Surgery, Gastroenterology and Oncology

|
|
Vascular Endothelial Growth Factor (VEGF-C) as a Perioperative Serum Biomarker for Gastric Cancer Prognostication
Alok Dubey1, Pravindhira Sozhamadevi Narendhira1, Arunkumar Ramachandran2,
Arun Kasiviswanathan1, Sugumar Chidambaranathan1, Nagnath babu O L1, Chitra Ayyappan2, Karthick Rajendran2, Priyadarshini Panneerselvam2, Muthukumaran Rajaram2
1Institute of Surgical Gastroenterology, Madras Medical College, Tamil Nadu, Chennai, India
2Multidisciplinary Research Unit, Madras Medical College, Tamil Nadu, Chennai, India
ABSTRACT
Background: Vascular Endothelial Growth Factor (VEGF) plays a critical role in tumor angiogenesis, progression, and metastasis. Despite advances in gastric cancer treatment, the prognosis remains poor, necessitating better biomarkers for risk stratification and assessing tumor biology to provide individualised care. This study evaluates VEGF-C as a prognostic biomarker in patients with gastric carcinoma.
Methods: A prospective cohort of 46 histologically confirmed gastric carcinoma patients was studied. Serum VEGF-C levels were measured preoperatively and post-operatively using ELISA. Clinicopathological features and survival data were collected and analyzed using SPSS. Perioperative VEGF-C levels were correlated with patient-tumor characteristics and survival outcomes using t-tests, correlation analysis, and Kaplan-Meier survival curves.
Results: Most patients were male (78.3%) and over 60 years old (54.3%). A majority had distal gastric tumors (69.6%) and stage IV disease (43.5%). Elevated preoperative VEGF-C was significantly associated with lymphatic involvement (p < 0.001), systemic metastasis (p = 0.012), and poorer survival. VEGF-C levels significantly decreased post-surgery (mean difference: 437.67 pg/mL, p < 0.001). Kaplan–Meier analysis revealed patients with greater fall in VEGF-C levels (> 50% ) post-surgery had a better median survival (17 months) compared to those with no/poor fall in VEGF-C levels (17 months) (Log-rank p = 0.013).
Conclusion: Increased serum VEGF-C levels is significantly associated with aggressive tumor biology and adverse prognosis in gastric carcinoma patient. Monitoring VEGF-C levels in the perioperative period may guide therapeutic decisions and predict clinical outcomes.
INTRODUCTION
Gastric cancer remains one of the leading causes of cancer-related mortality globally, particularly in East and South Asia (1). In India, it accounts for the fifth most common malignancy in adult males and seventh most common cancer in adult females (2) Despite advancements in surgical techniques and adjuvant therapies, survival rates remain dismal largely due to its high metastatic potential, delayed presentation with advanced stage and delayed diagnosis (3). Tumor angiogenesis is essential for cancer growth and metastasis. Vascular Endothelial Growth Factor (VEGF) is a key angiogenic factor secreted by tumor cells promoting endothelial proliferation, vascular permeability, and neovascularization (4). Elevated VEGF expression has been reported in various malignancies, especially gastric cancer, and is associated with tumor aggressiveness and poor prognosis (5). Several studies have explored tissue-based VEGF expression using immunohistochemistry (IHC), yet serum-based VEGF measurements offer a minimally invasive alternative with potential for dynamic monitoring (6). VEGF-C has been overexpressed in 30-60% of gastric cancer patients and increased serum levels has been associated with poor prognosis (7). In this study, we evaluated the preoperative and post-operative levels of serum VEGF-C in gastric cancer patients to determine their association with clinicopathological features, and prognostic implications for survival.
