Surgery, Gastroenterology and Oncology
|
|
Methodology: A prospective comparative study was conducted at Ain Shams university hospitals between January 2017 and May 2021 including 80 cases of VAH with single defect 10 cm. The cases were divided into 2 groups; group (I) underwent ACS and group (II) underwent TAR.
Results: A significantly higher intraoperative failure rate was seen in the ACS group (20% versus 2.5%). TAR showed significantly lower rates of early postoperative wound complications (65% versus 22.5%) and recurrence (22.5% versus 2.5%) after 1 year.
Conclusion: TAR is a more effective technique in treatment of large VAH than ACS, with fewer postoperative complication and lower short-term recurrence.
INTRODUCTION
Ventral abdominal hernia (VAH) repair is considered a surgical challenge especially hernias with large defect size and loss of domain (1-3). The incidence of recurrence after VAH repair ranges from 18.1% in primary VAH to 30.6% in secondary VAH (incisional hernia) (4). The principle of any hernia repair is to achieve a tension free repair with re-enforcement by mesh to decrease the incidence of recurrence (5). Many techniques had been studied and evaluated to address the problem of complex VAH with large defects and loss domain (1,6).
In 1973, Rives et al. described their technique of retro rectus space dissection for incisional hernia (IH) repair allowing medial mobilization of myofascial flap and sub-lay mesh insertion (7, 8). But this technique has limitation in cases of large VAH as it doesn’t allow major myofascial advancement due to lateral limitation of linea semilunaris (LS) (9).
In 1990, Ramirez et al. at described their technique of open anterior component separation (ACS) (10). In this technique, the external oblique aponeurosis is separated from internal oblique lateral to LS to achieve more medial mobilization of myofascial flap (9). ACS was successfully utilized in cases of large VAH, but the technique had its drawbacks. ACS was found to be associated with high rates of wound complications (seroma, wound infection, and flap necrosis) due to excess subcutaneous dissection with the possibility of lateral abdominal wall hernia at area of external oblique incision. The recurrence rate after ACS ranges from 9-18% (4).
In 2012, Novitsky et al. were the first to describe the technique of posterior component separation and transversus abdominis release (PCS-TAR). The technique involved retro rectus space dissection reaching the LS, then an incision is made at posterior rectus sheath to access the space between internal oblique and transversus abdominis muscles. Dissection is carried on in this space until reaching psoas muscle laterally. This technique has the advantage of allowing maximum medial myofascial mobilization without major subcutaneous dissection. In addition, the technique allows for sub-lay mesh placement (11).
All these different component separation techniques aim to reestablish a functional abdominal wall through autologous tissue repair by separating between myofascial layers of anterior abdominal wall (AAW). This allows medial mobilization of myofascial flap to obliterate large size defects (12-14). Due to the relative novelty of these techniques, a knowledge gap still persists regarding which CS technique is suitable to different types of VAH. The aim of our study is to compare the outcomes of TAR with ACS as regard short term postoperative complication and recurrence rate.
PATIENT AND METHODS
Study design
This study was a prospective comparative study conducted at our university hospitals between January 2017 and May 2021. All procedures performed in this study were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. The approval of the ethical committee of general surgery department, Ain Shams University was obtained before the start of the study.
The study included 80 patients with age ranging from 18-65 years old. Our inclusion criteria required that the patients should have VAH with a single midline defect which is ≥ 10 cm [measured by computerized tomography (CT) scan] in diameter.
Exclusion criteria: We excluded patients with lateral defect, previous abdominoplasty, those with loss of domain [identified clinically and by CT scan (15)], patients with recurrence after any type of CS, patients with previous abdominal flap reconstruction or stoma, patients with complicated VAH (obstructed or strangulated), and patients with inflammatory bowel disease.
Patients enrolled in the study were divided sequentially to two groups:
- Group (I): 40 patients treated with ACS.
- Group (II): 40 patients treated with TAR.
The operation intended was thoroughly discussed with all patients and an informed consent was obtained.
Preoperative workup
All patients underwent full clinical assessment in addition to contrast enhanced CT scan of the abdomen and pelvis commenting on defect size, number of defects, loss of domain, previous mesh insertion and associated intraabdominal pathology.
