremans RP, Lauwers PM. Planned relaparotomies in the surgical treatment of severe generalized peritonitis from intestinal origin. World J Surg 1983;7:762–6.
41. Biondo S, Ramos E, Fraccalvieri D, Kreisler E, Ragué JM, Jaurrieta E. Comparative study of left colonic Peritonitis Severity Score and Mannheim Peritonitis Index. Br J Surg. 2006;93(5):616-22.
42. Schietroma M, Cappelli S, Carlei F, Pescosolido A, Lygidakis NJ, Amicucci G. "Acute abdomen": early laparoscopy or active laparotomic-laparoscopic observation? Hepatogastroenterology 2007; 54(76):1137-41.
43. Kologlu M, Elker D, Altun H, Sayek I. Validation of MPI and PIA II in two different groups of patients with secondary peritonitis. Hepatogastroenterology 2001;48(37):147-51.
44. Sawyer RG, Claridge JA, Nathens AB, Rotstein OD, Duane TM, Evans HL, et al. Trial of short-course antimicrobial therapy for intraabdominal infection. N Engl J Med 2015;372: 1996-2005.
45. Schein M, Decker GA. The Hartmann procedure: extended indications in severe intra-abdominal infection. Dis Colon Rectum 1988;31:126–9.
46. Schietroma M, Cecilia EM, De Santis G, Carlei F, Pessia B, Amicucci G. Supplemental Peri-Operative Oxygen and Incision Site Infection after Surgery for Perforated Peptic Ulcer: A Randomized, Double- Blind Monocentric Trial. Surg Infect (Larchmt) 2016;17(1):106-13.
47. Marchese M, Romano L, Giuliani A, Cianca G, Di Sibio A, Carlei F, et al. A case of intrasplenic displacement of an endoscopic doublepigtail stent as a treatment for laparoscopic sleeve gastrectomy leak [published correction appears in Int J Surg Case Rep. 2019;56:49]. Int J Surg Case Rep. 2018;53:367–369.
48. Giuliani A, Romano L, Marchese M, Necozione S, Cianca G, Schietroma M, et al. Gastric leak after laparoscopic sleeve gastrectomy: management with endoscopic double pigtail drainage. A systematic review. Surg Obes Relat Dis 2019;15(8):1414-1419.
49. Schietroma M, Pessia B, Carlei F, Amicucci G. Septic complications after pancreatoduodenectomy for pancreatic adenocarcinoma: are increased gut permeability and inflammatory serum markers responsible? Pancreas 2016;45(9):e47-8.
50. Schein M, Saadia R, Freinkel Z, Decker GA. Aggressive treatment of severe diffuse peritonitis: a prospective study. Br J Surg 1988;75: 173–6.
51. Knaus WA, Zimmerman JE, Wagner DP, Draper EA, Lawrence DE. APACHE –acute physiology and chronic health evaluation: a physiologically based classification system. Crit Care Med 1981; 9: 591–7.
52. Knaus WA, Draper EA, Wagner DP, Zimmerman JE. APACHE II: a severity of disease classification system. Crit Care Med 1985;13: 818–29.
53. Le Gall J-R, Loirat P, Alperovitch A. Simplified acute physiological score for intensive care patients. Lancet 1983; ii: 741.
54. Le Gall JR, Loirat P, Alperovitch A, Glaser P, Granthil C, Mathieu D, et al. A simplified acute physiology score for ICU patients. Crit Care Med 1984;12: 975–7.
55. Elebute EA, Stoner HB. The grading of sepsis. Br J Surg 1983; 70: 29–31.
56. Goris RJ, te Boekhorst TP, Nuytinck JK, Gimbr?re JS. Multiple-organ failure: generalized autodestructive inflammation? Arch Surg 1985; 120: 1109–15.
57. Muralidhar V A, Madhu CP, Sudhir S, Madhu S. Mannheim peritonitis index –prediction of risk of death from peritonitis: construction of a statistical and validation of an empirically based index. Theoretical Surgery 1987;1: 169–77.
58. Imrie CW, Benjamin IS, Ferguson JC, McKay AJ, Mackenzie I, O'Neill J, et al. A single-centre double-blind trial of Trasylol therapy in primary acute pancreatitis. Br J Surg 1978;65: 337–41.
59. Bosscha K, Reijnders K, Hulstaert PF, Algra A, van der Werken C. Prognostic scoring system to predict outcome in peritonitis and intra-abdominal sepsis. BJS 1997;84:1532-4.