Figure 1 - Study Flow diagram
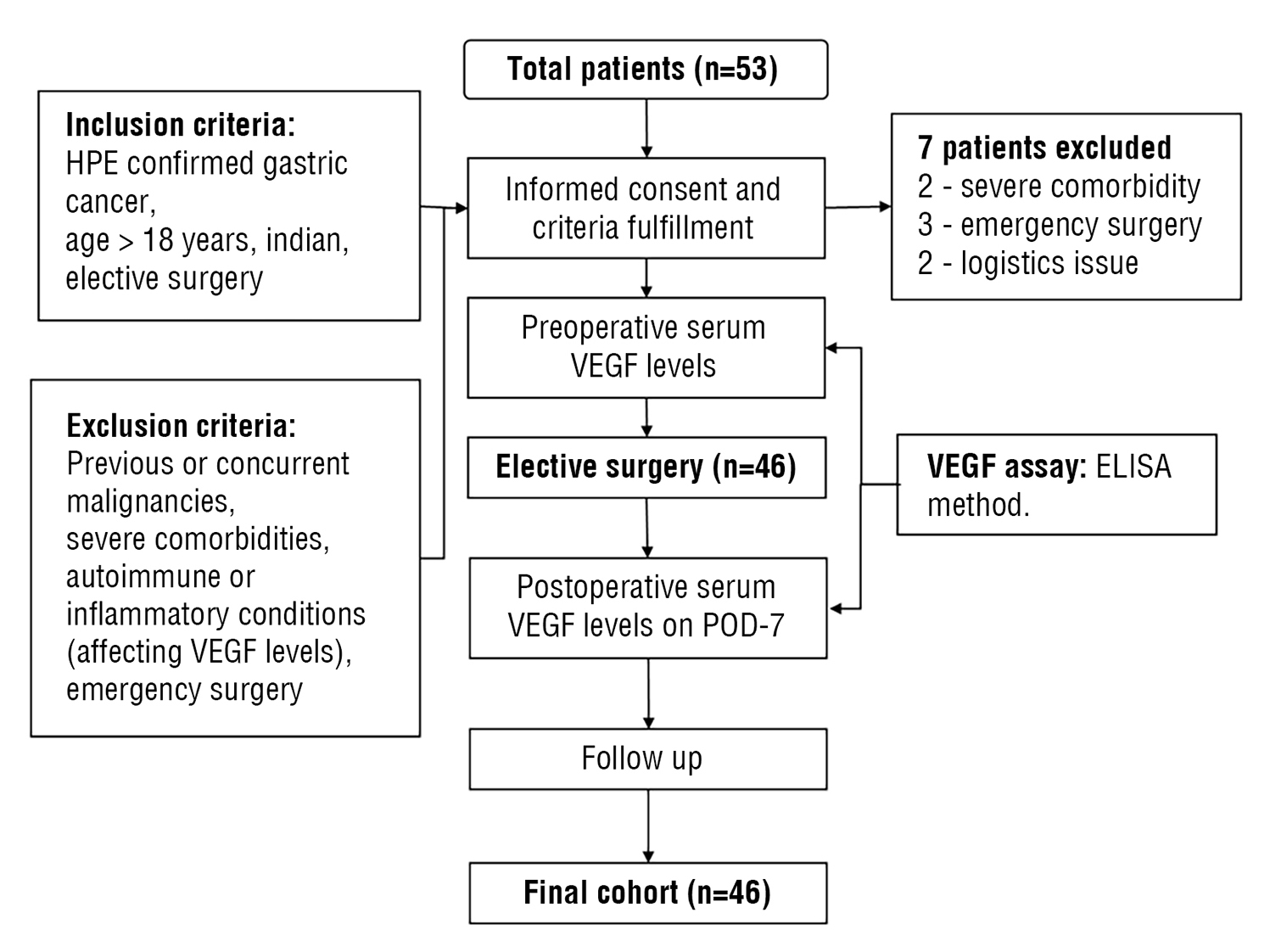
MATERIALS AND METHODS
Study Design and Participants
This prospective observational study was conducted on 46 patients with histopathologically confirmed gastric carcinoma at the Institute of Surgical Gastroenterology, Madras Medical College, Rajiv Gandhi Government General Hospital, Chennai – 600003 between July 2022 to June 2025 (including a two year follow up for survival analysis). Indian Patients aged >18 years, who underwent elective surgery were included. Patients with previous / concurrent history of other malignancies, emergency procedures, severe comorbidities, auto-immune and inflammatory conditions are excluded.