Surgical technique
Transverses abdominis release After midline laparotomy, exploration of abdomen and complete adhesiolysis was done.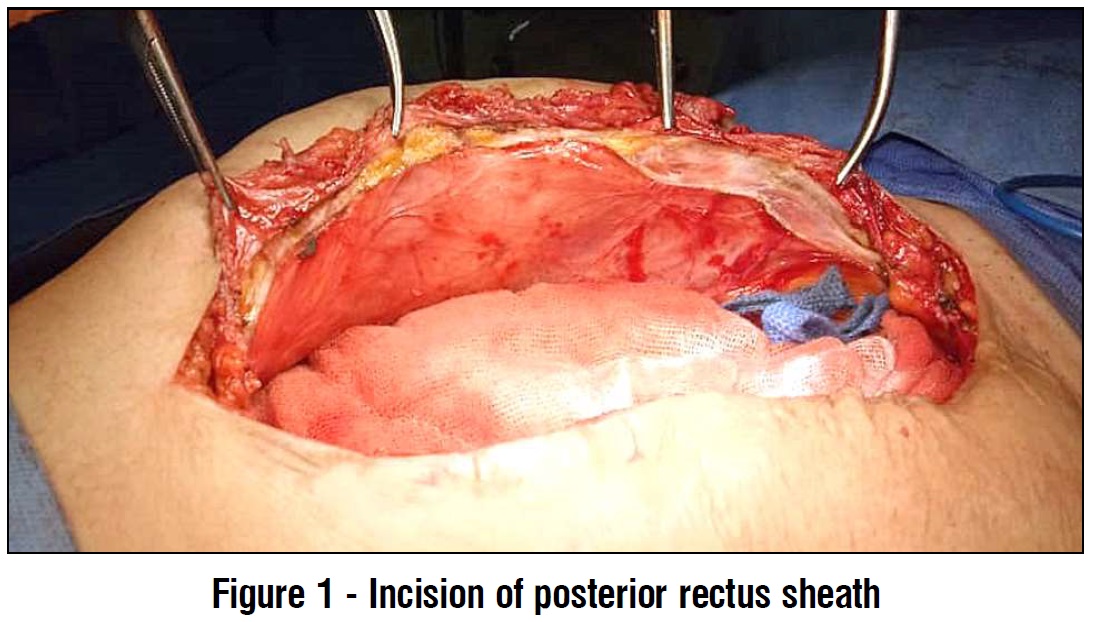 The posterior rectus sheath was incised at one cm lateral linea alba to access the retro rectus space (fig. 1). Blunt dissection of the retro rectus space was done using a peanut gauze until reaching LS laterally (taking care to preserve the neurovascular bundles to rectus muscle) (fig. 2).
The posterior rectus sheath was incised at one cm lateral linea alba to access the retro rectus space (fig. 1). Blunt dissection of the retro rectus space was done using a peanut gauze until reaching LS laterally (taking care to preserve the neurovascular bundles to rectus muscle) (fig. 2).
An incision was made at posterior rectus sheath one cm medial to LS (just medial to the neurovascular bundles) starting at the upper part of abdomen to expose the underneath transversus abdominis muscle (TAM). Then we divided TAM by mono polar diathermy to access the space between