60. Sartelli M, Abu-Zidan FM, Catena F, Griffiths EA, Di Saverio S, et al. Global validation of the WSES Sepsis Severity Score for patients with complicated intra-abdominal infections: a prospective multicentre study (WISS Study). World J Emerg Surg 2015;10:61.
61. Knaus WA, Wagner DP, Draper EA, Zimmerman JE, Bergner M, Bastos PG, et al. The APACHE III prognostic system. Risk prediction of hospital mortality for critically ill hospitalized adults. Chest 1991; 100:1619–36.
62. Prytherch DR, Whiteley MS, Higgins B, Weaver PC, Prout WG, Powell SJ. POSSUM and Portsmouth-POSSUM for predicting mortality. Br J Surg. 1998; 85: 1217-1220.
63. Bohnen J, Boulanger M, Meakins JL, McLean AP. Prognosis in generalized peritonitis. Relation to cause and risk factors. Arch Surg. 1983;118(3):285–290.
64. Meakins JL, Solomkin JS, Allo MD, Dellinger EP, Howard RJ, Simmons RL. A proposed classification of intra-abdominal infections. Stratification of etiology and risk for future therapeutic trials. Arch Surg 1984;119(12):1372–1378.
65. Dellinger EP, Wertz MJ, Meakins JL, Solomkin JS, Allo MD, Howard RJ, et al. Surgical infection stratification system for intra-abdominal infection. Multicenter trial. Arch Surg 1985;120(1):21–29.
66. Giuliani A, Romano L, Coletti G, Walid A Fatayer M, Calvisi G, Maffione F, et al. Lymphangiomatosis of the ileum with perforation: A case report and review of the literature. Ann Med Surg (Lond) 2019;41:6-10.
67. Giuliani A, Romano L, Papale E, et al. Post-surgical abdominal damage: management and treatment with vacuum therapy and biological mesh. Chirurgia 2019;32:275-9.
68. Arici C, Mesci A, Dincer D, Dinckan A, Colak T. Analysis of risk factors predicting (affecting) mortality and morbidity of peptic ulcer perforations. Int Surg 2007; 92(3):147–154.
69. Forsmo HM, Glomsaker T, Vandvik PO. Perforated peptic ulcer - a 12-year material. Tidsskr Nor Laegeforen 2005; 125(13): 1822–1824.
70. Kim JM, Jeong SH, Lee YJ, Park ST, Choi SK, Hong SC, et al. Analysis of risk factors for postoperative morbidity in perforated peptic ulcer. Journal of gastric cancer 2012; 12(1):26–35.
71. Kujath P, Schwandner O, Bruch HP. Morbidity and mortality of perforated peptic gastroduodenal ulcer following emergency surgery. Langenbecks Arch Surg 2002; 387(7–8):298–302.
72. Lohsiriwat V, Prapasrivorakul S, Lohsiriwat D. Perforated peptic ulcer: clinical presentation, surgical outcomes, and the accuracy of the Boey scoring system in predicting postoperative morbidity and mortality. World J Surg 2009; 33(1):80–85.
73. Mishra A, Sharma D, Raina VK. A simplified prognostic scoring system for peptic ulcer perforation in developing countries. Indian J Gastroenterol 2003; 22(2):49–53.
74. Rajesh V, Chandra SS, Smile SR. Risk factors predicting operative mortality in perforated peptic ulcer disease. Trop Gastroenterol 2003; 24(3):148–150.
75. Buck DL, Vester-Andersen M, Møller MH. Accuracy of clinical prediction rules in peptic ulcer perforation: an observational study. Scand J Gastroenterol 2012;47(1):28-35.
Full Text Sources:
Abstract:
Views: 7022

Methods: We included 183 patients with diagnosis of complicated PPU. We defined categories T (Temperature), N (Neutrophils count) and M (MOF); then, patients were grouped in stages (0-IV). Variables analysed were age, sex, ASA, blood transfusion, causes of sepsis, temperature, neutrophils count, preoperative organ failure, immune-compromised status, stage (0-IV).
Results: Patients were grouped as follows: none at stage 0; 6 at stage I; 72 at stage II, 72 at stage III; 33 at stage IV. ASA score, neutrophils count, preoperative organ failure, stage IIIIV emerged as statistically significant different prognostic factors. ASA score and stage were significant independent predictors of post-operative mortality in multivariate analysis.
Conclusion: Our proposed system could define and help to assess the mortality risk.