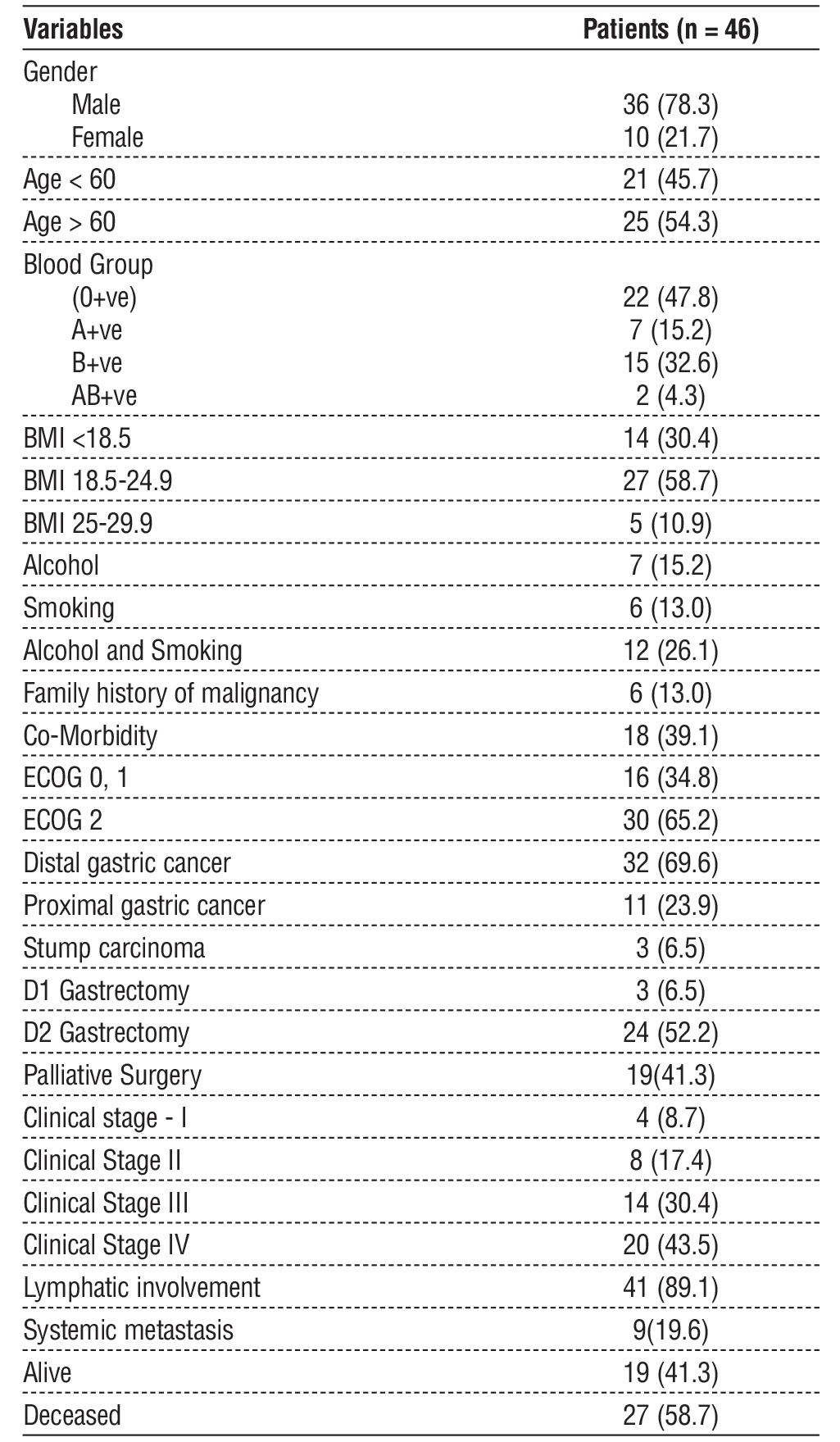
Table 1 - Demographics and clinicopathological features of patients
Ethical Approval
The study was approved by the Institutional Ethics Committee (IEC no. 16072022) and a written informed consent was obtained from all participants.
Data Collection
Demographic and clinical details including age, sex, BMI, blood group, comorbidities, past treatment
history, alcohol/smoking history, ECOG performance status, tumor location, staging, intraoperative findings, procedure details and postoperative recovery were documented and patients were followed up as shown by the study flow diagram (fig. 1).
VEGF Assay
Blood samples were collected from the patients preoperatively on the day before surgery and post-
operatively on postoperative day 7. Serum VEGF-C levels were measured using a commercially available ELISA kit (Elbascience Human VEGF-C ELISA) according to manufacturer instructions. All samples were run in duplicate.