TAM and fascia transversalis (fig. 3). Blunt dissection was carried on in this space very gently until reaching the retroperitoneal space laterally exposing the psoas muscle, iliac vessels (fig. 4).

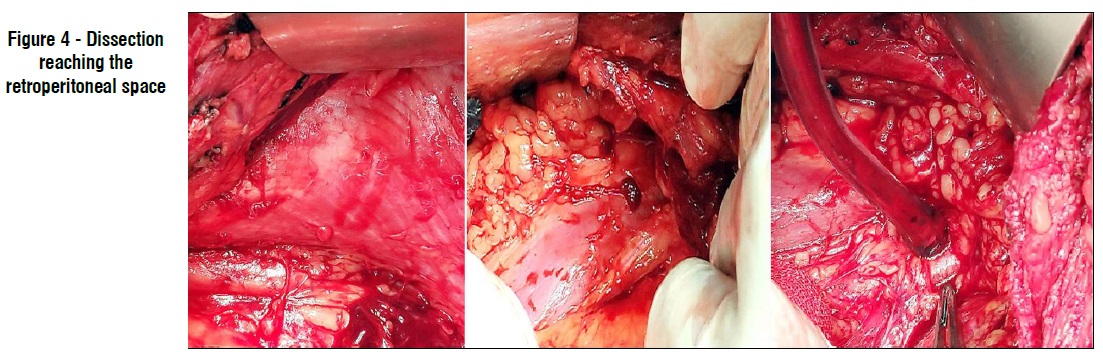
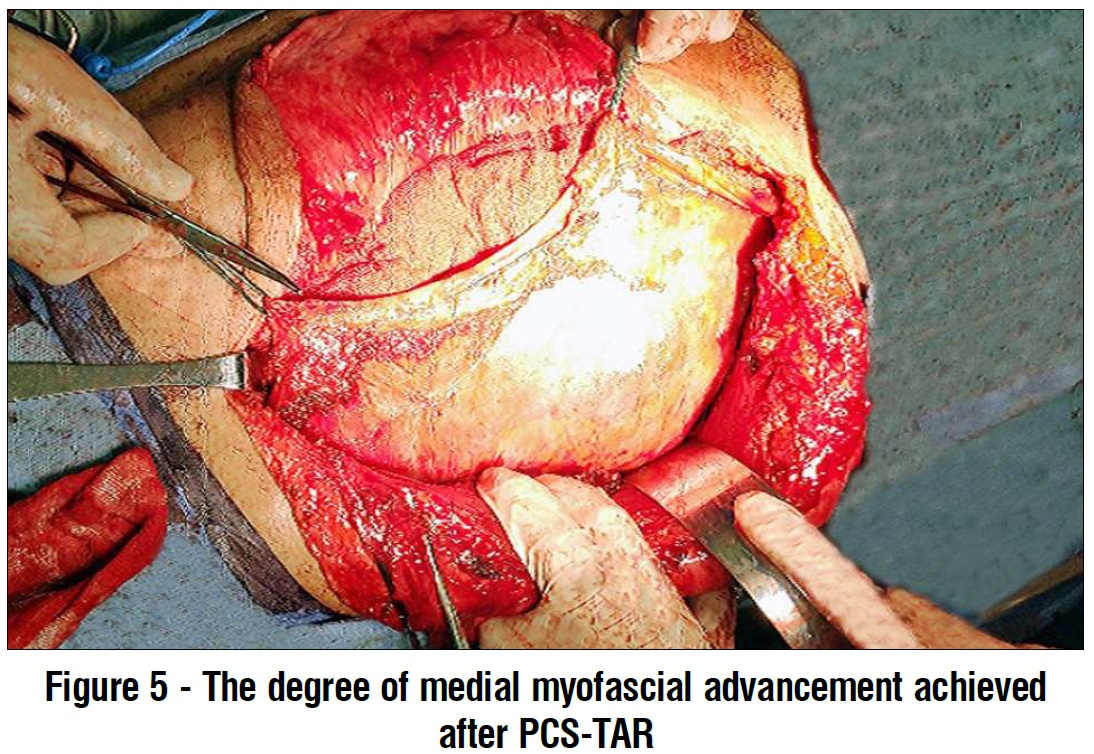
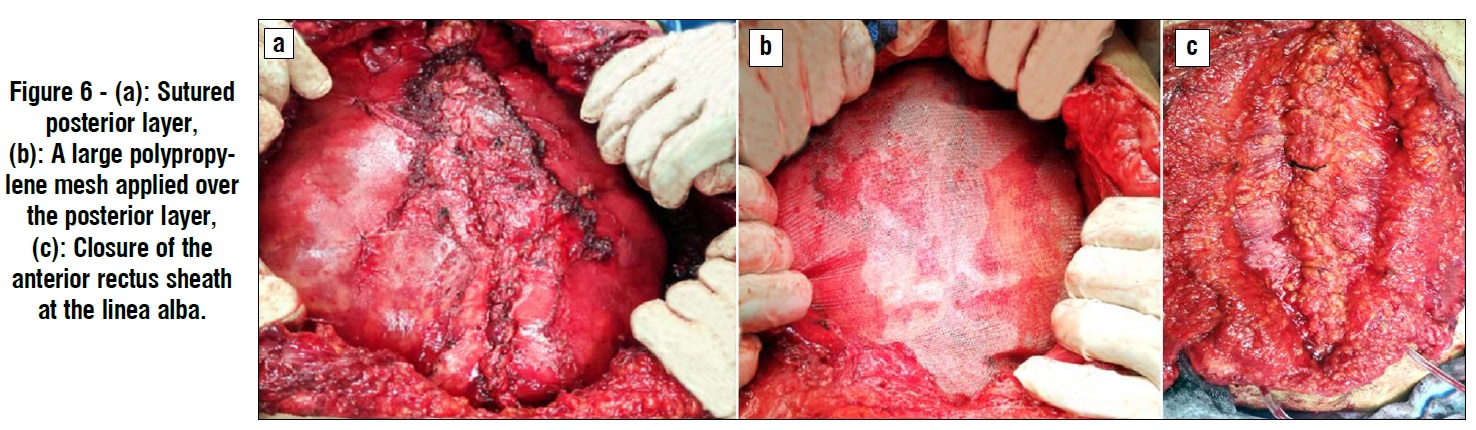
Dissection in the pre-transversalis space was continued inferiorly to space of Retzius (anterior to urinary bladder) exposing the pubic bone and Cooper's ligament, and superiorly to the costal margin and the subxiphoid space. After generous medial myofascial advancement is achieved (fig. 5) bilaterally, re-approximation of posterior rectus sheath at midline was done using Vicryle® (Ethicon) 2/0 continuous suturing (fig. 6a). Holes in the posterior layer are closed with Vicryle® (Ethicon) 2/0 continuous suture. After closure of the posterior layer, we applied a 30 x 30 cm polypropylene mesh (in diamond configuration) (fig. 6b). The mesh was fixed to Cooper's ligaments using by 2/0 Proline® (Ethicon) sutures.