INTRODUCTION
Peptic ulcer disease, both duodenal and gastric, despite the widespread availability of effective acid reduction agents and antibiotic therapy for Helicobacter pylori (1), is associated with potentially life-threatening complications, including bleeding, perforation, penetration and obstruction. Intraabdominal sepsis (IAS) after perforation is the second most frequent complication after bleeding (2,3). A high risk for morbidity (20-50%) and mortality (1.3-40%) is encountered in surgically treated perforated peptic ulcer (PPU) patients (4-9). Patients with PPU present with signs of sepsis and by a careful preoperative assessment of the patients’ severity grade, appropriate management can be offered to achieve an optimal outcome of disease (10,11). Many scoring systems (Boey score, Peptic Ulcer Perforated (PULP) Score, American Society of Anaesthesiologists (ASA) score) have been proposed to predict mortality after PPU (12-14). PULP score seems to be the most reliable, but it is very complex to use (12). Boey score is easier but its predictability value is not consistent (12,15-17). ASA score is a general surgical risk score not intended for PPU patients in particular, and its major drawback is its subjective assessment (12,15). Nowadays, in the clinical practice the grading systems are not always employed for PPU, although they seem to give precise clinical indications, because some of them are too complicated (PULP score) and others are too aspecific (ASA score). In our work, we tried to assess the severity of IAS as a complication of PPU using a new TNM score: T indicates Temperature, N Neutrophil count and M Multiple organ failure (MOF) (18,19). In this study we aimed to evaluate significance of this score to predict mortality of patients with complicated PPU.
MATERIALS AND METHODS
The TNM system was studied in 183 patients with complicated PPU and IAS, managed in General Surgery and Hepato-biliopancreatic Surgery at our Department of Surgery in the period between April 2012 and December 2019. Pregnant women, patients aged < 18, immune-compromised patients and those who underwent laparoscopic surgery were excluded.

At the presentation, patients were clinically evaluated; blood tests and imaging exams were performed. Intravenous antibiotic therapy was set up: Ciprofloxacin 200 mg or Amoxicillin-clavulanic 2 gr and Metronidazole 500 mg.
The anthropometric data were collected in an electronic database. According to clinical and laboratory characteristics, the patients were classified based on our system. Table 1 resumes the definitions. The classes of the patients is showed in table 2, which also shows the groupings in stages (stage 0-IV).
For the study of this system, we used retrospective data of 102 patients between January 2001 and January 2012 (control group); the study group was prospectively evaluated. TNM stage was firstly evaluated at the time of the presentation and then every day of recovery. The primary endpoint was to assess the efficacy of TNM score in forecasting mortality at 30 days. The work has been reported in line with the STROCSS criteria (20).
Statistical analysis
The characteristics of the study sample were analysed with descriptive statistics; the discrete and nominal variables were expressed using frequencies and percentages; for continuous variables, medians and range were reported. The frequency distribution of prognostic factors (age classes, sex, ASA score, blood transfusion, causes of sepsis, fever, neutrophil count, pre-operative organ failure, immuno-compromised status, TNM stage) were examined between outcome groups (alive or dead). Chi square ( 2) test was used to analyse statistical differences. Variables significantly different between the two groups were introduced in the multivariate logistic model to obtain independent predictors of death, with associations reported as odds ratios (ORs) and 95% confidence intervals (CIs). Model discrimination was evaluated using the receiver operating characteristics (ROC) curve. All data were electronically recorded; statistical analyses were performed using the Stata Statistical Software (Release 15/IC, College Station, TX: Stata Corp LP). All the tests were two-tailed, and p < 0.05 was considered statistically significant.
RESULTS
One hundred eighty-three consecutive patients were included; they had a mean age of 67.0 years (range 23 to 86). No significative differences of age between the sexes was reported. One hundred and seventeen patients (63.9%) were diagnosed with localized peritonitis or abscesses and sixty-six (36%) with generalized peritonitis.

The model has a good predictive power being the area under the ROC curve equal to 0.8058 (standard error 0.0342) (fig. 1). In the control group retrospectively analysed death occurred in 33.3% of patients, with no significant difference from the study group. The mortality increased among stages (13.1% at stage II, 28.5% at stage III and 100% at stage IV).