Statistical Analysis
Data were analyzed using SPSS v26. Continuous variables were expressed as mean ± SD, and categorical variables as frequency (%). Paired t-tests were used to compare pre- and post-op VEGF levels. Correlations were assessed using Pearson’s correlation coefficient. Group comparisons were performed using independent t-tests and ANOVA. Survival analysis was conducted using Kaplan-Meier curves, with comparisons via Log-rank, Breslow, and Tarone-Ware tests. A p-value < 0.05 was considered statistically significant.
RESULTS
Demographic and Clinical Profile
Out of 46 patients, 78.3% were male, and 54.3% were over 60 years of age. Most had a normal BMI (58.7%), while 30.4% were underweight. Blood group O+ve was the most common (47.8%). Substance use history included alcohol (15.2%), smoking (13%) or both (26.1%). Comorbidities were present in 39.1% and 13% had a family history of cancer (table 1).
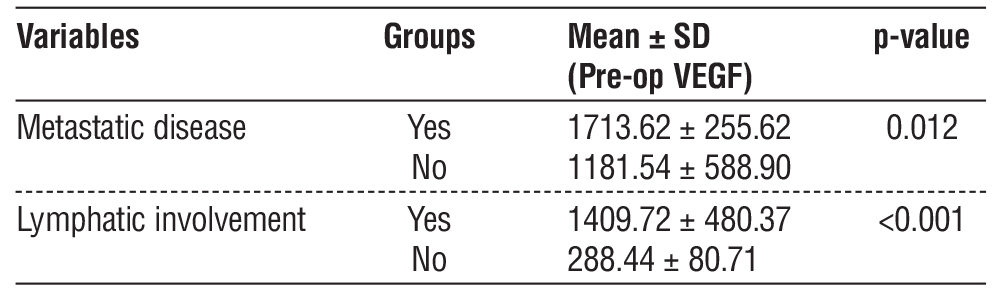
Table 2 - Comparison of metastatic and lymphatic involvements with VEGF levels
Tumor Characteristics
Distal gastric cancer accounted for more than two-third of the cases (69.6%) with a mean pre-operative VEGF-C level of 1287 (± 605) pg/ml. Proximal gastric cancers and stump carcinoma accounted for 23.9 % and 6.5% respectively with mean VEGF-C levels of 1292 (±559) pg/ml and 1259 (±531) pg/ml respectively. All patients were taken up for elective surgery and among them 52.2% and 6.5% underwent D2 and D1 gastrectomies respectively. As features of advanced (Stage IV) disease were noted intraoperatively in 41.3% of cases, they underwent palliative options of Gastro-jejunostomy or feeding jejunostomy. Postoperative histo-pathological examination revealed that 34.8% had moderately differentiated adenocarcinoma with lymphatic involvement in 89.1%, and systemic metastasis in 19.6% cases as represented in table 1.
VEGF Levels and Surgical Outcome
Preoperative VEGF levels averaged 1285.67 ± 578.53 pg/mL. Patients with metastatic disease had significantly higher preoperative VEGF (1713.62 ± 255.62 pg/mL) than those without metastasis (1181.54 ± 588.90 pg/mL, p = 0.012). VEGF was also markedly elevated in cases with lymphatic involvement (1409.72 ± 480.37 pg/mL vs. 288.44 ± 80.71 pg/mL, p < 0.001) (table 2).