A closed suction drain was inserted in pre-transversalis space on the mesh. Then re-approximation of anterior rectus sheath at linea alba was done with continuous suturing using double polydioxanone (PDS) loop® (Ethicon) No. 1. Then subcutaneous tissue and skin were closed (fig. 6c).
Anterior component separation
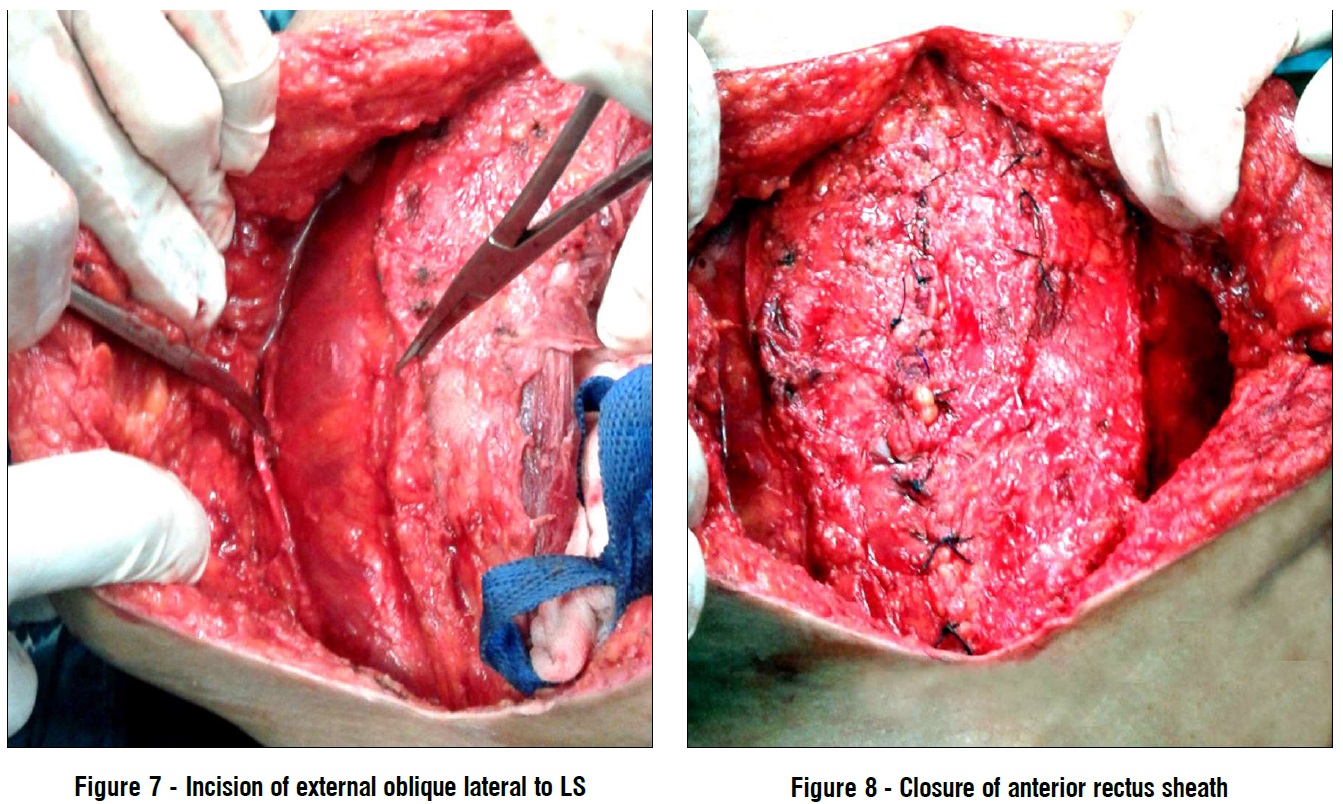
Anterior component separation After laparotomy and adhesiolysis, dissection of skin flap was done until 2 cm lateral to LS. Then incision of the external oblique aponeurosis was done 1 cm lateral to LS starting from the costal margin superiorly to pubic bone inferiorly exposing underlying internal oblique muscle. Blunt dissection was done separating both the external and internal obliques allowing medial myofascial mobilization (fig. 7). Posterior component separation was carried out in the same way described previously in PCS-TAR technique, till reaching the neurovascular bundles medial to LS.