DISCUSSION
Mortality is a serious complication in PPU. PPU carries a mortality ranging from 1.3% to 40% (4-9,21, 22). The mortality rate is as high as 12%-47% in elderly patients undergoing PPU surgery (23-25). Significant risk factors that lead to death are presence of patients factors (age > 65 years-old, female, underweight, presence of comorbidities, delay in presentation more than 24h, non-steroidal anti-inflammatory or steroid use), disease factors (shock at presentation, elevated urea or creatinine, metabolic acidosis, anemia, hypoalbuminemia), and treatment factors (resection surgery, blood transfusion, intensive care units) (26-33). Several different scoring systems used to predict outcome in PPU can be identified through the literature: the Boey score, the ASA score, the Sepsis score (SS), the Charlson Comorbidity Index (CCI), the Mannheim Peritonitis Index (MPI), the Acute Physiology and Chronic Health Evaluation II (APACHE II), the Simplified Acute Physiology score II (SAPS II), the Physiology and Operative Severity Score for the Enumeration of Mortality and Morbidity Physical Sub-score ( POSSUMphys score), the Mortality Probability Models II (MPM II), the PULP score, the Hacettepe score (HS), the Jabalpur score (JS), the Practical Scoring System of Mortality in Patients with Perforated Peptic Ulcer (POMPP) score, and the American Association for the Surgery of Trauma (AAST) Emergency General Surgical (EGS) grading system (AAST EGS grade) (34-37).
Anbalakan K. et al have validated ASA score, Boey’s score, MPI and PULP score and found that all the four systems have moderate accuracy of predicting mortality with area under the receiver operator curve of 72%- 77.2% (37). Other scoring systems are not widely used due a lack of validation or their complexity in clinical use. Our new scoring system (18,19) is simple to use and it seems to be a good predictor of mortality. We believe that the initial TNM stage can be easily adopted in the clinical practice to predict the surgical mortality of PPU patients. Early detection of patients at higher risk could be useful to choose other treatment strategies except surgery to decrease the risk of mortality. More consistent and careful perioperative cares should be adopted, among which respiratory support, circulatory stabilization and frequent monitorization (12,38). To early stage patients, a simple grading system may provide reduction in mortality rates.
The death rates related to complicated IAS is reported to be about 1% (39), 6.7% (40) up to 60% (41-50). The most important variable to explains the difference could be the heterogeneous population of patients and procedures (41,43,51-62). Both the anatomic source of infections and the physiologic impairment affect the outcome (63-67). In our present study we selected a homogeneous sample with the same diagnosis (complicated peptic ulcer), same operation (urgent open repair), same surgical incision (midline laparotomy).
Our results showed that TNM could help to classify patients based on their mortality risk. Moreover, some variables seem to be related to mortality: TNM stages III-IV, ASA score III-IV, neutrophil count and preoperative organ failure. Multivariate analysis, in fact, showed that TNM stage IV and ASA score IV themselves significantly influenced the mortality. Indeed, 90.9% (30/33) of the patients at stage IV died, and the high mortality rate (100%) for M2 patients was mainly reported for patients in the first period of the study (retrospective analysis), when treatment was still not so aggressive as in the last cases considered.
Our grading systems is simple and it allows a reevaluation of the patients based on the clinical picture. Some limitations have to be underlined. The prolonged period of data collection and the small sample size are the main ones, because these factors may influence the evaluation of the TNM. Indeed, our study population was only 183 patients, but this number was noticeable when compared with other studies in the literature (6,68-75), except cohort study of Møller 12 and the study of Hernandez (36). A large-scale clinical trial should be evaluated.
CONCLUSION
In our preliminary study, we want to describe our results about the use of TNM score to assess IAS after PPU. This “transfer” of TNM from cancer pathology to septic pathology could prove, if other studies confirm our results, to be extremely effective to define the mortality risk in patients with IAS after PPU.
Supported and conflict of interest statement
The authors declare no dedicated source of funding and no conflicts of interest related to this publication.
Ethics approval and consent to participate
This is an observational clinical study, so ethics approval is not required. Informed consent was obtained from all individual participants included in the study.
Competing interest
The authors declare that they have no conflict of interest.
Funding: No funding.
Authors contribution
M.S. and F.C. provided study conception and design. B.P., L.R., A.G. have acquired the data. A.M. analysed and interpreted these data. L.R. drafted the manuscript. All authors revised, read and approved the final manuscript.
REFERENCES
1. Testini M, Portincasa P, Piccinni G, Lissidini G, Pellegrini F, Greco L. Significant factors associated with fatal outcome in emergency open surgery for perforated peptic ulcer. World J Gastroenterol. 2003; 9(10):2338-40.
2. Milosavljevic T, Kostic-Milosavljevic M, Jovanovic I, Krstic M. Complications of peptic ulcer disease. Dig Dis 2011; 29(5):491–493.
3. Beatrice P, Lucia R, Antonio G, Domenico G, Mario S, Francesco C, et al. Rare case of upper gastrointestinal bleeding: Dieulafoy' s lesion of duodenum. A case report. Ann Med Surg (Lond). 2019; 45:19-21.