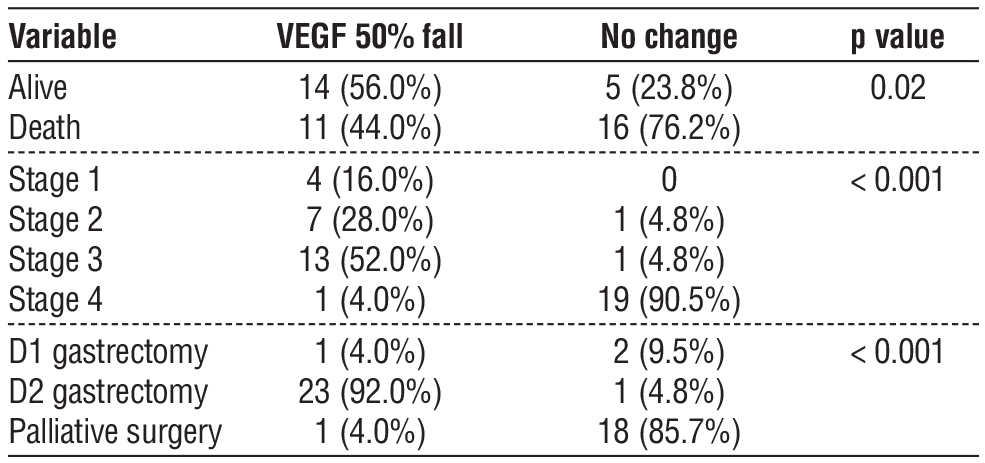
There was significant decline in the postoperative VEGF-C levels for the patients who underwent curative resections (D2 gastrectomy) had a mean fall of 798 pg/ml (more than 50% of their preop values) (table 3).
Table 3 - Comparison table for VEGF fall and other variable
Survival Analysis
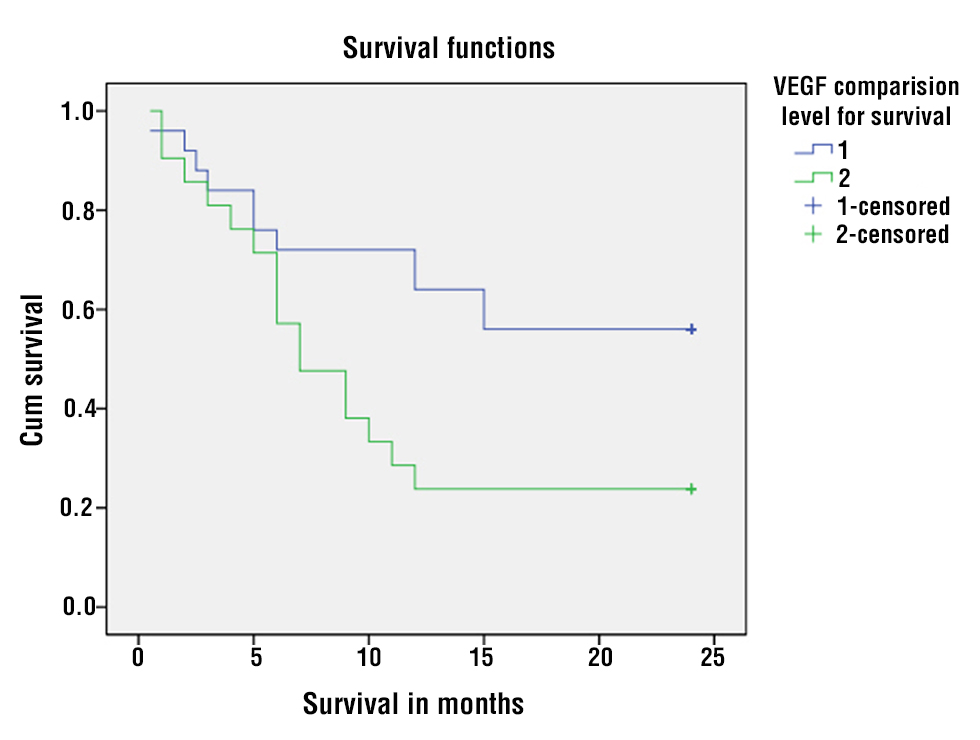
Kaplan-Meier survival analysis (table 4 and fig. 2) revealed significantly poorer survival among patients with high VEGF expression. The patients who presented with advanced stages and underwent palliative options had less than 50% fall in serum VEGF and a median survival of 7 months whereas the patients who underwent a complete curative resection had more than 50% fall in serum VEGF levels and a median survival of 17 months. Event rates were 76.2% and 44%, respectively. Mean survival times were 10.43 ± 1.59 months and 16.56 ± 1.81 months respectively. These findings are summarised in tables 3 and 4. Log-rank (p = 0.013), Breslow (p = 0.043), and Tarone-Ware (p = 0.031) tests confirmed these differences.