Closure of the anterior rectus sheath at linea alba was done with continuous suturing using double PDS loop® (Ethicon) No. 1 (fig. 8). A large sheet of polypropylene mesh (or even 2 meshes sutured together) was applied to cover the whole area (including the midline and the areas of component septation bilaterally) and fixed using 2/0 proline® (Ethicon) sutures. A closed suction drain was inserted on the mesh. Then sub-cutaneous tissue and skin were closed. In cases we were unable to close the defect without tension using one technique, an attempt to add the other technique either uni or bilaterally was carried on. If this attempt failed, a double face mesh was used as a bridge to aid closure (no cases required this in the study).
Postoperative care
Follow up of patients in the ward was done with administration of intravenous antibiotics, fluids, and potent analgesics. Patient started oral fluids once intestinal sounds are audible. Drains were removed once the output was ≤ 50 ml per day. Patient were followed in the outpatient clinic (OPC) one week after discharge and periodically at 3, 6, and 12 months postoperative. If recurrence was clinically suspected, CT abdomen was done to confirm that.
Collected data included 1) preoperative data: patient demographics, smoking, associated comorbidities (including, diabetes mellitus (D.M), chronic obstructive pulmonary disease (COPD), and hypertension), type of VAH (non-recurrent or recurrent with/without mesh), number of previous abdominal surgeries, previous abdominal cancer, and previous wound infection. 2) Intraoperative data: operative time, defect size (width in cm), intraoperative blood loss, concomitant surgery with hernia repair, visceral or vascular injury, the change in intraoperative airway pressure before and after defect closure and failure of closure (defined as either inability to close the defect or closure under tension with increase in airway pressure more than 40 mmHg). 3) Postoperative data: postoperative hemorrhage, length of hospital stay, time of drains removal, time to regain of gastrointestinal tract (GIT) motility, postoperative wound complication (wound infection, seroma, skin dehiscence and flap necrosis) and recurrence.
Statistics
Data were revised, coded, entered on a computer and analyzed using SPSS package version number 20. Student t-test was used for comparing quantitative variables between two study groups. Chi-square and Fisher exact tests were used to test the association between qualitative variables. P-value ≤ 0.05 was considered significant and P-value ≤ 0.001 was considered highly significant.
RESULTS
Eighty patients were enrolled in the study. Patients were sequentially divided into two groups; group (I): 40 patients treated with ACS and group (II): 40 patients treated with TAR. No statistically significant difference was found between both groups regarding patients' demographics and preoperative characteristics as shown in tables 1 and 2.




Two patients had another concomitant surgery in addition to VAH repair (one patient in group (I) underwent open cholecystectomy and one patient in group (II) underwent open cholecystectomy and left oophorectomy.
The study had shown that the difference in mean operative time between both groups was not statistically significant (215.4±52.4 minutes in group (I) versus 226±51.9 minutes in group (II). Also, no statistically significant difference was found between both groups regarding the mean intraoperative blood loss (420±157.2 ml in group (I) versus 452±151.7 ml in group (II). We usually measure the intraoperative airway pressure change (before and after defect closure), to see if the closure caused a rise in intraabdominal pressure. A statistically significant difference in intraoperative airway pressure change was found between both groups in favor of PCS-TAR group as shown in fig. 9.
Failure (either inability to close the defect or closure with increase of airway pressure ≥40 mmHg) was encountered in nine patients. Eight of them were in the ACS group (20% of the group patients), and one in the PCS-TAR group (2.5% of the group patients). This difference was statistically significant. Operative data are shown in table 3.
The mean length of the period required for drain removal showed a highly significant difference in favor of the PCS-TAR group (14.5±5.4 days in group (I) versus 10.3±4.7 days in group (II)). Despite that, the mean length of hospital stay showed no statistically significant difference between both groups (4.7±1.4 days in group (I) versus 5.2±1.7 days in group (II)) (table 4).