4. Christensen S, Riis A, Norgaard M, Sørensen HT, Thomsen RW. Short-term mortality after perforated or bleeding peptic ulcer among elderly patients: a population-based cohort study. BMC Geriatr 2007; 7:8.
5. Christiansen C, Christensen S, Riis A, Thomsen RW, Johnsen SP, Tonnesen E, et al. Antipsychotic drugs and short-term mortality after peptic ulcer perforation: a population-based cohort study. Aliment Pharmacol Ther 2008;28(7):895–902.
6. Thorsen K, Glomsaker TB, von Meer A, Soreide K, Soreide JA. Trends in diagnosis and surgical management of patients with perforated peptic ulcer. J Gastrointest Surg 2011;15(8):1329–1335.
7. Bertleff MJ, Lange JF. Perforated peptic ulcer disease: a review of history and treatment. Dig Surg 2010;27(3):161–169.
8. Lau JY, Sung J, Hill C, Henderson C, Howden CW, Metz DC. Systematic review of the epidemiology of complicated peptic ulcer disease: incidence, recurrence, risk factors and mortality. Digestion 2011;84(2):102–113.
9. Bae S, Shim KN, Kim N, Kang JM, Kim DS, Kim KM, et al. Incidence and short-term mortality from perforated peptic ulcer in korea: a population-based study. J Epidemiol 2012;22(6):508–516.
10. Moller MH, Shah K, Bendix J, Jensen AG, Zimmermann-Nielsen E, Adamsen S, et al. Risk factors in patients surgically treated for peptic ulcer perforation. Scand J Gastroenterol 2009; 44(2):145–152.
11. Moller MH, Adamsen S, Thomsen RW, Moller AM. Multicentre trial of a perioperative protocol to reduce mortality in patients with peptic ulcer perforation. Br J Surg 2011; 98(6):802–810.
12. Møller MH, Engebjerg MC, Adamsen S, Bendix J, Thomsen RW. The Peptic Ulcer Perforation (PULP) score: a predictor of mortality following peptic ulcer perforation. A cohort study. Acta Anaesthesiol Scand. 2012; 56:655–62.
13. Boey J, Choi SK, Poon A, Alagaratnam TT. Risk stratification in perforated duodenal ulcers. A prospective validation of predictive factors. Ann Surg 1987;205:22–6.
14. Mäkelä JT, Kiviniemi H, Ohtonen P, Laitinen SO. Factors that predict morbidity and mortality in patients with perforated peptic ulcers. Eur J Surg 2002;168:446–51.
15. Thorsen K, Søreide JA, Søreide K. Scoring systems for outcome prediction in patients with perforated peptic ulcer. Scand J Trauma Resusc Emerg Med 2013;21:25.
16. Thorsen K, Søreide JA, Søreide K. What is the best predictor of mortality in perforated peptic ulcer disease? A population-based, multivariable regression analysis including three clinical scoring systems. J Gastrointest Surg 2014;18:1261–8.
17. Mishra A, Sharma D, Raina VK. A simplified prognostic scoring system for peptic ulcer perforation in developing countries. Indian J Gastroenterol 2003;22:49–53.
18. Schietroma M, Pessia B, Mattei A, Romano L, Giuliani A, Carlei F. Temperature-Neutrophils-Multiple Organ Failure Grading for Complicated Intra-Abdominal Infections. Surg Infect (Larchmt) 2020;21:69-74.
19. Schietroma M, Romano L, Pessia B, Mattei A, Fiasca F, Carlei F, et al. TNM: a simple classification system for complicated intraabdominal sepsis after acute appendicitis [published online ahead of print, 2020 Aug 6]. Minerva Chir. 2020;10.23736/S0026- 4733.20.08274-7.
20. Agha R, Abdall-Razak A, Crossley E, Dowlut N, Iosifidis C, Mathew G; STROCSS Group. STROCSS 2019 Guideline: Strengthening the reporting of cohort studies in surgery. Int J Surg 2019;72:156-165.
21. Hermansson M, Staël von Holstein C, Zilling T. Surgical approach and prognostic factors after peptic ulcer perforation. Eur J Surg 1999; 165: 566-572.
22. Rajesh V, Chandra SS, Smile SR. Risk factors predicting operative mortality in perforated peptic ulcer disease. Trop Gastroenterol 2003; 24: 148-150.
23. Blomgren LG. Perforated peptic ulcer: long-term results after simple closure in the elderly. World J Surg 1997; 21:412-414.
24. Svanes C, Salvesen H, Stangeland L, Svanes K, Søreide O. Perforated peptic ulcer over 56 years. Time trends in patients and disease characteristics. Gut 1993; 34:1666-1671.