Table 4 - Kaplan-Meier survival analysis
Figure 2 - Kaplan–Meier Survival Analysis
DISCUSSION
Previous studies have established VEGF as a central mediator of angiogenesis and a poor prognostic factor in gastric cancer (8,9). Yildirim et al. observed that elevated VEGF levels were predictive of metastasis and recurrence (10). Similarly, Seo et al. reported serum VEGF as an independent predictor of overall survival in gastric carcinoma (11). VEGF family of glycoproteins consists of VEGF-A, VEGF-B, VEGF-C, VEGF-D, PlGF (Placenta Growth factor), VEGF-E, VEGF-F, interacting with specific tyrosine kinase receptors VEGFR - 1,2,3. VEGF-C interacts with VEGFR-3/FLT-4 and play an important role lymphangiogenisis (12). Increased VEGF-C and VEGFR3 expression in histo-pathological examination of gastric cancer patients was associated with increased lymphatic spread (13). Our findings demonstrate that elevated serum VEGF-C levels are significantly associated with aggressive tumor behavior like lymphatic and systemic spread and reduced survival in gastric carcinoma. The postoperative decline in VEGF-C levels (more than 50% of their preop values) reflects the reduction in tumor burden in patients undergoing D1/D2 gastrectomies. The strong correlation between VEGF-C and advanced disease in our cohort underscores its potential utility in preoperative risk stratification. Unlike static histological parameters, serum VEGF-C levels can be dynamically monitored, providing insights into disease progression or therapeutic response. Our survival analysis highlights that high VEGF-C expression correlates with worse outcomes. This supports its potential as a negative prognostic biomarker and highlights the therapeutic promise of targeted therapies (Anti-VEGF) in selected patients (14,15). Significant fall in serum VEGF-C levels (more than 50 % of its preoperative values) in the immediate postoperative period may be useful in assessing adequate tumor clearance and predicting survival as indicated by a better survival compared to those with no fall/ increase in postoperative VEGF-C levels (p = 0.02). Thus perioperative monitoring of VEGF-C levels may serve as a real-time indicator of surgical clearance and treatment response.
CONCLUSION
Increased serum VEGF-C levels are associated with aggressive tumor biology like lymph node involvement, metastasis, and reduced survival in gastric cancer patients establishing the role of VEGF-C as a potential negative biomarker for gastric cancer prognostication. Thus, monitoring the perioperative levels of VEGF-C can aid in the assessment of disease progression, treatment response and surgical clearance and guide therapeutic decision-making, enhancing the personalized care for gastric cancer patients.
Limitations
Our study is limited by its single-center design and relatively small sample size with a short follow up. There was no serial assessment of VEGF-C levels during follow up and the study was not powered to asses other prognostic factors or targeted therapy. Thus future multicentric RCTs with improved sample size and longer follow up is required to validate these findings and establish serial VEGF-C levels monitoring protocols and explore its integration into clinical algorithms.
Author’s Contributions
All authors contributed equally to the study.
Acknowledgement
The Multidisciplinary Research Unit, a unit of the Department of Health Research, Ministry of Health and Family Welfare, New Delhi, India was gratefully acknowledged for supporting this study. We extend our gratitude to the Institute of Surgical Gastro-enterology and Madras Medical College for providing the necessary facilities and support.
Conflict of Interest: None declared.
Funding Statement
This research is supported by the Department of Health Research (DHR), Government of India, under the Multidisciplinary Research Unit (MRU).
REFERENCES
1. Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, et al. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin. 2021;71(3):209-249.
2. Sharma A, Radhakrishnan V. Gastric cancer in India. Indian J Med Paediatr Oncol. 2011;32(1):12-6.
3. Smyth EC, Nilsson M, Grabsch HI, van Grieken NC, Lordick F. Gastric cancer. Lancet. 2020;396(10251):635-648.
4. Ferrara N. VEGF as a therapeutic target in cancer. Oncology. 2005;69 Suppl 3:11-6.
5. Takahashi Y, Kitadai Y, Bucana CD, Cleary KR, Ellis LM. Expression of vascular endothelial growth factor and its receptor, KDR, correlates with vascularity, metastasis, and proliferation of human colon cancer. Cancer Res. 1995;55(18):3964-8.
6. Szajewski M, Ciesielski M, P?ksa R, Kurek P, Sta?czak M, Walczak J, et al. VEGF-C and Lymphatic Vessel Density in Tumor Tissue of Gastric Cancer: Correlations with Pathoclinical Features and Prognosis. Cancers. 2025; 17(9):1406.