The overall rate of early postoperative wound complication showed a difference in favor of PCS-TAR with the difference being statistically significant (it was significant in rates of seroma and wound infection and non-significant in rates of flap necrosis and wound dehiscence). These data are shown in fig. 10 and table 5. All cases of wound complication were treated with parenteral antibiotics and dressing, with no further operative intervention needed.
After one year of follow up, ten cases of recurrence of the VAH were recorded. Nine patients (22.5%) had recurrence after ACS and one patient (2.5%) had recurrence after PCS-TAR, with the difference being highly statistically significant. Recurrent cases after ACS were planned for repair by PCS-TAR and the recurrent case after PCS-TAR was planned for repair by laparoscopic intraperitoneal on lay mesh repair (IPOM). Postoperative data are shown in table 4.


DISCUSSION
Large VAH is a complex and difficult surgical problem, with high morbidity and risk of recurrence. The aim of VAH repair is to restore the functional abdominal wall with prosthetic reinforcement (11). PCS-TAR technique first described by Novitsky et al. aims to achieve significant medial advancement of the posterior fascial layer by dissection in a potential and safe space between the transversus abdominis muscle and fascia transversalis with peritoneum (11). This space is devoid of neurovascular bundles (which pass in the space between TAM and internal oblique muscle). This technique allows for creation of a large space (reaching laterally to the retroperitoneum) for tension free repair plus mesh placement (16, 17). Moreover, applying the mesh in this space restores the native biomechanics of abdominal wall (11). We conducted this study to compare between ACS and TAR when utilized in the repair of large VAH. Our study did not show a difference in technical difficulty between both types of repairs. This was shown by demonstrating no significant difference between both groups regarding the operative time and the intraoperative blood loss. This similarity may be attributed to the fact that both procedures mostly have the same steps except the plane of lateral myofascial dissection (subcutaneous in ACS and pre-transversalis in PCS-TAR).

Most of the published literature reflected a similar degree of technical difficulty for these techniques. For PCS-TAR, Novitsky et al., reported a mean operative time of 235 minutes and Krpata et al. reported a mean operative time of 228 minutes (both of which are close to our time) (11,18). On the other hand, a far longer mean operative time (383 minutes) was reported by Appleton et al. in their study (a case series on PCS-TAR) (19). The longer mean operative time in Appleton et al.'s study may be related to the small sample size (12 patients), or the fact that 41.7% of them had previous VAH repair (versus only 7.5% in our study). Also, Appleton et al.'s study, included three cases of concomitant major surgery (radical cystectomy with ileal conduit and pelvic lymphadenectomy, completion proctectomy with end ileostomy and Hartman procedure) (19). We had only one simple concomitant procedure in each of our study groups.
In Krpata et al.'s study (a retrospective comparative study between ACS and PCS-TAR), the mean operative time for ACS technique was 285 min which is longer than ours. This longer operative time is mostly because they did panniculectomy in 37.5% of ACS (18).
The PCS-TAR demonstrated a better efficacy in closure of large abdominal than ACS. This was shown by the significantly lower failure rate in our study (2.5% versus 20%). This may be explained by the larger distance of flap mobilization achieved by the technique (may reach more than 10 cm on each side).
This efficacy of PCS-TAR was reported by other authors in their studies. Novitsky et al. and Appleton et al. both reported no cases of failure in their previously mentioned studies (11,19).
In our study, the PCS-TAR technique showed a significantly lower postoperative wound complication rate than ACS technique (22.5% versus 65%). This may be explained by fact that PCS-TAR avoids excessive subcutaneous dissection that accompanies ACS. This in turn makes it less likely to compromise the perforating vessels supplying the overlying tissue. Also, the subcutaneous mesh placement in ACS increases the rate of seroma formation in comparison to the pre-transversalis placement in PCS-TAR (11).
Krpata et al. reported a wound complication rate of 25.5% in PCS-TAR group versus 48.2% in ACS group in their study (18). In Holihan et al. study (a retrospective comparative study comparing between PCS-TAR, ACS and other component separation techniques operated by multiple different surgical teams), they reported a wound complication rate of 29.2% in PCS-TAR group in comparison to 32% in ACS group (20). While both studies demonstrated lower wound complication rates after PCS-TAR similar to us, their wound complication rates after ACS were significantly lower than ours. This may be explained by the fact that both Krpata et al. and Holihan et al. studies used light weight synthetic and biological meshes in their ACS repairs, in contrast to the heavy weight synthetic mesh used in all our patients. Also, Krpata et al. performed panniculectomy in 37.5% of their ACS cases (decreasing the size of the flaps and hence the chance of developing complications) (18,20).