25. Bulut OB, Rasmussen C, Fischer A. Acute surgical treatment of complicated peptic ulcers with special reference to the elderly. World J Surg 1996; 20: 574-577.
26. Buck DL, Møller MH. Influence of body mass index on mortality after surgery for perforated peptic ulcer. Br J Surg 2014; 101: 993-999.
27. Noguiera C, Silva AS, Santos JN, Silva AG, Ferreira J, Matos E, et al. Perforated peptic ulcer: main factors of morbidity and mortality. World J Surg 2003; 27:782-787.
28. Agrez MV, Henry DA, Senthiselvan S, Duggan JM. Changing trends in perforated peptic ulcer during the past 45 years. Aust N Z J Surg 1992; 62:729-732.
29. Svanes C, Lie RT, Lie SA, Kv?le G, Svanes K, S?reide O. Survival after peptic ulcer perforation: a time trend analysis. J Clin Epidemiol 1996; 49:1363-1371.
30. Walt R, Katschinski B, Logan R, Ashley J, Langman M. Rising frequency of ulcer perforation in elderly people in the United Kingdom. Lancet 1986; 1: 489-492.
31. Kocer B, Surmeli S, Solak C, Unal B, Bozkurt B, Yildirim O, et al. Factors affecting mortality and morbidity in patients with peptic ulcer perforation. J Gastroenterol Hepatol 2007;22:565-570.
32. Schietroma M, Colozzi S, Romano L, Pessia B, Giuliani A, Vicentini V, et al. Short- and long-term results after laparoscopic floppy Nissen fundoplication in elderly versus non-elderly patients. J Minim Access Surg. 2020;16(3):256 263.
33. Giuliani A, Romano L, Papale E, Puccica I, Di Furia M, Salvatorelli A, et al. Complications of postlaparoscopic sleeve gastric resection: review of surgical technique. Minerva Chir. 2019 Jun;74(3):213-217.
34. Knudsen NV, M?ller MH. Association of mortality with out-of-hours admission in patients with perforated peptic ulcer. Acta Anaesthesiol Scand 2015; 59: 248-254.
35. Menekse E, Kocer B, Topcu R, Olmez A, Tez M, Kayaalp C. A practical scoring system to predict mortality in patients with perforated peptic ulcer. World J Emerg Surg 2015 Feb 21;10:7.
36. Hernandez MC, Thorn MJ, Kong VY, Aho JM, Jenkins DH, Bruce JL, et al. Validation of the AAST EGS grading system for perforated peptic ulcer disease. Surgery 2018;164(4):738-745.
37. Anbalakan K, Chua D, Pandya GJ, Shelat VG. Five year experience in management of perforated peptic ulcer and validation of common mortality risk prediction models - are existing models sufficient? A retrospective cohort study. Int J Surg 2015; 14: 38-44.
38. M?ller MH, Adamsen S, Thomsen RW, M?ller AM. Peptic Ulcer Perforation (PULP) trial group. Multicentre trial of a perioperative protocol to reduce mortality in patients with peptic ulcer perforation. Br J Surg. 2011;98:802–10.
39. Teichmann W, Wittmann DH, Andreone PA. Scheduled reoperations (etappenlavage) for diffuse peritonitis. Arch Surg 1986;121: 147–52.
40. Penninckx FM, Kerremans RP, Lauwers PM. Planned relaparotomies in the surgical treatment of severe generalized peritonitis from intestinal origin. World J Surg 1983;7:762–6.
41. Biondo S, Ramos E, Fraccalvieri D, Kreisler E, Ragué JM, Jaurrieta E. Comparative study of left colonic Peritonitis Severity Score and Mannheim Peritonitis Index. Br J Surg. 2006;93(5):616-22.
42. Schietroma M, Cappelli S, Carlei F, Pescosolido A, Lygidakis NJ, Amicucci G. "Acute abdomen": early laparoscopy or active laparotomic-laparoscopic observation? Hepatogastroenterology 2007; 54(76):1137-41.
43. Kologlu M, Elker D, Altun H, Sayek I. Validation of MPI and PIA II in two different groups of patients with secondary peritonitis. Hepatogastroenterology 2001;48(37):147-51.
44. Sawyer RG, Claridge JA, Nathens AB, Rotstein OD, Duane TM, Evans HL, et al. Trial of short-course antimicrobial therapy for intraabdominal infection. N Engl J Med 2015;372: 1996-2005.
45. Schein M, Decker GA. The Hartmann procedure: extended indications in severe intra-abdominal infection. Dis Colon Rectum 1988;31:126–9.