7. Laterza MM, Orditura M, Fabozzi A, Ventriglia J, Savastano B, Petrillo A, et al. 660P - Increased Vegf-C Serum Levels are Predictive of a Poor Outcome in Patients with Resectable Gastric Cancer, Annals of Oncology, Volume 25, Supplement 4, 2014, Page iv225, ISSN 0923-7534.
8. Bilgiç CI, Tez M. Serum VEGF levels in gastric cancer patients: correlation with clinicopathological parameters. Turk J Med Sci. 2015;45(1):112-7.
9. Kruszyna ?, Murawa D, Jagodzi?ski PP, Oszkinis G, Krasi?ski Z. The Expression and Prognostic Significance of VEGF and CXCR4 in Gastric Cancer: Correlation with Angiogenesis, Lymphangiogenesis and Progression. Molecular Biology. 2022;44(7):3075-3088.
10. Dai Y, Jiang J, Wang Y, Jin Z, Hu S. The correlation and clinical implication of VEGF-C expression in microvascular density and lymph node metastasis of gastric carcinoma. Am J Transl Res. 2016; 8(12):5741-5747.
11. Karayiannakis AJ, Syrigos KN, Polychronidis A, Zbar A, Kouraklis G, Simopoulos C, et al. Circulating VEGF levels in the serum of gastric cancer patients: correlation with pathological variables, patient survival, and tumor surgery. Ann Surg. 2002;236(1):37-42.
12. Liu W, Xu H, Ying X, Zhang D, Lai L, Wang L, et al. Radiofrequency Ablation (RFA) Combined with Transcatheter Arterial Chemo-embolization (TACE) for Patients with Medium-to-Large Hepatocellular Carcinoma: A Retrospective Analysis of Long-Term Outcome. Med Sci Monit. 2020;26:e923263.
13. Liu XE, Sun XD, Wu JM. Expression and significance of VEGF-C and FLT-4 in gastric cancer. World J Gastroenterol. 2004;10(3): 352-5.
14. Fuchs CS, Tomasek J, Yong CJ, Dumitru F, Passalacqua R, Goswami C, et al. Ramucirumab monotherapy for previously treated gastric cancer. Lancet. 2014;383(9911):31-39.
15. Van Cutsem E, Kang YK, Chung HC, Shen L, Sawaki A, Lordick F, et al. Phase III study of ramucirumab plus paclitaxel in advanced gastric cancer. Lancet Oncol. 2014;15(11):1224–1235.
Full Text Sources:
Abstract:
Views: 616

For Authors
Journal Subscriptions

Dec 2025
Supplements
Instructions for authors
Online submission
Contact
e-ISSN: 2601 - 1700 (online)
ISSN-L: 2559 - 723X
Journal Abbreviation: Surg. Gastroenterol. Oncol.
Surgery, Gastroenterology and Oncology (SGO) is indexed in:
- SCOPUS
- EBSCO
- DOI/Crossref
- Google Scholar
- SCImago
- Harvard Library
- Open Academic Journals Index (OAJI)
Surgery, Gastroenterology and Oncology (SGO) is an open-access, peer-reviewed online journal published by Celsius Publishing House. The journal allows readers to read, download, copy, distribute, print, search, or link to the full text of its articles.
Time to first editorial decision: 25 days
Rejection rate: 61%
CiteScore: 0.2

Meetings and Courses in 2025
Meetings and Courses in 2024
Meetings and Courses in 2023
Meetings and Courses in 2022
Meetings and Courses in 2021
Meetings and Courses in 2020
Meetings and Courses in 2019
Verona expert meeting 2019

Surgery, Gastroenterology and Oncology applies the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits readers to copy and redistribute the material in any medium or format, remix, adapt, build upon the published works non-commercially, and license the derivative works on different terms, provided the original material is properly cited and the use is non-commercial. Please see: https://creativecommons.org/licenses/by-nc/4.0/
Publisher’s Note:
The opinions, statements, and data contained in article are solely those of the authors and not of Surgery, Gastroenterology and Oncology journal or the editors. Publisher and the editors disclaim responsibility for any damage resulting from any ideas, instructions, methods, or products referred to in the content.
 IASGO Society News
IASGO Society News