Our study had shown a significantly lower recurrence rate following PCS-TAR after one year of follow-up. This difference may be secondary to lower postoperative wound complication rates after PCS-TAR compared to ACS. Also, the fact that PCS-TAR results in a lower mean intrabdominal tension (as demonstrated by the measurement of airway pressure in our study) may contribute to this.
Previously published data mostly support our results. Novitsky et al. reported a recurrence rate was 4.7% after PCS-TAR, with a mean follow up duration of 26.1 months. This recurrence rate is marginally higher than ours but with longer follow-up duration (11). Appleton et al., did not record any case of recurrence after PCS-TAR in their study, with a median follow up of twenty-four months (19). Their results are significantly better than ours with a longer follow-up duration. Appleton's study population were generally of low mean BMI & non-smokers, so this may contribute their excellent results (11,19).
The recurrence rate after ACS in Holihan et al.'s study was lower than ours (16% versus 22.5%). This may be attributed to lower incidence of wound complications in their study (32% versus 65% in ours), and to the fact that the ratio of patients with associated comorbidities and smokers was lower in their study (20).
On the other hand, Holihan et al.'s study reported a high recurrence rate after PCS-TAR reaching 20.8%. Their study population had a high incidence of previous wound infection (12.5% versus 5% in our study), and previous VAH repair (70.8% versus 7.5% in our study), which may explain their results. The results of Holihan et al. study may indicate that they are more experienced in ACS than PCS-TAR (20).
Our study has its limitations. First, the follow up duration was relatively short to properly address the problem of recurrence which may occur after a long period. Second, the study was comparative but not randomized and not multicenter (it is our experience in our center), limiting its power to represent a wider type of practice. At last, our study did not include VAH with multiple defects, those with stoma, and complicated cases. These types of patients could have changed the results if they were included. In the future, larger, multicenter, randomized controlled trials with longer follow up duration are needed for more solid results.
CONCLUSION
TAR is a more effective technique in treatment of large ventral abdominal hernia than ACS, with fewer postoperative complication and lower short-term recurrence.
Authors, contributions
• Study conception and design: all authors;
• Acquisition of data: Hossam S Abdelrahim, Ehab Mohammed Ali Fadl;
• Analysis and interpretation of data: Haitham M. Elmaleh, Mohammed H Goda;
• Drafting of manuscript: Hossam S Abdelrahim, Ahmed F Amer;
• Critical revision of manuscript: all authors.
Funding and Financial support
This research did not receive any specific grant from funding agencies in the public, commercial or notfor- profit sectors.
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
IRB approval number: 0006379. Clinical trial registration number: PACTR202109549974384 (retrospectively registered).
REFERENCES
1. Sleiwah A, McAllister S. Posterior components separation with transversus abdominis release (TAR) versus alternative techniques of components separation: systematic review. Int Surg. 2018;103 (7-8):355–365.
2. Mudge M, Hughes LE. Incisional hernia: a 10 year prospective study of incidence and attitudes. Br J Surg. 1985;72(1):70-1.
3. Ng JK, Parnianpour M, Richardson CA, Kippers V. Functional roles of abdominal and back muscles during isometric axial rotation of the trunk. J Orthop Res. 2001;19(3):463-71.
4. Hodgkinson JD, Leo CA, Maeda Y, Bassett P, Oke SM, Vaizey CJ, et al. A meta-analysis comparing open anterior component separation with posterior component separation and transversus abdominis release in the repair of midline ventral hernias. Hernia. 2018;22(4): 617-626.
5. Pereira JA, Bravo-Salva A, Montcusí B, Pérez-Farre S, Fresno de Prado L, LópezCano M. Incisional hernia recurrence after open elective repair: expertise in abdominal wall surgery matters. BMC Surg. 2019;19(1):103.
6. Ventral Hernia Working Group, Breuing K, Butler CE, Ferzoco S, Franz M, Hultman CS, Kilbridge JF, et al. Incisional ventral hernias: review of the literature and recommendations regarding the grading and technique of repair. Surgery. 2010;148(3):544-58.
7. Stoppa RE. The treatment of complicated groin and incisional hernias. World J Surg. 1989;13(5):545-54.
8. Rives J, Pire JC, Flament JB, Palot JP, Body C. Treatment of large eventrations. New therapeutic indications apropos of 322 cases. Chirurgie.1985;111(3):215-25.