46. Schietroma M, Cecilia EM, De Santis G, Carlei F, Pessia B, Amicucci G. Supplemental Peri-Operative Oxygen and Incision Site Infection after Surgery for Perforated Peptic Ulcer: A Randomized, Double- Blind Monocentric Trial. Surg Infect (Larchmt) 2016;17(1):106-13.
47. Marchese M, Romano L, Giuliani A, Cianca G, Di Sibio A, Carlei F, et al. A case of intrasplenic displacement of an endoscopic doublepigtail stent as a treatment for laparoscopic sleeve gastrectomy leak [published correction appears in Int J Surg Case Rep. 2019;56:49]. Int J Surg Case Rep. 2018;53:367–369.
48. Giuliani A, Romano L, Marchese M, Necozione S, Cianca G, Schietroma M, et al. Gastric leak after laparoscopic sleeve gastrectomy: management with endoscopic double pigtail drainage. A systematic review. Surg Obes Relat Dis 2019;15(8):1414-1419.
49. Schietroma M, Pessia B, Carlei F, Amicucci G. Septic complications after pancreatoduodenectomy for pancreatic adenocarcinoma: are increased gut permeability and inflammatory serum markers responsible? Pancreas 2016;45(9):e47-8.
50. Schein M, Saadia R, Freinkel Z, Decker GA. Aggressive treatment of severe diffuse peritonitis: a prospective study. Br J Surg 1988;75: 173–6.
51. Knaus WA, Zimmerman JE, Wagner DP, Draper EA, Lawrence DE. APACHE –acute physiology and chronic health evaluation: a physiologically based classification system. Crit Care Med 1981; 9: 591–7.
52. Knaus WA, Draper EA, Wagner DP, Zimmerman JE. APACHE II: a severity of disease classification system. Crit Care Med 1985;13: 818–29.
53. Le Gall J-R, Loirat P, Alperovitch A. Simplified acute physiological score for intensive care patients. Lancet 1983; ii: 741.
54. Le Gall JR, Loirat P, Alperovitch A, Glaser P, Granthil C, Mathieu D, et al. A simplified acute physiology score for ICU patients. Crit Care Med 1984;12: 975–7.
55. Elebute EA, Stoner HB. The grading of sepsis. Br J Surg 1983; 70: 29–31.
56. Goris RJ, te Boekhorst TP, Nuytinck JK, Gimbr?re JS. Multiple-organ failure: generalized autodestructive inflammation? Arch Surg 1985; 120: 1109–15.
57. Muralidhar V A, Madhu CP, Sudhir S, Madhu S. Mannheim peritonitis index –prediction of risk of death from peritonitis: construction of a statistical and validation of an empirically based index. Theoretical Surgery 1987;1: 169–77.
58. Imrie CW, Benjamin IS, Ferguson JC, McKay AJ, Mackenzie I, O'Neill J, et al. A single-centre double-blind trial of Trasylol therapy in primary acute pancreatitis. Br J Surg 1978;65: 337–41.
59. Bosscha K, Reijnders K, Hulstaert PF, Algra A, van der Werken C. Prognostic scoring system to predict outcome in peritonitis and intra-abdominal sepsis. BJS 1997;84:1532-4.
60. Sartelli M, Abu-Zidan FM, Catena F, Griffiths EA, Di Saverio S, et al. Global validation of the WSES Sepsis Severity Score for patients with complicated intra-abdominal infections: a prospective multicentre study (WISS Study). World J Emerg Surg 2015;10:61.
61. Knaus WA, Wagner DP, Draper EA, Zimmerman JE, Bergner M, Bastos PG, et al. The APACHE III prognostic system. Risk prediction of hospital mortality for critically ill hospitalized adults. Chest 1991; 100:1619–36.
62. Prytherch DR, Whiteley MS, Higgins B, Weaver PC, Prout WG, Powell SJ. POSSUM and Portsmouth-POSSUM for predicting mortality. Br J Surg. 1998; 85: 1217-1220.
63. Bohnen J, Boulanger M, Meakins JL, McLean AP. Prognosis in generalized peritonitis. Relation to cause and risk factors. Arch Surg. 1983;118(3):285–290.
64. Meakins JL, Solomkin JS, Allo MD, Dellinger EP, Howard RJ, Simmons RL. A proposed classification of intra-abdominal infections. Stratification of etiology and risk for future therapeutic trials. Arch Surg 1984;119(12):1372–1378.
65. Dellinger EP, Wertz MJ, Meakins JL, Solomkin JS, Allo MD, Howard RJ, et al. Surgical infection stratification system for intra-abdominal infection. Multicenter trial. Arch Surg 1985;120(1):21–29.