9. Siegal SR, Pauli EM. Posterior component separation/transversus abdominis release. Plast Aesthet Res. 2019;6:25.
10. Ramirez OM, Ruas E, Dellon AL. "Components separation" method for closure of abdominal-wall defects: an anatomic and clinical study. Plast Reconstr Surg. 1990;86(3):519-26.
11. Novitsky YW, Elliott HL, Orenstein SB, Rosen MJ. Transversus abdominis muscle release: a novel approach to posterior component separation during complex abdominal wall reconstruction. Am J Surg. 2012;204(5):709-16.
12. Scheuerlein H, Thiessen A, Schug-Pass C, Köckerling F. What do we know about component separation techniques for abdominal wall rernia repair? Front Surg. 2018;5:24.
13. Nockolds CL, Hodde JP, Rooney PS. Abdominal wall reconstruction with components separation and mesh reinforcement in complex hernia repair. BMC Surg. 2014;14:25.
14. Bleichrodt RP, de Vries Reilingh TS, Malyar A, van Goor H, Hansson B, van der Kolk B. Component separation technique to repair large midline hernias. Operative Techniques in General Surgery. 2004; 6(3):179-188.
15. Tanaka EY, Yoo JH, Rodrigues AJ Jr, Utiyama EM, Birolini D, Rasslan S. A computerized tomography scan method for calculating the hernia sac and abdominal cavity volume in complex large incisional hernia with loss of domain. Hernia. 2010;14(1):63-9.
16. Carbonell AM, Cobb WS, Chen SM. Posterior components separation during retromuscular hernia repair. Hernia. 2008;12(4): 359-62.
17. Jones CM, Winder JS, Potochny JD, Pauli EM. Posterior Component Separation with Transversus Abdominis Release: Technique, Utility, and Outcomes in Complex Abdominal Wall Reconstruction. Plast Reconstr Surg. 2016;137(2):636-646.
18. Krpata DM, Blatnik JA, Novitsky YW, Rosen MJ. Posterior and open anterior components separations: a comparative analysis. Am J Surg; 2012, 203(3):31822; discussion 322.
19. Appleton ND, Anderson KD, Hancock K, Scott MH, Walsh CJ. Initial UK experience with transversus abdominis muscle release for posterior components separation in abdominal wall reconstruction of large or complex ventral hernias: a combined approach by general and plastic surgeons. Ann R Coll Surg Engl. 2017;99(4):265-270.
20. Holihan JL, Askenasy EP, Greenberg JA, Keith JN, Martindale RG, Roth JS, et al. Ventral Hernia Outcome Collaboration Writing Group. Component Separation vs. Bridged Repair for Large Ventral Hernias: A Multi-Institutional Risk-Adjusted Comparison, Systematic Review, and MetaAnalysis. Surg Infect (Larchmt). 2016;17(1):17-26.
Full Text Sources:
Abstract:
Views: 7217

For Authors
Journal Subscriptions

Dec 2025
Supplements
Instructions for authors
Online submission
Contact
e-ISSN: 2601 - 1700 (online)
ISSN-L: 2559 - 723X
Journal Abbreviation: Surg. Gastroenterol. Oncol.
Surgery, Gastroenterology and Oncology (SGO) is indexed in:
- SCOPUS
- EBSCO
- DOI/Crossref
- Google Scholar
- SCImago
- Harvard Library
- Open Academic Journals Index (OAJI)
Surgery, Gastroenterology and Oncology (SGO) is an open-access, peer-reviewed online journal published by Celsius Publishing House. The journal allows readers to read, download, copy, distribute, print, search, or link to the full text of its articles.
Time to first editorial decision: 25 days
Rejection rate: 61%
CiteScore: 0.2

Meetings and Courses in 2025
Meetings and Courses in 2024
Meetings and Courses in 2023
Meetings and Courses in 2022
Meetings and Courses in 2021
Meetings and Courses in 2020
Meetings and Courses in 2019
Verona expert meeting 2019

Surgery, Gastroenterology and Oncology applies the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits readers to copy and redistribute the material in any medium or format, remix, adapt, build upon the published works non-commercially, and license the derivative works on different terms, provided the original material is properly cited and the use is non-commercial. Please see: https://creativecommons.org/licenses/by-nc/4.0/
Publisher’s Note:
The opinions, statements, and data contained in article are solely those of the authors and not of Surgery, Gastroenterology and Oncology journal or the editors. Publisher and the editors disclaim responsibility for any damage resulting from any ideas, instructions, methods, or products referred to in the content.
 IASGO Society News
IASGO Society News