66. Giuliani A, Romano L, Coletti G, Walid A Fatayer M, Calvisi G, Maffione F, et al. Lymphangiomatosis of the ileum with perforation: A case report and review of the literature. Ann Med Surg (Lond) 2019;41:6-10.
67. Giuliani A, Romano L, Papale E, et al. Post-surgical abdominal damage: management and treatment with vacuum therapy and biological mesh. Chirurgia 2019;32:275-9.
68. Arici C, Mesci A, Dincer D, Dinckan A, Colak T. Analysis of risk factors predicting (affecting) mortality and morbidity of peptic ulcer perforations. Int Surg 2007; 92(3):147–154.
69. Forsmo HM, Glomsaker T, Vandvik PO. Perforated peptic ulcer - a 12-year material. Tidsskr Nor Laegeforen 2005; 125(13): 1822–1824.
70. Kim JM, Jeong SH, Lee YJ, Park ST, Choi SK, Hong SC, et al. Analysis of risk factors for postoperative morbidity in perforated peptic ulcer. Journal of gastric cancer 2012; 12(1):26–35.
71. Kujath P, Schwandner O, Bruch HP. Morbidity and mortality of perforated peptic gastroduodenal ulcer following emergency surgery. Langenbecks Arch Surg 2002; 387(7–8):298–302.
72. Lohsiriwat V, Prapasrivorakul S, Lohsiriwat D. Perforated peptic ulcer: clinical presentation, surgical outcomes, and the accuracy of the Boey scoring system in predicting postoperative morbidity and mortality. World J Surg 2009; 33(1):80–85.
73. Mishra A, Sharma D, Raina VK. A simplified prognostic scoring system for peptic ulcer perforation in developing countries. Indian J Gastroenterol 2003; 22(2):49–53.
74. Rajesh V, Chandra SS, Smile SR. Risk factors predicting operative mortality in perforated peptic ulcer disease. Trop Gastroenterol 2003; 24(3):148–150.
75. Buck DL, Vester-Andersen M, Møller MH. Accuracy of clinical prediction rules in peptic ulcer perforation: an observational study. Scand J Gastroenterol 2012;47(1):28-35.
Full Text Sources:
Abstract:
Views: 7022

Watch Video Articles
For Authors
Journal Subscriptions

Current Issue
Dec 2025
Supplements
Instructions for authors
Online submission
Contact
ISSN: 2559 - 723X (print)
e-ISSN: 2601 - 1700 (online)
ISSN-L: 2559 - 723X
Journal Abbreviation: Surg. Gastroenterol. Oncol.
Surgery, Gastroenterology and Oncology (SGO) is indexed in:
e-ISSN: 2601 - 1700 (online)
ISSN-L: 2559 - 723X
Journal Abbreviation: Surg. Gastroenterol. Oncol.
Surgery, Gastroenterology and Oncology (SGO) is indexed in:
- SCOPUS
- EBSCO
- DOI/Crossref
- Google Scholar
- SCImago
- Harvard Library
- Open Academic Journals Index (OAJI)
Open Access Statement
Surgery, Gastroenterology and Oncology (SGO) is an open-access, peer-reviewed online journal published by Celsius Publishing House. The journal allows readers to read, download, copy, distribute, print, search, or link to the full text of its articles.
Surgery, Gastroenterology and Oncology (SGO) is an open-access, peer-reviewed online journal published by Celsius Publishing House. The journal allows readers to read, download, copy, distribute, print, search, or link to the full text of its articles.
Journal Metrics
Time to first editorial decision: 25 days
Rejection rate: 61%
CiteScore: 0.2
Time to first editorial decision: 25 days
Rejection rate: 61%
CiteScore: 0.2

Meetings and Courses in 2025
Meetings and Courses in 2024
Meetings and Courses in 2023
Meetings and Courses in 2022
Meetings and Courses in 2021
Meetings and Courses in 2020
Meetings and Courses in 2019
Verona expert meeting 2019

Surgery, Gastroenterology and Oncology applies the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits readers to copy and redistribute the material in any medium or format, remix, adapt, build upon the published works non-commercially, and license the derivative works on different terms, provided the original material is properly cited and the use is non-commercial. Please see: https://creativecommons.org/licenses/by-nc/4.0/
Publisher’s Note:
The opinions, statements, and data contained in article are solely those of the authors and not of Surgery, Gastroenterology and Oncology journal or the editors. Publisher and the editors disclaim responsibility for any damage resulting from any ideas, instructions, methods, or products referred to in the content.
 IASGO Society News
IASGO Society News